Share This Page
Drug Price Trends for RYBELSUS
✉ Email this page to a colleague
Average Pharmacy Cost for RYBELSUS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
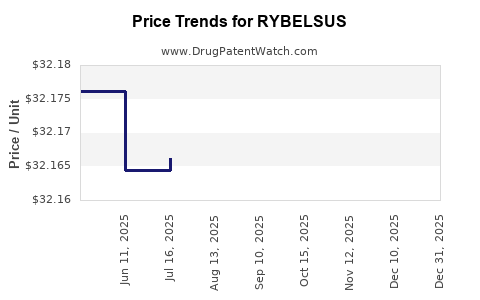
| RYBELSUS 14 MG TABLET | 00169-4314-30 | 33.16713 | EACH | 2026-01-01 |
| RYBELSUS 7 MG TABLET | 00169-4307-30 | 33.17299 | EACH | 2026-01-01 |
| RYBELSUS 3 MG TABLET | 00169-4303-30 | 33.16024 | EACH | 2026-01-01 |
| RYBELSUS 14 MG TABLET | 00169-4314-30 | 32.20110 | EACH | 2025-12-17 |
| RYBELSUS 3 MG TABLET | 00169-4303-30 | 32.19441 | EACH | 2025-12-17 |
| RYBELSUS 7 MG TABLET | 00169-4307-30 | 32.20679 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
RYBELSUS Market Analysis and Price Projections
Market Landscape and Competitive Environment
Rybelsus (semaglutide oral), developed by Novo Nordisk, is a glucagon-like peptide-1 (GLP-1) receptor agonist approved for the treatment of type 2 diabetes mellitus. Its primary differentiation lies in its oral formulation, offering an alternative to injectable GLP-1 RAs.
Key Competitors in the GLP-1 RA Market:
- Injectable Semaglutide (Ozempic): Also from Novo Nordisk, Ozempic is a weekly injectable semaglutide. It is a direct competitor with a more established track record and a broader indication for cardiovascular risk reduction in certain patients with type 2 diabetes.
- Liraglutide (Victoza): Developed by Novo Nordisk, Victoza is a daily injectable GLP-1 RA. It has been available longer than semaglutide products and also holds a cardiovascular outcome indication.
- Dulaglutide (Trulicity): Eli Lilly and Company's weekly injectable GLP-1 RA. It is a significant player in the market, offering similar efficacy and safety profiles.
- Exenatide (Byetta/Bydureon): AstraZeneca's GLP-1 RA, available in both twice-daily and weekly formulations. Byetta was one of the first GLP-1 RAs approved.
- Tirzepatide (Mounjaro): Eli Lilly and Company's novel dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 receptor agonist. Mounjaro has demonstrated superior glycemic control and weight loss in clinical trials compared to existing GLP-1 RAs, positioning it as a significant future competitor.
Market Penetration Drivers for Rybelsus:
- Oral Convenience: Eliminates the need for injections, which is a major barrier for many patients considering GLP-1 RAs. This is Rybelsus's primary competitive advantage.
- Glycemic Control: Rybelsus has demonstrated significant reductions in HbA1c levels comparable to injectable GLP-1 RAs.
- Weight Management: Like other GLP-1 RAs, Rybelsus can contribute to modest weight loss, an important benefit for many individuals with type 2 diabetes.
- Cardiovascular Outcome Trials (CVOTs): While Rybelsus has initiated CVOTs, current data regarding its cardiovascular benefits are less established than for Ozempic and Victoza. However, the ongoing SELECT trial is expected to provide critical information on its cardiovascular risk reduction capabilities.
Market Challenges for Rybelsus:
- Cost: GLP-1 RAs are generally high-cost medications. The price of Rybelsus is positioned to reflect this premium.
- Absorption Variability: Oral semaglutide absorption can be variable, requiring specific administration instructions (e.g., on an empty stomach with a small amount of water).
- Gastrointestinal Side Effects: Nausea, vomiting, and diarrhea are common side effects, though they are generally dose-dependent and transient.
- Competition from Newer Agents: The development of tirzepatide (Mounjaro) with superior efficacy in head-to-head trials poses a significant competitive threat, potentially capturing market share from existing GLP-1 RAs, including Rybelsus.
Pricing Strategy and Projections
Novo Nordisk has adopted a premium pricing strategy for Rybelsus, reflecting its novel oral delivery mechanism and the established value of GLP-1 receptor agonists in managing type 2 diabetes.
