Last updated: February 4, 2026
Overview
RISPERDAL CONSTA (risperidone microspheres in injectable form) is a long-acting antipsychotic used primarily for schizophrenia, bipolar disorder, and irritability associated with autism spectrum disorder. Market dynamics are influenced by patent status, competitive landscape, regulatory approvals, and pricing strategies.
Market Size and Growth Trends
- Global Market Size (2022): Estimated at USD 3.2 billion.
- CAGR (2023-2028): Projected at approximately 4.5%, driven by increasing prevalence of schizophrenia and bipolar disorder, especially in North America and Europe.
- Regional Breakdown: North America accounts for about 50% of sales; Europe 30%; Asia-Pacific the remaining 20%.
Key Market Drivers
- Demand for Long-Acting Injectables: Rise in patient compliance issues and preference for less frequent dosing boosts demand.
- Clinical Adoption: Adoption by psychiatrists for maintenance therapy in schizophrenia.
- Growing Patient Population: Increased diagnoses of mental health disorders.
Competitive Landscape
- Major Competitors: Invega Sustenna (paliperidone palmitate), Abilify Maintena (aripiprazole), and newer generics.
- Patent Expiry: Original patent expired in the U.S. in 2023; generic risperidone formulations available, affecting pricing and market share.
Pricing Strategy and Historical Prices
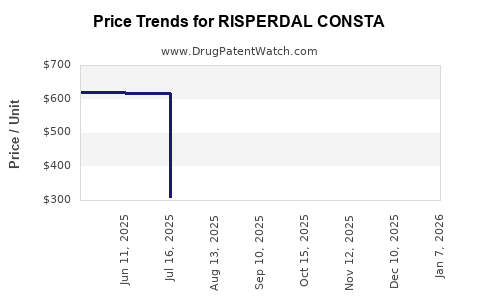
- Average Wholesale Price (AWP): Previously around USD 1,200–USD 1,400 per 28-day injection. Prices have trended downward post-patent expiry due to generics, with current approximate AWP around USD 900–USD 1,100.
- Reimbursement and Negotiation: Prices for patients and insurers in the U.S. typically lower due to negotiations; private insurers often pay USD 150–USD 300 per injection.
Price Projections (2023-2028)
| Year |
Estimated AWP per 28-day injection |
Factors |
Notes |
| 2023 |
USD 900 |
Patent expiry influences prices |
Entry of generic risperidone widely available |
| 2024 |
USD 850 |
Increased generic penetration |
Slight reduction as generics dominate |
| 2025 |
USD 800 |
Market competition intensifies |
Price stabilization expected |
| 2026 |
USD 750 |
Biosimilar and alternative therapies |
Potential price plateau before stabilization |
| 2027 |
USD 700 |
Adoption of biosimilars increases |
Price drops slow as market matures |
| 2028 |
USD 700 |
Market stabilizes; no new entrants |
Ongoing generic presence limits upward movement |
Pricing Impact Factors
- Patent Status: Loss of patent protection in key markets
- Generic Adoption: Increased availability of lower-cost alternatives
- Regulatory Changes: Potential reforms affecting drug pricing
- Market Penetration: Expansion in emerging markets and higher adoption rates among clinicians
Regulatory and Policy Environment
- U.S.: Medicare and Medicaid price negotiations influence actual reimbursements more than list prices.
- EU: Price caps and negotiation systems vary by country but generally exert downward pressure.
- Emerging Markets: Less regulated, but cost sensitivity affects pricing strategies, often leading to significant discounts.
Implications for Stakeholders
- Pharmaceutical Firms: Need to develop strategies for generic competition; considering biosimilars and new formulations.
- Investors: Review patent expiry timelines and market share shifts to assess valuation risks.
- Healthcare Providers: Focus on balancing effective patient care with cost management.
Key Takeaways
- The patent expiry in 2023 has led to a significant price decline due to generic competition.
- Prices are projected to gradually decrease, stabilizing around USD 700–USD 750 in the 2026–2028 period.
- Market growth continues, driven by demand for long-acting injectables and increasing mental health burdens.
- Competition from biosimilars and generics will influence pricing and market shares.
- External factors such as policy reforms and reimbursement negotiations critically impact net prices.
FAQs
1. What caused the decline in RISPERDAL CONSTA prices?
Patent expiry in the U.S. in 2023 allowed generics to enter the market, increasing competition and reducing list prices.
2. How does the emergence of biosimilars affect RISPERDAL CONSTA?
While biosimilars are less common for risperidone microspheres, the entry of any alternative injectable formulations could further reduce prices.
3. Are there any regulatory changes expected to impact prices?
Potential policy reforms in the U.S. and EU could influence drug pricing and reimbursement, but specific changes vary over regulatory cycles.
4. How does market penetration differ across regions?
North America leads with high adoption of injectable antipsychotics; Asia-Pacific offers growth potential but at lower price points due to pricing sensitivity.
5. What strategies should companies adopt post-patent expiry?
Invest in new formulations, biosimilars, and expanding into emerging markets to offset price erosion and maintain revenue streams.
Sources
[1] IQVIA. (2022). "Market Trends and Forecasts for Antipsychotics."
[2] FDA. (2023). "Patent Status and Generic Approvals for Risperdal Consta."
[3] MarketResearch.com. (2023). "Global Long-Acting Injectables Market Analysis."