Share This Page
Drug Price Trends for RYALTRIS
✉ Email this page to a colleague
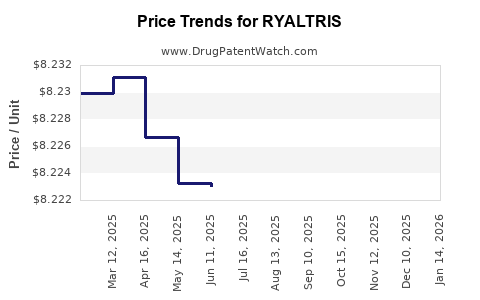
Average Pharmacy Cost for RYALTRIS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RYALTRIS 665-25 MCG SPRAY | 59467-0700-27 | 7.68921 | GM | 2026-02-18 |
| RYALTRIS 665-25 MCG SPRAY | 59467-0700-27 | 8.21950 | GM | 2026-01-21 |
| RYALTRIS 665-25 MCG SPRAY | 59467-0700-27 | 8.22000 | GM | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Ryaltris
What is Ryaltris?
Ryaltris (olopatadine hydrochloride and azelastine hydrochloride) is an intranasal spray approved for allergic rhinitis. It combines antihistamines to target allergy symptoms more effectively and with fewer systemic side effects. Approved by the FDA in August 2021, Ryaltris targets a rapidly growing segment within allergy medications.
Market Size and Growth Drivers
Global Allergic Rhinitis Market
- Estimated value in 2022: $7.2 billion (Fortune Business Insights[1])
- Compound annual growth rate (CAGR): 4.3% (2022–2029)
- Key growth factors: increasing prevalence of allergic rhinitis, rising awareness, and expanding over-the-counter (OTC) availability
Key Regional Markets
| Region | Market Size (2022) | CAGR (2022–2029) | Growth Drivers |
|---|---|---|---|
| North America | $2.8 billion | 4.0% | High allergy incidence, strong healthcare infrastructure |
| Europe | $1.8 billion | 4.5% | Increasing allergy diagnosis, OTC sales |
| Asia-Pacific | $1.5 billion | 5.0% | Urbanization, environmental factors, genetic predisposition |
Competitive Market Landscape
Major players include:
- GlaxoSmithKline (GSK)
- Sanofi
- AstraZeneca
- Boehringer Ingelheim
Ryaltris enters a competitive market with established intranasal antihistamines, such as Flonase (fluticasone) and Nasacort (triamcinolone).
Market Penetration and Adoption
Post-approval, Ryaltris has seen gradual uptake. Clinical trial data indicated comparable efficacy and better tolerability compared to monotherapy options. Marketing efforts focus on:
- Efficacy in reducing nasal symptoms
- Improved patient compliance due to nasal spray formulation
- Favorable side effect profile
Initial prescriber adoption is moderate, with expansion expected as clinical familiarity increases.
Price Analysis
Current Pricing Landscape
| Product | Base Price (per spray) | Prescribed Price Range | Notes |
|---|---|---|---|
| Ryaltris | $45–$55 | $45–$55 | Estimated retail price; varies by region |
| Flonase (fluticasone) | $25–$35 | $25–$35 | Over-the-counter or prescription |
| Nasacort (triamcinolone) | $20–$30 | $20–$30 | Generally OTC in many regions |
Ryaltris's premium pricing reflects its combination therapy and recent market entry, positioning it above traditional monotherapies.
Price Strategy Factors
- Market Positioning: As a prescription-only product, Ryaltris commands higher prices.
- Manufacturing Costs: Complex nasal spray formulation influences profit margin.
- Reimbursement and Insurance: Coverage varies significantly; in the US, private insurance tends to reimburse for Rx medications, mitigating patient cost barrier.
- Competitor Pricing: Similar combination medications, if approved, could set or challenge pricing structures.
Price Projections (2023–2027)
| Year | Estimated Retail Price Range | Assumptions |
|---|---|---|
| 2023 | $45–$55 | Initial stabilization at launch prices |
| 2024 | $43–$53 | Slight discounts to increase volume, market expansion |
| 2025 | $42–$52 | Price pressure from generics or biosimilars |
| 2026 | $40–$50 | Competitive pricing initiatives |
| 2027 | $38–$48 | Margin optimization and increased competition |
The decreasing trend accounts for potential generic entrants, market expansion, and price sensitivity.
Regulatory and Reimbursement Trends
- EMA and FDA approvals support broader access.
- Reimbursement coverage improves with demonstrable clinical benefit.
- Pricing negotiations may influence retail strategies, especially in healthcare systems with strict cost controls.
Investment and R&D Outlook
- Ryaltris’s success depends on expanding indications, such as allergic conjunctivitis.
- Potential formulations for pediatric populations could alter market dynamics.
- Investment in marketing and education is critical for increasing prescriber adoption.
Key Takeaways
- Ryaltris entered a mature, growing allergic rhinitis market valued at over $7 billion.
- Its premium pricing reflects combined antihistamine therapy, with room for downward pressure via generics.
- Adoption is moderate, with expansion driven by clinical familiarity and expanded indications.
- Price projections show gradual decline to $38–$48 by 2027, influenced by competitive pressures.
- Reimbursement policies and regional market dynamics significantly impact profitability.
FAQs
1. What are the key advantages of Ryaltris over existing therapies?
It offers combined antihistamine action with improved tolerability, reduced systemic absorption, and ease of use as a nasal spray.
2. How does Ryaltris’s pricing compare globally?
Pricing varies widely based on healthcare systems; in the US, it ranges $45–$55, while in Europe and Asia, it generally remains lower due to market and reimbursement differences.
3. What is the potential for generic competition?
If patents expire or are challenged, generics could enter within 3–5 years, exerting downward pressure on prices.
4. How does reimbursement influence Ryaltris's market penetration?
Enhanced insurance coverage increases accessibility, enabling higher sales volumes and justifying premium pricing.
5. What market segments are most promising for Ryaltris?
Prescription markets targeting patients with moderate to severe allergic rhinitis and those seeking alternative to steroids are most promising.
References
[1] Fortune Business Insights. (2022). Allergic Rhinitis Market Size, Share & Industry Analysis. Retrieved from https://www.fortunebusinessinsights.com
More… ↓