Current Pricing Landscape:
- List Price: The manufacturer's list price for Rybelsus varies by dosage strength and formulation. For example, the 7 mg once-daily tablet had a U.S. list price of approximately $891 per 30-day supply in early 2023. The 14 mg once-daily tablet had a list price of approximately $916 per 30-day supply during the same period. (Source: GoodRx data, March 2023).
- Net Price: Actual prices paid by patients and payers are subject to significant discounts through rebates, pharmacy benefit manager (PBM) negotiations, and commercial insurance formularies. Net prices are typically lower than list prices, but precise figures are proprietary and vary widely.
- Comparative Pricing: Injectable semaglutide (Ozempic) has a similar list price range, typically between $800 and $900 per 30-day supply, depending on dosage. Liraglutide (Victoza) and dulaglutide (Trulicity) generally have slightly lower list prices, often in the $700 to $800 range per 30-day supply.
Factors Influencing Future Pricing:
- Market Share and Uptake: The rate at which Rybelsus gains market share and patient acceptance will influence its long-term pricing power. Strong uptake driven by convenience could support current price levels.
- Clinical Trial Data (SELECT Trial): Positive results from the SELECT trial demonstrating significant cardiovascular risk reduction for Rybelsus could lead to broader payer coverage and potentially support higher price points or justify current pricing by adding a critical secondary benefit.
- Competitive Pressures: The emergence and increasing market penetration of tirzepatide (Mounjaro) will exert downward pressure on GLP-1 RA pricing across the board. If Mounjaro proves to be more efficacious and cost-effective for a broader patient population, payers may demand greater price concessions for Rybelsus and other GLP-1 RAs.
- Payer Negotiations: Payers continuously assess the value proposition of drugs. Intensified negotiations and formulary restrictions could lead to greater price erosion.
- Generic Entry: As Rybelsus is a relatively new molecule, generic competition is not an immediate concern. However, patent expiry in the distant future will ultimately lead to price declines.
- Inflation and Economic Conditions: General inflation rates and broader economic conditions can influence pharmaceutical pricing strategies.
Price Projection Scenarios:
-
Scenario 1: Stable Pricing (2-4% annual increase)
- Assumption: Rybelsus maintains strong market positioning due to its oral convenience, and the SELECT trial yields positive cardiovascular outcomes.
- Projection: Annual price increases are expected to align with historical pharmaceutical price inflation, averaging 2-4% per year over the next five years. Net prices will continue to be influenced by payer rebates.
- Example: By 2028, the list price for the 14 mg dose could reach approximately $990-$1,070 per 30-day supply.
-
Scenario 2: Moderate Price Erosion (0-2% annual increase or slight decrease)
- Assumption: Competition from tirzepatide intensifies, and payers leverage this competition to negotiate steeper discounts. The SELECT trial results are neutral or less impactful than anticipated.
- Projection: Price growth will be significantly curtailed, or prices may see slight declines as Novo Nordisk competes more aggressively on price to defend market share.
- Example: By 2028, the list price for the 14 mg dose could remain around $916-$950 per 30-day supply, with net prices potentially declining.
-
Scenario 3: Significant Price Pressure (Negative annual growth)
- Assumption: Tirzepatide demonstrates overwhelming superiority and cost-effectiveness, leading to widespread formulary restrictions and reduced prescribing of Rybelsus. The SELECT trial shows no significant benefit.
- Projection: Novo Nordisk may be forced to implement price reductions to maintain market access and volume.
- Example: By 2028, the list price for the 14 mg dose could fall below $850 per 30-day supply.
Impact of Market Access and Formulary Placement:
- Payer Coverage: The availability of Rybelsus on commercial and government formularies is a critical determinant of its market penetration and, consequently, its pricing power. Early adoption by major PBMs and health plans, often with tiered co-pays, has been instrumental in driving initial uptake.
- Prior Authorization and Step-Therapy: Requirements for prior authorization or step-therapy (requiring patients to try other, often less expensive, medications first) can limit Rybelsus's accessibility and necessitate greater price flexibility.
Patent Landscape and Exclusivity
The patent landscape for Rybelsus is crucial for understanding its long-term market exclusivity and potential for generic entry. Novo Nordisk has secured a comprehensive portfolio of patents covering the compound, its formulations, manufacturing processes, and methods of use.
