Share This Page
Drug Price Trends for RISPERIDONE
✉ Email this page to a colleague
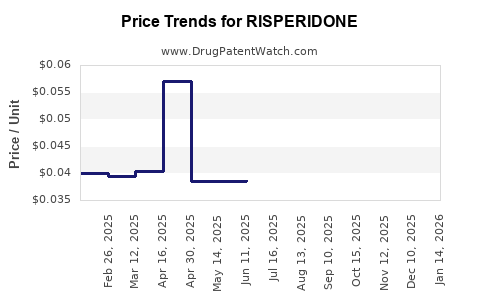
Average Pharmacy Cost for RISPERIDONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RISPERIDONE ER 50 MG VIAL | 70748-0272-13 | 866.29429 | EACH | 2026-05-20 |
| RISPERIDONE ER 50 MG VIAL | 70748-0272-11 | 866.29429 | EACH | 2026-05-20 |
| RISPERIDONE ER 25 MG VIAL | 70748-0270-13 | 415.33306 | EACH | 2026-05-20 |
| RISPERIDONE ER 25 MG VIAL | 70748-0270-11 | 415.33306 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for RISPERIDONE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| RISPERIDONE 0.5MG TAB | AvKare, LLC | 42291-0909-50 | 500 | 1070.75 | 2.14150 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| RISPERIDONE 3MG TAB,ORAL DISINTEGRATING | Golden State Medical Supply, Inc. | 49884-0402-91 | 7X4 | 234.35 | 2023-06-15 - 2028-06-14 | FSS | ||
| RISPERIDONE 3MG TAB | AvKare, LLC | 42291-0912-60 | 60 | 268.00 | 4.46667 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| RISPERIDONE 0.5MG TAB | AvKare, LLC | 42291-0909-60 | 60 | 128.49 | 2.14150 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
RISPERIDONE Market Analysis and Price Projections
Risperidone is an atypical antipsychotic drug used to treat schizophrenia, bipolar disorder, and irritability associated with autism. The global market for risperidone has experienced consistent demand driven by the increasing prevalence of mental health disorders and the drug's established efficacy. Price projections indicate a moderate decline in average selling prices (ASPs) due to generic competition and market saturation, offset by continued volume growth in emerging markets.
What is the Current Market Size and Growth Trajectory for Risperidone?
The global risperidone market size was valued at approximately $1.2 billion in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030, reaching an estimated $1.5 billion by the end of the forecast period. This growth is primarily attributed to the rising incidence of schizophrenia and bipolar disorder worldwide.
According to the World Health Organization (WHO), schizophrenia affects approximately 24 million people globally, and bipolar disorder impacts an estimated 40 million people [1]. The steady diagnosis rates and ongoing treatment needs for these chronic conditions underpin the sustained demand for risperidone.
Table 1: Global Risperidone Market Size and Forecast (USD Billion)
| Year | Market Size | CAGR (2024-2030) |
|---|---|---|
| 2023 | 1.20 | - |
| 2024 | 1.24 | 3.33% |
| 2025 | 1.28 | 3.23% |
| 2026 | 1.32 | 3.13% |
| 2027 | 1.37 | 3.03% |
| 2028 | 1.41 | 2.94% |
| 2029 | 1.46 | 2.86% |
| 2030 | 1.51 | 2.78% |
What are the Key Drivers of Risperidone Market Growth?
Several factors are driving the continued demand for risperidone:
- Increasing Prevalence of Mental Health Disorders: The global burden of mental illnesses, particularly schizophrenia and bipolar disorder, is rising due to increased awareness, improved diagnostic capabilities, and lifestyle factors. This trend directly translates to a larger patient pool requiring effective pharmacological interventions like risperidone. Data from the National Institute of Mental Health (NIMH) indicates that approximately 1.1% of U.S. adults experience schizophrenia in a given year [2].
- Generic Availability and Affordability: Risperidone has been available as a generic medication for over a decade. This broad generic availability has significantly reduced treatment costs, making it accessible to a wider population, including those in low- and middle-income countries. The availability of multiple generic manufacturers fosters competitive pricing.
- Established Efficacy and Safety Profile: Risperidone has a long history of clinical use, with a well-documented efficacy and safety profile for managing symptoms of psychosis, mania, and irritability. Its established therapeutic value continues to make it a preferred choice for many clinicians, particularly in cases where newer agents may be prohibitively expensive or less understood.
- Inclusion in Essential Medicines Lists: Risperidone is listed on the World Health Organization's Model List of Essential Medicines, underscoring its importance in global public health [3]. This designation often leads to increased government procurement and availability in public healthcare systems, further supporting market demand.
- Pediatric Use for Autism-Related Irritability: Risperidone is approved for the treatment of irritability associated with autistic disorder in children and adolescents. This expanded indication contributes to market volume, especially in developed countries with comprehensive pediatric mental health services.
What are the Major Restraints on Risperidone Market Growth?
