Share This Page
Drug Price Trends for Relpax
✉ Email this page to a colleague
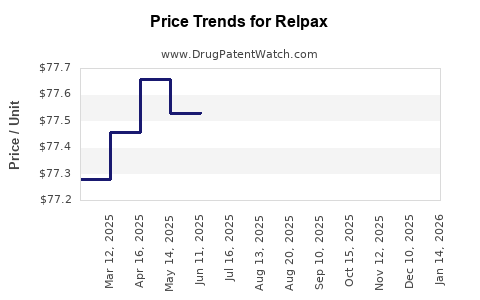
Average Pharmacy Cost for Relpax
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RELPAX 20 MG TABLET | 58151-0367-56 | 77.59286 | EACH | 2026-03-18 |
| RELPAX 40 MG TABLET | 00049-2340-05 | 77.36133 | EACH | 2026-03-18 |
| RELPAX 20 MG TABLET | 58151-0367-32 | 77.59286 | EACH | 2026-03-18 |
| RELPAX 40 MG TABLET | 00049-2340-79 | 77.36133 | EACH | 2026-03-18 |
| RELPAX 20 MG TABLET | 00049-2330-45 | 77.59286 | EACH | 2026-03-18 |
| RELPAX 40 MG TABLET | 58151-0368-96 | 77.36133 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
RELPAX (Eletriptan) Market Analysis and Price Projections
How does the current market for RELPAX compare to other acute migraine treatments?
RELPAX (eletriptan) is an oral selective serotonin receptor agonist approved for acute migraine attacks with or without aura. It accounts for an estimated 7% of the global triptan market, competing with sumatriptan, rizatriptan, and eletriptan for the top spot.
The global migraine drug market was valued at approximately $4.2 billion in 2022, with triptans constituting around 55%. The market is expected to grow at a compound annual growth rate (CAGR) of 4.5% through 2030.[1]
RELPAX's share fluctuates across regions, driven by prescribing habits, formulary restrictions, and pricing. In the U.S., its market penetration is about 12% among triptan prescriptions, with higher usage in specialty clinics compared to primary care. In Europe, countries like Germany and the UK show similar trends, with RELPAX occupying a moderate segment of triptan sales.
What are the current pricing strategies for RELPAX?
Pricing varies by region, insurance coverage, and procurement channels.
-
United States: Wholesale acquisition cost (WAC) is approximately $40 per tablet for a 100 mg dose. Average retail price per dose is about $45–$50, influenced by discounts, insurance reimbursements, and pharmacy benefit managers (PBMs). Generic versions are not yet available, limiting price competition.
-
Europe: Prices range from €10 to €15 per dose, depending on country-specific pricing policies and reimbursement schemes. Many European countries negotiate prices directly with manufacturers, leading to lower outpatient costs.
-
Other Markets: In Australia, retail prices hover around AUD 60 (~USD 42) per dose. In Asian markets, prices are generally lower, often around USD 20–30 per tablet, owing to local pricing regulations and market dynamics.
What are the projected market growth and pricing trends?
The global market for triptans, including RELPAX, is expected to grow modestly at a CAGR of around 4%, driven by increased migraine prevalence, advancements in formulation, and expanding indications for combination therapies.
Pricing trends forecast a gradual decline in retail costs over the next five years:
-
United States: Prices are expected to decrease by 10–15%, primarily due to increasing competition from generics and biosimilars once exclusivity expires or off-patent status is achieved.
-
Europe: Negotiated reimbursement prices are expected to stabilize or slightly decline, driven by cost-containment policies and increased adoption of telemedicine.
-
Emerging Markets: Price reductions are contingent on local policies but could fall by 20–30% with increased generic entry and regional price negotiations.
How might patent status and generic entry affect future prices?
RELPAX's patent protection expired in the U.S. in late 2022, with generic versions now available. Initial generic prices are approximately 40–50% lower than brand-name RELPAX, with further reductions expected as market consolidation occurs.
In Europe, patent exclusivity opposed by patent litigation or delayed approval could postpone generic entry until 2024–2025, resulting in sustained higher prices during this period.
What strategic considerations should manufacturers and payers observe?
Manufacturers should focus on:
-
Differentiation: highlighting unique formulations such as higher bioavailability or faster onset.
-
Pricing flexibility: employing tiered pricing models in emerging markets.
-
Patient access programs: discounts for uninsured or underinsured patients.
Payers aim to:
-
Negotiate preferred formulary placements.
-
Use step therapy protocols to minimize costs.
-
Promote generic substitution when available to curb expenses.
Key Takeaways
- RELPAX holds a moderate share in the triptan segment, with regional variations shaping its market dynamics.
- Current pricing in the U.S. is around $45–$50 per tablet; Europe sees lower costs due to negotiated prices.
- Market growth remains steady, but upcoming generic entry is expected to lead to price declines.
- Patent expiration and regulatory differences will influence future pricing strategies.
- Both manufacturers and payers are adopting strategies to optimize access and control costs.
FAQs
Q1: When did RELPAX lose patent protection in the United States?
A1: Patent protection expired in late 2022, enabling generic versions to enter the U.S. market.
Q2: How does RELPAX compare to other triptans in terms of efficacy?
A2: Efficacy is comparable among triptans; RELPAX has a similar onset of action but may offer improved tolerability for some patients based on individual response.
Q3: What are the main factors influencing RELPAX's pricing in different regions?
A3: Pricing factors include patent status, regulatory policies, insurance coverage, negotiations with healthcare authorities, and market competition.
Q4: Are there any new formulations or indications for RELPAX?
A4: As of 2023, no new formulations or additional indications have been approved; focus remains on optimizing existing formulations.
Q5: What is the outlook for RELPAX's market share post-generic entry?
A5: Market share is expected to decline, but brand recognition and physician preferences may sustain a niche segment, especially if differentiated by clinical benefits.
References
[1] Grand View Research. (2023). Migraine Drugs Market Size, Share & Trends Analysis. Grand View Research. https://www.grandviewresearch.com/industry-analysis/migraine-drugs-market
More… ↓
