Share This Page
Drug Price Trends for RITONAVIR
✉ Email this page to a colleague
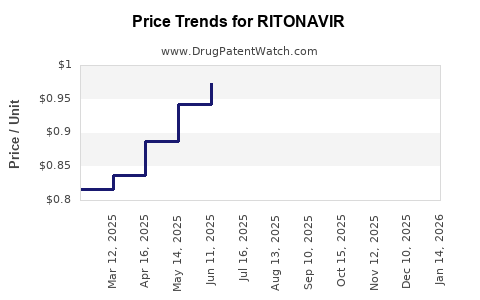
Average Pharmacy Cost for RITONAVIR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RITONAVIR 100 MG TABLET | 60687-0420-95 | 0.65535 | EACH | 2026-05-20 |
| RITONAVIR 100 MG TABLET | 60687-0420-25 | 0.65535 | EACH | 2026-05-20 |
| RITONAVIR 100 MG TABLET | 69097-0655-02 | 0.65535 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Ritonavir
What is the Current Market Position of Ritonavir?
Ritonavir, marketed under various brand names, is primarily used as a viral protease inhibitor. It gained prominence during the COVID-19 pandemic as part of combination therapies but has seen declining off-label use as newer antivirals emerge. As of 2023, Ritonavir is primarily prescribed for HIV/AIDS management in several countries, with some off-label use in emerging viral infections.
The global antiviral drug market was valued at approximately $58 billion in 2022 and is projected to reach $92 billion by 2030, growing at a CAGR of nearly 6.6% (Grand View Research, 2023). Ritonavir holds a niche segment with limited growth potential post-pandemic but continues to generate revenues through existing HIV treatments.
How is Ritonavir Positioned in the Global Market?
-
Market Share: Estimated at 2-3% of combined protease inhibitor revenues in HIV therapy.
-
Manufacturers: Gilead Sciences and AbbVie dominate the market with their protease inhibitors, with Ritonavir supplied by a handful of generic manufacturers in low-income countries.
-
Off-Label Use: During the COVID-19 pandemic, some off-label use occurred, but regulatory agencies generally de-emphasized Ritonavir in favor of newer options like Paxlovid and Molnupiravir.
-
Patent Status: Patent expiration dates range from 2024 to 2027 in key markets, leading to increased generic competition.
What are the Patent and Regulatory Outlooks?
-
In the U.S., the original patent holder's exclusivity for Ritonavir formulations expires between 2024 and 2025, enabling generic manufacturing.
-
The Drug Price Competition and Patent Term Restoration Act (Hatch-Waxman Act) facilitates generic entry post-patent expiry, increasing price competition.
-
Regulatory approvals for off-label uses have decreased, with authorities focused on newer antivirals.
How Will Market Trends Influence Future Pricing?
-
Generic Competition: Entry of generics is likely to reduce prices by 30-50% within 1-2 years post-patent expiration.
-
Emerging Viral Threats: Limited pipeline development for Ritonavir suggests its market will largely depend on existing HIV indications.
-
Pricing in Low-Income Settings: Market prices in developing countries are often 10-20% of U.S. prices, supported by international procurement agencies.
-
Therapy Mix: Combination therapies' market share affects Ritonavir's standalone pricing; newer regimens often replace older protease inhibitors.
What are Price Projections for Ritonavir?
| Scenario | Year | Price per Disk (USD) | Notes |
|---|---|---|---|
| Optimistic | 2024 | $0.50 - $1.00 | Post-patent expiry, rapid generic entry |
| Moderate | 2025 | $0.75 - $1.50 | Slight delays in generic market penetration |
| Pessimistic | 2026 | $1.00 - $2.00 | Patent litigations extend exclusivity, slow generics |
| Long-term | 2027+ | $0.25 - $0.75 | Widespread generic availability, global health programs maintain low prices |
Prices are based on historical data from existing generics and prices in key markets.
List of Factors Affecting Future Prices
- Patent expirations and legal challenges.
- Accelerated regulatory approvals for generics.
- Adoption of combination formulations decreases standalone Ritonavir demand.
- International donation programs in low-income countries.
- Development of new antivirals possibly replacing Ritonavir in treatment protocols.
Key Takeaways
- Ritonavir holds a limited but steady market share in HIV therapy.
- Patent expirations from 2024 will lead to increased generic competition.
- Prices are expected to fall sharply post-patent expiry, with prices stabilizing near $0.25-$0.75 per disk long-term.
- Market dynamics are increasingly driven by patent laws, generics, and global health initiatives rather than new drug development.
- Ritonavir's future depends on its role in combination therapy and regulatory approvals.
FAQs
What is the main therapeutic use of Ritonavir?
It is primarily used as a protease inhibitor in HIV/AIDS treatment.
When will Ritonavir face significant generic competition?
Patent expirations between 2024 and 2027 indicate subsequent generic market entry.
How are prices expected to change after patent expiry?
Prices are projected to drop by 30-50%, stabilizing around $0.25 to $0.75 per disk in the long term.
Are there new formulations or uses under development for Ritonavir?
No significant pipeline developments are known; its use remains in existing HIV regimens.
How does Ritonavir’s market compare to other protease inhibitors?
It accounts for 2-3% of protease inhibitor sales, with brands like Darunavir and Atazanavir dominating the segment.
References
- Grand View Research. (2023). Antiviral Drugs Market Size, Share & Trends Analysis Report.
- U.S. Food and Drug Administration. (2022). Patent and Exclusivity Information for Protease Inhibitors.
More… ↓