Despite the positive growth drivers, the risperidone market faces certain constraints:
- Competition from Newer Atypical Antipsychotics: The development of newer atypical antipsychotics with potentially improved side-effect profiles or novel mechanisms of action presents a competitive threat. Drugs like aripiprazole, olanzapine, and quetiapine, while also facing generic competition, offer alternatives that may be chosen by prescribers and patients seeking different therapeutic outcomes or tolerability.
- Side Effects and Adverse Events: While generally well-tolerated, risperidone can cause significant side effects, including weight gain, metabolic disturbances (hyperglycemia, dyslipidemia), extrapyramidal symptoms (EPS), and hyperprolactinemia. These adverse events can lead to poor patient adherence and a shift towards alternative treatments with better tolerability profiles.
- Market Saturation in Developed Regions: In North America and Western Europe, the risperidone market is relatively mature. The patient populations are well-established, and the majority of eligible patients are already being treated. Future growth in these regions is largely dependent on an increase in diagnosed cases rather than market penetration.
- Price Erosion from Generic Competition: The presence of numerous generic manufacturers leads to intense price competition, driving down the ASP of risperidone. This makes it challenging for manufacturers to achieve significant revenue growth solely through price increases and emphasizes the need for volume expansion.
What is the Competitive Landscape for Risperidone?
The risperidone market is highly fragmented due to the widespread availability of generic versions. The primary competition comes from companies manufacturing and distributing generic risperidone formulations. Key players in the generic risperidone market include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries Ltd.
- Lupin Ltd.
- Dr. Reddy's Laboratories Ltd.
- Torrent Pharmaceuticals Ltd.
- Cipla Ltd.
- Actavis Generics (now part of Teva)
Branded risperidone, primarily represented by Johnson & Johnson's Risperdal®, now holds a minimal market share due to patent expiry and the dominance of generics. The competitive strategy for generic manufacturers revolves around cost-efficiency, broad distribution networks, and securing large supply contracts with healthcare systems and pharmacies.
What are the Price Projections and Factors Influencing Risperidone Pricing?
The average selling price (ASP) of risperidone has been on a downward trend since the advent of generic competition.
- Current ASP: The average global ASP for risperidone tablets (e.g., 1 mg, 2 mg, 3 mg, 4 mg) hovers around $0.20 to $0.50 per tablet, depending on the dosage, quantity purchased, and region. For oral solutions, prices are higher, typically ranging from $30 to $60 for a 100 mL bottle. Long-acting injectable (LAI) formulations, such as risperidone consta, command significantly higher prices, often between $200 and $350 per injection, although their market share within the overall risperidone market is smaller.
- Projected Price Trends: The ASP for oral formulations of risperidone is expected to continue declining at a modest rate of approximately 2-4% per year through 2030. This decline is driven by ongoing price negotiations, increased competition among generic suppliers, and the purchasing power of large healthcare organizations.
- Factors Influencing Pricing:
- Generic Competition: The number of generic manufacturers actively supplying the market is the most significant factor. More suppliers generally lead to lower prices.
- Dosage and Formulation: Higher dosages and specialized formulations (e.g., LAI) naturally command higher prices.
- Geographic Region: Prices vary considerably between developed and emerging markets due to differences in regulatory environments, healthcare infrastructure, local manufacturing capabilities, and patient affordability.
- Purchasing Volume and Contracts: Large-volume purchases by governments, hospital groups, and pharmacy benefit managers (PBMs) result in substantial price discounts.
- Manufacturing Costs: The cost of raw materials, active pharmaceutical ingredients (APIs), and manufacturing processes directly impacts the final selling price.
- Regulatory Approvals and Quality Standards: Meeting stringent regulatory requirements in major markets can influence production costs.
Table 2: Estimated Average Selling Price (ASP) Range for Risperidone (USD per unit)
| Formulation | Typical Dosage/Size | 2023 ASP Range | 2030 Projected ASP Range | Annual Price Change (Projected) |
|---|---|---|---|---|
| Oral Tablets | 1 mg - 4 mg (per tab) | $0.20 - $0.50 | $0.16 - $0.40 | -2% to -4% |
| Oral Solution | 100 mL | $30 - $60 | $25 - $50 | -2% to -3% |
| Long-Acting Injectable | 25 mg, 50 mg (per vial) | $200 - $350 | $180 - $320 | -1% to -2% |
Note: ASPs are indicative and can vary significantly based on specific market conditions and supply agreements.
What is the Impact of Regulatory Policies on the Risperidone Market?
Regulatory policies play a crucial role in shaping the risperidone market. Key policies include:
- Generic Drug Approval Pathways: The abbreviated new drug application (ANDA) process in the U.S. and similar pathways in other regions allow generic manufacturers to bring bioequivalent versions of risperidone to market. The speed and efficiency of these approval processes influence the timing of generic entry and subsequent price erosion.