Key Patent Areas:
- Composition of Matter Patents: These are fundamental patents that protect the semaglutide molecule itself. These patents typically have the longest protection periods. U.S. Patent No. 9,700,720 for semaglutide, filed in 2016 and granted in 2017, is a key example.
- Formulation Patents: These patents protect the specific oral formulation of semaglutide, including excipients and methods of stabilization required for oral delivery. U.S. Patent Nos. such as 10,500,412, 10,603,480, and 10,717,984 cover aspects of the oral delivery technology. These patents are vital for Rybelsus's differentiation.
- Manufacturing Process Patents: These patents cover the specific chemical synthesis and manufacturing processes used to produce semaglutide and its oral formulations.
- Method of Use Patents: These patents protect specific therapeutic uses of semaglutide, such as for the treatment of type 2 diabetes.
- Polymorph Patents: Patents may also cover specific crystalline forms (polymorphs) of semaglutide, which can affect stability and bioavailability.
Patent Exclusivity Timeline:
-
U.S. Market: The primary composition of matter patent for semaglutide is expected to expire around 2031. However, formulation and method-of-use patents often extend exclusivity periods. Novo Nordisk has strategically filed numerous patents to create a layered defense.
- The U.S. Orange Book lists multiple patents for Rybelsus, with various expiration dates ranging from the late 2020s to the mid-2030s. For instance, U.S. Patent No. 10,717,984 has an expiry date in 2034.
- Regulatory Exclusivity: In addition to patent protection, Rybelsus benefits from regulatory exclusivities granted by regulatory agencies. In the U.S., new chemical entities (NCEs) receive 5 years of data exclusivity, and if approved for a new indication after the initial approval, can receive an additional 3 years of exclusivity. Rybelsus, as an NCE formulation, may also be eligible for certain pediatric exclusivity extensions.
-
European Market: Similar to the U.S., Rybelsus is protected by a portfolio of patents in Europe. European Patent EP 2613550 B1, covering semaglutide, has an expiry date around 2026, but a supplementary protection certificate (SPC) could extend this. The oral formulation patents are critical for extending market exclusivity.
Potential for Generic Competition:
- Patent Challenges: Generic manufacturers will likely challenge Novo Nordisk's patents once they are eligible for review. Successful challenges could lead to earlier generic entry.
- ANDA Filings: Following patent expiry or successful challenges, generic companies can file Abbreviated New Drug Applications (ANDAs) with the FDA. The first generic to file a successful ANDA may be eligible for 180 days of market exclusivity.
- Formulation Complexity: The complex formulation technology required for oral semaglutide may present technical hurdles for generic manufacturers, potentially delaying their entry. Demonstrating bioequivalence for an oral peptide formulation can be more challenging than for small molecules.
Impact of Biosimilars:
- Semaglutide is a peptide, not a biologic, so the term "biosimilar" is not applicable. However, generic versions of semaglutide will eventually enter the market.
Market Size and Growth Forecasts
The global market for type 2 diabetes treatments is substantial and projected to continue growing, driven by increasing prevalence of obesity and diabetes worldwide, as well as advancements in therapeutic options. The GLP-1 RA class, in particular, has experienced rapid growth.
Historical Market Performance (GLP-1 RA Class):
- The GLP-1 RA market has grown from a few billion dollars annually in the early 2010s to over $20 billion globally in recent years, largely due to the introduction of more effective and convenient formulations like weekly injectables and now oral semaglutide. (Source: Various market research reports).
Rybelsus Specific Market Share and Revenue:
- 2020: Rybelsus launched in the U.S. in September 2020. Its initial market penetration was moderate due to its novelty and the need for prescriber and patient education.
- 2021-2022: Rybelsus demonstrated strong growth, with sales reaching approximately DKK 8.9 billion (US$1.3 billion) in 2022, representing a significant increase from its launch year. (Source: Novo Nordisk Annual Reports).
- Projected Market Share: Analysts project Rybelsus to capture a significant share of the oral antidiabetic market and a notable portion of the overall GLP-1 RA market. Its oral convenience is expected to be a primary driver of its uptake, particularly among patients who are reluctant to use injectable medications.
Factors Driving Market Growth:
- Increasing Diabetes Prevalence: Global rates of type 2 diabetes are rising due to lifestyle changes, aging populations, and increasing obesity.
- Improved Glycemic Control: GLP-1 RAs offer superior glycemic control compared to older classes of antidiabetics, leading to better patient outcomes and reduced risk of complications.
- Weight Management Benefits: The weight loss associated with GLP-1 RAs is a highly attractive feature for a large segment of the type 2 diabetes population.
