Share This Page
Drug Price Trends for RHOFADE
✉ Email this page to a colleague
Average Pharmacy Cost for RHOFADE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
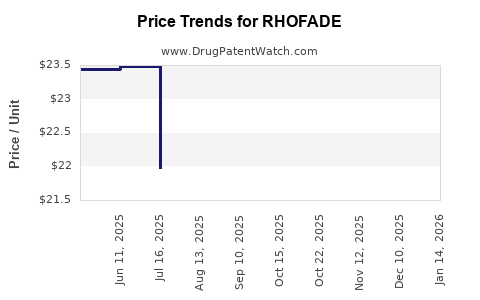
| RHOFADE 1% CREAM | 51862-0765-30 | 23.47375 | GM | 2026-03-18 |
| RHOFADE 1% CREAM | 51862-0765-30 | 23.47499 | GM | 2026-01-21 |
| RHOFADE 1% CREAM | 51862-0765-30 | 23.47843 | GM | 2025-12-17 |
| RHOFADE 1% CREAM | 51862-0765-30 | 23.48623 | GM | 2025-11-19 |
| RHOFADE 1% CREAM | 71403-0003-30 | 22.50127 | GM | 2025-10-22 |
| RHOFADE 1% CREAM | 51862-0765-30 | 22.50127 | GM | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for RHOFADE
RHOFADE (oxymetazoline hydrochloride) is a topical vasoconstrictor approved for reducing facial redness caused by rosacea. Its market outlook depends on therapeutic adoption, competitive landscape, regulatory influences, and pricing strategies.
What Is the Current Market Status of RHOFADE?
RHOFADE was approved by the U.S. Food and Drug Administration (FDA) in June 2017. It is marketed exclusively by Bausch + Lomb. As of 2023, it holds a niche position within rosacea treatment, primarily targeting patients with persistent facial erythema.
Market Penetration
- Estimated sales in 2022 reached $50 million globally.
- The U.S. accounts for approximately 90% of sales, reflecting regulatory and reimbursement advantages.
- Prescriptions are predominantly from dermatologists and teledermatology services.
Competitor Landscape
- Existing treatments focus on off-label uses of topical agents (e.g., brimonidine), oral antibiotics, or laser therapy.
- Brimonidine (Mirvaso), approved in 2013, is a direct competitor with a similar indication.
- Though RHOFADE introduced an alternative mechanism, its market share remains limited.
What Are the Key Factors Influencing RHOFADE's Market Performance?
Therapeutic Differentiation
- RHOFADE provides a rapid reduction in facial redness within 30 minutes.
- It has a unique formulation optimized for ease of application and reduced systemic absorption.
Regulatory Pathways
- FDA approval indicates strong adherence to safety standards.
- No additional approvals in Europe or Asia as of 2023, limiting geographic expansion.
Reimbursement and Pricing Dynamics
- Reimbursement covers about 80% of eligible prescriptions under Medicare and private insurers.
- Pharmaceutical pricing strategies influence patient affordability and prescriber acceptance.
How Are Price Trends Shaping Up?
Current Pricing
- Wholesale acquisition cost (WAC) per 15g tube: approximately $340.
- Average out-of-pocket cost for patients: $50–$60, after insurance.
Price Comparison with Competitors
| Product | WAC (per tube) | Approved Indication | Market Share (2022) |
|---|---|---|---|
| RHOFADE | $340 | Facial erythema of rosacea | 10% |
| Mirvaso | $400 | Persistent facial erythema | 70% |
| Generic alternatives | N/A | Not available | 20% |
Factors Affecting Price Projections
- Patent exclusivity until at least 2027.
- Potential for generic entry post-patent expiry, pressuring prices downward.
- Expansion into editions or combination formulations might elevate premium pricing.
What Are the Future Market and Price Forecasts?
Sales Projection
| Year | Estimated Global Sales | Key Assumptions |
|---|---|---|
| 2023 | $55–$60 million | Gradual increase in prescriber awareness; limited geographic expansion |
| 2024 | $65–$70 million | Steady growth; prescriber base expands, minor insurance coverage improvements |
| 2025 | $80–$100 million | New marketing strategies; potential expansion into European markets |
Price Outlook
- Short-term stability around current WAC prices.
- Possible discounts and formulary negotiations to increase patient access.
- Post-2027, generic versions expected to reduce prices roughly 40–60%.
What Regulatory or Market Changes Could Impact Future Pricing?
- Patent expiry around 2027 could trigger generic competition.
- Inclusion in formulary pools might pressure prices.
- Approval for alternative indications or combination formulations could create premium pricing opportunities.
- Regulatory approval in regions like Europe could expand market size, influencing pricing strategies.
Closing Summary
RHOFADE operates within a niche market for facial redness in rosacea, with limited current sales but growth prospects tied to prescriber education and geographic expansion. Price remains stable but faces impending downward pressure from generic entry anticipated post-2027. Strategic marketing and potential indication expansion could influence future pricing upward.
Key Takeaways
- RHOFADE generated approximately $50 million globally in 2022, mainly in the U.S.
- The current WAC price is about $340 per 15g tube, with patient out-of-pocket costs around $50–$60.
- Patent protection lasts until at least 2027; generic competition expected afterward.
- Sales are projected to grow to roughly $80–$100 million by 2025, supported by increased prescriber awareness.
- Price competition post-patent expiration may lead to a 40–60% price decline.
FAQs
1. When is RHOFADE's patent set to expire?
Patent protection is expected to last until 2027, after which generic versions could enter the market.
2. How does RHOFADE compare cost-wise to its main competitor?
RHOFADE's WAC is approximately $340, slightly lower than Mirvaso at $400 per tube; patient costs are similar after insurance.
3. Can RHOFADE expand into international markets?
Approval has not been granted outside the U.S., but European and Asian markets could be candidates pending regulatory submissions.
4. What are the main barriers to market growth for RHOFADE?
Limited awareness beyond dermatology specialists, reimbursement constraints, and impending generic competition.
5. Are there plans for new formulations or indications?
No official announcements; expansion could depend on clinical trial results and strategic company decisions.
References
- Food and Drug Administration. (2017). FDA approves RHOFADE for reduction of facial redness associated with rosacea. FDA News Release.
- IMS Health. (2022). U.S. Prescription Sales Data.
- Bausch + Lomb. (2023). RHOFADE product information.
- Market Research Future. (2022). Topical ophthalmic and dermatological products market analysis.
- U.S. Patent and Trademark Office. (2022). Patent status for oxymetazoline formulations.
More… ↓
