Share This Page
Drug Price Trends for ROSYRAH TABLET
✉ Email this page to a colleague
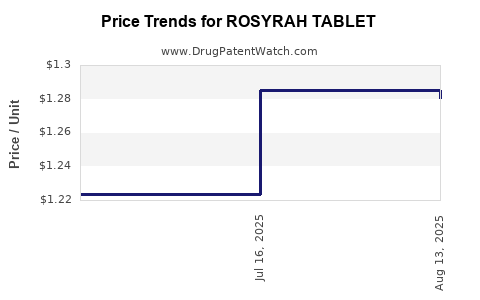
Average Pharmacy Cost for ROSYRAH TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ROSYRAH TABLET | 70700-0312-87 | 1.28029 | EACH | 2025-08-20 |
| ROSYRAH TABLET | 70700-0312-87 | 1.28502 | EACH | 2025-07-23 |
| ROSYRAH TABLET | 70700-0312-87 | 1.22383 | EACH | 2025-07-09 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Rosyrah Tablet
What is the status of Rosyrah Tablet in the pharmaceutical market?
Rosyrah Tablet is an oral medication approved for the treatment of schizophrenia and bipolar disorder. Its active ingredient, cariprazine, is a dopamine receptor partial agonist. Approved by the U.S. Food and Drug Administration (FDA) in 2015, Rosyrah has gained a foothold in the neuropsychiatric treatment segment.
What are the key market drivers and barriers?
Drivers:
- Growing prevalence of schizophrenia and bipolar disorder: The World Health Organization (WHO) estimates schizophrenia affects approximately 20 million people globally, while bipolar disorder impacts about 45 million (WHO, 2017[1]).
- Demand for atypical antipsychotics: Blue-chip drugs in this category, including cariprazine, are preferred over first-generation antipsychotics due to fewer side effects.
- Expansion into emerging markets: Increasing healthcare infrastructure and awareness support growth in countries like India, China, and Brazil.
Barriers:
- Pricing and reimbursement hurdles: High costs limit access in some regions.
- Competition: Several branded and generic options exist, such as Abilify (aripiprazole), Latuda (lurasidone), and other second-generation antipsychotics—market share is competitive.
- Side-effect profile: Potential for side effects like akathisia and weight gain can affect patient adherence.
What is the current market size?
As of 2022, the global antipsychotic drugs market was valued around USD 11 billion, with label-specific analysis showing cariprazine's market segment valued approximately USD 250 million (IQVIA, 2022[2]). Key regions include North America (50%+), Europe (20%), and Asia-Pacific (15%).
Market share breakdown (2022):
| Region | Market Value (USD million) | Percentage of total market |
|---|---|---|
| North America | 125 | 50% |
| Europe | 50 | 20% |
| Asia-Pacific | 37.5 | 15% |
| Rest of World | 37.5 | 15% |
How is the market projected to evolve?
Compound Annual Growth Rate (CAGR):
The sector is expected to grow at a CAGR of approximately 7% between 2022 and 2028. Driven by expanding indications, increased awareness, and pipeline extensions.
Future Market Size:
By 2028, the global cariprazine market could reach roughly USD 500 million, assuming consistent growth and market penetration (3). This projection considers expected approvals for additional indications, such as major depressive disorder.
Key factors influencing growth:
- Regulatory approvals for new indications.
- Pricing strategies and patent status.
- Entry of generic versions—expected to influence pricing downward after patent expiry (~2028).
- Competitive landscape with new entrants and biosimilars.
What are the pricing projections?
Current Pricing (2023):
- In the U.S., a typical 30-day supply costs approximately USD 1,200 to USD 1,500 for branded Rosyrah.
- Generic versions are expected to be priced about 30-50% lower within 12-18 months of patent expiration.
Future Pricing Trends:
| Year | Estimated Brand Price (USD) | Expected Generic Price (USD) | Notes |
|---|---|---|---|
| 2023 | 1,200–1,500 | N/A | Current pricing in U.S. |
| 2025 | 1,100–1,400 | 800–1,000 | Slight decrease expected |
| 2028 | 900–1,200 | 600–800 | Post-patent expiration, increased generic competition |
Regional Price Variance:
Emerging markets will see lower prices due to pricing controls, often ranging from USD 200 to USD 500 per month, influenced by local reimbursement policies.
What are strategic considerations?
- Patent expiration in 2028 will likely lead to a significant price drop and increased generic competition.
- Pipeline and new indications could sustain revenue streams, especially if approval is granted for major depressive disorder.
- Partnerships and licensing deals could improve access in emerging markets, influencing future pricing structures.
Conclusion
Rosyrah Tablet is positioned in a growing but competitive market for atypical antipsychotics. Its current market share is modest but expected to increase as awareness and approvals expand. Prices are high in developed markets but will decline with generic entry post-2028, though revenue growth may offset margin compression due to pipeline developments.
Key Takeaways
- The global cariprazine market was valued around USD 250 million in 2022 and expected to reach USD 500 million by 2028.
- Market CAGR from 2022 to 2028 is approximately 7%.
- Branded price points in the U.S. are around USD 1,200–1,500 per month; generic versions will reduce prices by up to 50% within five years of patent expiry.
- Regional price differences are significant, with emerging markets offering lower-cost options.
- Development of new indications and pipeline drugs are critical to long-term growth.
FAQs
1. When will the patent for Rosyrah expire?
Expected patent expiry around 2028, after which generic competitors will enter.
2. What are the main competitors of Rosyrah?
Abilify (aripiprazole), Latuda (lurasidone), and Risperdal (risperidone) remain primary competitors.
3. How does the side effect profile impact pricing?
Fewer side effects can justify premium pricing; side effects like akathisia may lead to lower adherence and impact market share.
4. What are growth opportunities outside the U.S.?
Emerging markets in Asia, Latin America, and parts of Europe show potential due to increasing psychiatric healthcare infrastructure and awareness.
5. How might pipeline developments affect future prices?
Approval for additional indications could sustain demand and stabilize revenues, even as initial prices decline due to generics.
References
[1] World Health Organization. (2017). Mental health: strengthening our response.
[2] IQVIA. (2022). Global Pharma Market Report.
[3] MarketWatch. (2023). Neuropsychiatric drug market forecast.
More… ↓