- Cardiovascular Risk Reduction: Emerging data and ongoing trials for cardiovascular benefits add significant value to the GLP-1 RA class, potentially expanding their use to a broader patient population beyond glycemic control.
- Innovations in Delivery: The success of Rybelsus validates the demand for convenient oral administration of once-daily medications.
Forecasted Market Size for Rybelsus:
- Short-Term (1-3 years): Rybelsus sales are expected to continue their rapid growth trajectory, likely reaching DKK 15-20 billion (US$2.2-2.9 billion) annually by 2025, driven by expanding indications and market access.
- Medium-Term (3-7 years): Growth will likely moderate as competition intensifies and patent expirations approach. However, Rybelsus is expected to remain a significant product. Annual sales could stabilize in the range of DKK 20-25 billion (US$2.9-3.6 billion) by 2028, contingent on the SELECT trial's success and the competitive landscape.
- Long-Term (7+ years): Sales will be impacted by generic competition. However, if Rybelsus demonstrates robust long-term benefits, it may retain a market presence even with generics, albeit at a lower price point.
Impact of Tirzepatide (Mounjaro):
- The introduction and widespread adoption of tirzepatide (Mounjaro) represents a significant potential headwind for Rybelsus and the broader GLP-1 RA market. Mounjaro's superior efficacy in clinical trials suggests it could capture substantial market share. This competitive pressure could dampen the growth projections for Rybelsus, particularly in the medium to long term, potentially leading to greater price erosion than initially anticipated.
Regional Market Dynamics:
- North America: Largest market due to high diabetes prevalence, advanced healthcare infrastructure, and willingness to adopt innovative therapies.
- Europe: Significant market with strong demand for advanced diabetes treatments, though pricing and reimbursement policies vary by country.
- Asia-Pacific: Fastest-growing market due to rising prevalence of diabetes and increasing access to healthcare. Japan and China are key markets.
- Rest of the World: Emerging markets present growth opportunities, but adoption may be slower due to cost constraints.
Key Takeaways
Rybelsus has established a strong initial market presence in the type 2 diabetes market, primarily driven by its novel oral formulation. Its pricing strategy reflects a premium for this convenience, with list prices comparable to injectable GLP-1 receptor agonists. The patent landscape provides robust exclusivity through the mid-2030s, although generic challenges are anticipated. Market growth is fueled by rising diabetes prevalence and the therapeutic benefits of GLP-1 RAs, with Rybelsus projected to achieve substantial revenue. However, the emergence of highly effective competitors like tirzepatide (Mounjaro) poses a significant risk of increased price pressure and potentially reduced market share growth in the medium to long term. Positive outcomes from the ongoing SELECT trial are critical for reinforcing Rybelsus's value proposition and supporting its future pricing and market positioning.
Frequently Asked Questions
-
What is the primary competitive advantage of Rybelsus compared to other GLP-1 receptor agonists? Rybelsus's primary advantage is its oral formulation, offering a non-injectable alternative for patients treating type 2 diabetes.
-
When are the key patents for Rybelsus expected to expire in the U.S. market? While the core semaglutide composition of matter patents expire around 2031, formulation and method-of-use patents extend Rybelsus's exclusivity period, with some expiring in the mid-2030s, such as U.S. Patent No. 10,717,984 in 2034.
-
How might the results of the SELECT trial impact Rybelsus's market price? Positive results from the SELECT trial, demonstrating significant cardiovascular risk reduction, could strengthen Rybelsus's value proposition, potentially supporting current pricing levels or allowing for modest increases by adding a critical secondary benefit.
-
What is the expected impact of tirzepatide (Mounjaro) on Rybelsus's market growth projections? Tirzepatide, with its demonstrated superior efficacy in clinical trials, is expected to exert significant competitive pressure, potentially dampening Rybelsus's growth projections and leading to greater price erosion than previously anticipated.
-
Are there any significant technical challenges for generic manufacturers in developing Rybelsus generics? Yes, the complex formulation technology required for oral peptide delivery may present technical hurdles for generic manufacturers, potentially affecting their ability to demonstrate bioequivalence and delaying market entry.
Citations
[1] GoodRx. (March 2023). Semaglutide Prices, Coupons, and Patient Assistance Programs. GoodRx.com. [2] Novo Nordisk. (2023). Annual Report 2022. Novo Nordisk A/S. [3] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. FDA.gov.
More… ↓