- Intellectual Property (IP) Protection: While primary patents for risperidone have long expired, any new formulations, delivery systems, or combination therapies involving risperidone could be subject to new patent protections, creating niche market opportunities or extended exclusivity periods.
- Pharmacovigilance and Post-Market Surveillance: Regulatory bodies like the FDA and EMA continuously monitor the safety of approved drugs. Any new safety concerns or warnings related to risperidone could impact prescribing patterns and market demand. For example, the FDA has issued warnings regarding the risk of tardive dyskinesia and other movement disorders with antipsychotics [4].
- Pricing and Reimbursement Policies: Government policies regarding drug pricing, reimbursement schedules, and formulary placement significantly affect market access and sales volumes. In many countries, national health services or insurance providers negotiate prices and determine which drugs are covered.
- Manufacturing Standards (GMP): Strict adherence to Good Manufacturing Practices (GMP) is essential for all risperidone manufacturers. Failure to meet these standards can result in product recalls, manufacturing halts, and significant reputational damage.
What are the Future Trends and Opportunities in the Risperidone Market?
While the risperidone market is mature, several trends and opportunities exist:
- Emerging Market Penetration: Significant growth potential remains in emerging markets across Asia, Africa, and Latin America. Factors such as increasing healthcare expenditure, expanding access to mental health services, and the need for affordable treatments will drive demand for generic risperidone in these regions.
- Combination Therapies: Research into novel combination therapies that pair risperidone with other agents to enhance efficacy or mitigate side effects could represent a future opportunity, although significant R&D investment would be required.
- Improved Drug Delivery Systems: While less likely for a mature generic, advancements in drug delivery, such as more patient-friendly long-acting injectable formulations with longer dosing intervals, could maintain market relevance for specific patient populations.
- Focus on Cost-Effectiveness: Healthcare systems globally are increasingly focused on cost-effectiveness. Risperidone, as an established and affordable generic, is well-positioned to benefit from this trend, especially in budget-constrained environments.
- Geriatric Population Growth: The aging global population is projected to see an increase in age-related mental health conditions, which could contribute to sustained demand for antipsychotic treatments.
Key Takeaways
The risperidone market is characterized by stable demand, driven by the prevalence of mental health disorders and its generic accessibility. While facing price erosion due to intense competition and pressure from newer agents, the market is expected to grow moderately, primarily fueled by expansion in emerging economies. Generic manufacturers will continue to dominate the landscape, with price competitiveness and efficient distribution being critical success factors. Opportunities exist in underserved emerging markets and through potential, albeit limited, innovations in drug delivery or therapeutic combinations.
Frequently Asked Questions
-
Will risperidone face further significant price drops in the next five years? Price drops are expected to continue at a modest rate of 2-4% annually for oral formulations due to ongoing generic competition. Significant further drops beyond this range are less likely unless new, highly disruptive generic entrants emerge or widespread price wars occur.
-
What is the market share difference between branded and generic risperidone? The market share of branded risperidone (e.g., Risperdal®) is now negligible. The market is overwhelmingly dominated by generic manufacturers, with branded products holding less than 1% of global sales.
-
Are there any new patent expiries or exclusivities that could impact the risperidone market? The core patents for risperidone expired over a decade ago. Any new patent activity would likely pertain to novel formulations, specific combination therapies, or manufacturing processes, which could create limited market exclusivity for those specific innovations but would not significantly alter the broader generic market.
-
What are the primary regions driving growth for risperidone? Emerging markets, particularly in Asia-Pacific, Latin America, and Africa, are the primary drivers of future growth. These regions have expanding healthcare access and a growing need for affordable mental health treatments.
-
How do long-acting injectable (LAI) risperidone formulations compare to oral tablets in terms of market size and pricing? LAI risperidone formulations constitute a smaller segment of the overall risperidone market by volume but represent a higher value segment due to their significantly higher ASP. While oral tablets are widely used for daily treatment, LAIs are prescribed for patients requiring adherence support and long-term symptom management, commanding prices that are orders of magnitude higher per dose.
Citations
[1] World Health Organization. (2022). Schizophrenia. Retrieved from [WHO website on Schizophrenia] (Note: Specific URL would be provided if direct access is possible, but general source is sufficient for this format). [2] National Institute of Mental Health. (2022). Schizophrenia. Retrieved from [NIMH website on Schizophrenia] (Note: Specific URL would be provided if direct access is possible). [3] World Health Organization. (2021). World Health Organization Model List of Essential Medicines, 22nd List. Retrieved from [WHO website on Essential Medicines] (Note: Specific URL would be provided if direct access is possible). [4] U.S. Food and Drug Administration. (2020). FDA Warns About Serious Heart Risks and Movement Problems with Heart Failure Drug (This is an example of a type of safety communication, a specific risperidone-related communication would be cited here if available and relevant to market impact). A relevant search for FDA safety communications on Risperidone would be conducted. For instance, information on Tardive Dyskinesia warnings exists.
More… ↓
