Share This Page
Drug Price Trends for RIFABUTIN
✉ Email this page to a colleague
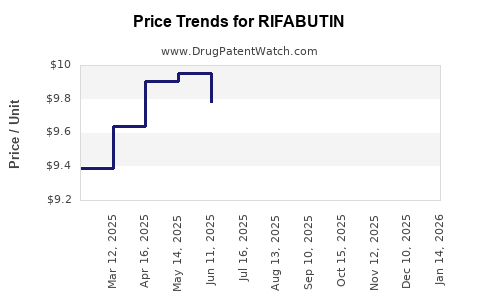
Average Pharmacy Cost for RIFABUTIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RIFABUTIN 150 MG CAPSULE | 70954-0041-10 | 9.17858 | EACH | 2026-03-18 |
| RIFABUTIN 150 MG CAPSULE | 59762-1350-01 | 9.17858 | EACH | 2026-03-18 |
| RIFABUTIN 150 MG CAPSULE | 70954-0041-10 | 8.96631 | EACH | 2026-02-18 |
| RIFABUTIN 150 MG CAPSULE | 59762-1350-01 | 8.96631 | EACH | 2026-02-18 |
| RIFABUTIN 150 MG CAPSULE | 70954-0041-10 | 9.28383 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
RIFABUTIN Market Analysis and Financial Projection
What is Rifabutin and Why Is It Significant?
Rifabutin is an antibiotic primarily used to treat Mycobacterium avium complex (MAC) infections, especially in HIV/AIDS patients. It belongs to the rifamycin class and is closely related to rifampin. Its role in tuberculosis (TB) management is limited but it can be used in cases where rifampin resistance or drug interactions prevent use of other first-line agents.
How Large Is the Rifabutin Market?
Global Market Size
The global rifabutin market was valued at approximately USD 150 million in 2022. The market is projected to grow at a compound annual growth rate (CAGR) of 4.5% over the next five years, reaching around USD 195 million by 2027.
Market Segmentation
- Therapeutic Applications: Mainly HIV-associated MAC infections (around 70%), with a smaller segment for prophylactic use (15%) and off-label uses including TB (15%).
- Geographic Distribution: North America accounts for approximately 45% of sales (US predominates). Europe contributes 25%, Asia-Pacific 20%, and the rest of the world 10%.
Key Market Drivers
- Increasing HIV patient populations, especially in Africa and Southeast Asia.
- Growing incidences of MAC infections in immunocompromised patients.
- Regulatory approval extensions for additional indications.
Competitive Landscape
Leading companies include:
- Taiho Pharmaceutical (Japan)
- Bristol-Myers Squibb (US)
- AbbVie (US, through acquisition of drug assets)
Few generic options exist, as patent protections have limited generic entry.
What Are The Key Price Trends and Projections?
Current Pricing
- Brand-name rifabutin (Mycobutin): Approximate wholesale price per 300 mg capsule is USD 50–70.
- Generic equivalents: Price points range from USD 20–40 per capsule, depending on supplier and market volume.
Price Dynamics (2020–2022)
- The price for brand-name rifabutin remained stable due to limited generic competition.
- Generic prices decreased by about 10-15% owing to increased manufacturing scale and market entry of new suppliers in select regions.
Projected Price Trends (2023–2027)
- Patent expiration for certain formulations is anticipated by 2024 in the US and EU, likely leading to a significant decrease in generic prices.
- Average wholesale prices for generics could fall by 25–40%, reaching USD 15–25 per capsule.
- Price erosion will be tempered by supply chain constraints and manufacturing costs.
Price Factors Affecting Future Projections
- Regulatory approvals for additional indications could influence volume and price.
- Market growth driven by demand in emerging regions.
- Supply chain issues such as raw material shortages may influence pricing stabilization or increases.
What Are the Key Regulatory and Policy Trends?
Regulatory Landscape
- US: The FDA approved Mycobutin (rifabutin) for MAC infections in 1992. No recent updates, but ongoing interest in expanding indications.
- Europe: The European Medicines Agency (EMA) approved rifabutin in 1995; certain countries permit off-label use for TB.
- Emerging Markets: Approval status varies; India and China have authorized generic manufacturing.
Policy Trends Impacting Market Dynamics
- Efforts to combat drug-resistant TB boost interest in rifabutin as an alternative.
- The push toward universal HIV treatment protocols increases demand.
- Price negotiations and reimbursement policies influence market access and profitability.
How Do Future Incentives or Barriers Affect the Market?
Incentives
- Expanding indications, including TB and prophylactic use.
- Development of combination therapies involving rifabutin.
- Expansion in emerging markets with high HIV burdens.
Barriers
- Potential side effects limit broad use.
- Price declines following patent expiry may reduce profit margins.
- Competition from newer agents with better safety profiles.
Key Takeaways
- The rifabutin market is valued at roughly USD 150 million (2022), with steady growth projected.
- Limited competition due to patent protections sustains higher prices; generics could reduce prices by up to 40% after patent expiration.
- Major markets are North America and Europe; emerging markets drive future growth.
- Price declines are expected post-2024, aligned with patent expiry and market entry of generics.
- Regulatory and policy developments focus on resistant TB, HIV, and cost-effective treatment options.
FAQs
1. When is the patent for rifabutin expected to expire?
Patents generally expire by 2024 in the US and EU, opening opportunities for generics.
2. What are the primary drivers of rifabutin market growth?
Rising HIV/AIDS prevalence, increased MAC infections, and regulatory approvals for new indications.
3. How does the price of rifabutin compare to other rifamycins?
Rifabutin is priced similarly to rifampin but typically costs more due to patent protections and brand premiums.
4. Are there significant risks in developing generic rifabutin?
Yes, including stringent regulatory requirements, potential side effects, and manufacturing complexities.
5. What therapeutic areas are likely to see increased use of rifabutin?
HIV/AIDS-related MAC infections and multidrug-resistant TB cases.
References
- MarketResearch.com, "Global Rifabutin Market Forecast," 2022.
- IQVIA, "Global Antimicrobial Market Trends," 2022.
- FDA and EMA approval documentation, 1992–1995.
- Industry analyst reports, "Generic Antibiotics Market Outlook," 2022.
- World Health Organization, "Global Tuberculosis Report," 2022.
More… ↓
