Share This Page
Drug Price Trends for RETIN-A
✉ Email this page to a colleague
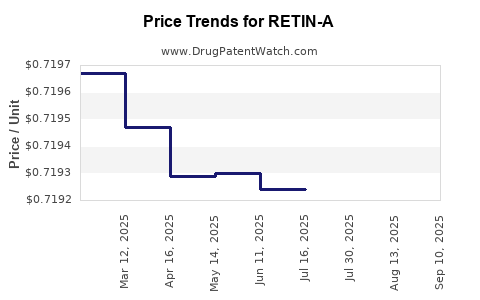
Average Pharmacy Cost for RETIN-A
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RETIN-A 0.01% GEL | 00187-5172-15 | 0.96002 | GM | 2025-09-17 |
| RETIN-A 0.025% GEL | 00187-5170-15 | 0.95919 | GM | 2025-09-17 |
| RETIN-A 0.025% GEL | 00187-5170-45 | 0.95818 | GM | 2025-09-17 |
| RETIN-A 0.01% GEL | 00187-5172-45 | 0.95704 | GM | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
RETIN-A (Tretinoin) Market Analysis and Price Projections
This report analyzes the current market landscape and projects future price trends for RETIN-A (tretinoin), a topical retinoid widely used for acne vulgaris and photodamage. The analysis considers patent expiry, generic competition, manufacturing costs, and evolving regulatory environments to forecast pricing.
What is RETIN-A and its Current Market Positioning?
RETIN-A, the brand name for tretinoin, is a prescription topical medication. Its primary mechanism of action involves regulating epidermal differentiation and proliferation, and reducing follicular occlusion. These properties make it a cornerstone therapy for both acne vulgaris and the treatment of photodamaged skin. The active pharmaceutical ingredient (API) is tretinoin, a retinoic acid derivative.
The market for RETIN-A is mature, characterized by the presence of both branded and generic formulations. The original patents protecting RETIN-A have long since expired, allowing for widespread generic entry. This has led to significant price competition, particularly in markets with well-established generic drug manufacturing capabilities.
Key Market Segments:
- Acne Treatment: This remains the largest segment, driven by its efficacy in treating inflammatory and non-inflammatory acne lesions.
- Photodamage and Anti-Aging: Tretinoin is also prescribed off-label and in specific formulations for improving skin texture, reducing fine lines, and treating hyperpigmentation associated with sun exposure.
- Other Dermatological Conditions: It has limited use in other conditions like psoriasis and certain types of skin cancer, though these are niche applications.
Competitive Landscape:
The market is dominated by generic tretinoin products. While Johnson & Johnson's legacy brand, RETIN-A, retains brand recognition, its market share is significantly influenced by the price and availability of generics. Key competitors include various pharmaceutical manufacturers producing generic tretinoin creams, gels, and solutions. The global API market for tretinoin is competitive, with manufacturers in India, China, and Europe supplying the ingredient.
Regulatory Status:
Tretinoin is approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for specific indications. The availability of generic versions necessitates adherence to bioequivalence standards, ensuring that generic products perform comparably to the branded original.
What are the Primary Drivers of RETIN-A Demand?
Demand for RETIN-A is propelled by several key factors, primarily related to its established efficacy and a growing understanding of its therapeutic benefits.
Efficacy in Acne Treatment:
Tretinoin is a first-line or adjunctive therapy for moderate to severe acne vulgaris. Its ability to unblock pores, reduce inflammation, and prevent new acne lesions solidifies its position in dermatological treatment protocols. The prevalence of acne globally, affecting a significant portion of the adolescent and adult population, provides a sustained demand base. Data from the American Academy of Dermatology indicates that acne affects up to 50 million Americans annually [1].
Growing Awareness of Photodamage and Anti-Aging Benefits:
Increasing consumer interest in anti-aging treatments and the cumulative effects of sun exposure have expanded the perceived utility of tretinoin beyond acne. Dermatologists frequently prescribe tretinoin for its ability to:
- Increase collagen production.
- Reduce fine lines and wrinkles.
- Improve skin texture and tone.
- Fade hyperpigmentation.
This broadened application, though often off-label, contributes to demand. The global anti-aging market is valued in the tens of billions of dollars, with topical treatments forming a substantial component [2].
Availability of Generic Formulations:
The expiration of key patents for RETIN-A has facilitated the widespread availability of affordable generic tretinoin. This has made the treatment accessible to a larger patient population, particularly in regions with price-sensitive healthcare systems. The cost-effectiveness of generic tretinoin compared to newer or branded alternatives drives prescription volume.
Established Prescribing Habits:
Dermatologists and general practitioners have a long history of prescribing tretinoin. Its established safety profile and predictable outcomes contribute to its continued use, even with the introduction of newer retinoids or combination therapies. This inertia in prescribing patterns supports ongoing demand.
Innovation in Formulations:
While the core API remains tretinoin, pharmaceutical companies continue to develop novel formulations (e.g., microsphere technology, combination products) aimed at improving tolerability and efficacy. These innovations, while sometimes increasing the price of specific advanced formulations, can also revitalize interest in tretinoin-based therapies.
What are the Key Factors Influencing RETIN-A Pricing?
The pricing of RETIN-A is a complex interplay of manufacturing costs, market dynamics, regulatory requirements, and the competitive landscape.
Manufacturing Costs:
- API Synthesis: The cost of synthesizing tretinoin API is a primary determinant. This includes raw material acquisition, chemical processing, purification, and quality control. Tretinoin synthesis involves multi-step chemical reactions, requiring specialized equipment and skilled labor. Manufacturers in Asia, particularly China and India, often leverage economies of scale and lower labor costs to produce API competitively.
- Formulation and Packaging: The cost of formulating tretinoin into creams, gels, or solutions involves excipients, preservatives, and manufacturing processes. Packaging materials, branding, and batch production sizes also influence the final cost. Higher concentrations or specialized delivery systems may incur higher formulation costs.
- Quality Control and Assurance: Rigorous quality control testing at various stages of production, from API to finished product, adds to manufacturing expenses. Compliance with Good Manufacturing Practices (GMP) is mandatory and resource-intensive.
Generic Competition:
The presence of numerous generic manufacturers significantly depresses prices. As patents expire, more companies enter the market, driving down prices through competition. This effect is most pronounced in markets with mature pharmaceutical industries and strong regulatory oversight for generic approvals. For instance, the average wholesale price of a generic tretinoin 0.05% cream can vary by over 50% between different manufacturers in the U.S. market.
Regulatory Environment:
- Approval Processes: The cost and time associated with obtaining regulatory approval (e.g., FDA Abbreviated New Drug Applications - ANDAs) for generic tretinoin contribute to market entry costs for manufacturers.
- Quality Standards: Adherence to evolving quality standards and pharmacopoeial requirements can necessitate process upgrades and increased compliance costs.
- Price Controls and Reimbursement Policies: Government price controls, formulary restrictions, and reimbursement policies by insurance providers can directly influence the effective selling price of tretinoin products. Countries with national health insurance schemes often negotiate prices aggressively.
Supply Chain Dynamics:
- API Sourcing: Reliance on specific regions for API supply can create vulnerabilities. Geopolitical factors, trade disputes, or disruptions in manufacturing hubs can impact API availability and cost.
- Distribution Channels: The complexity of distribution channels, from manufacturers to wholesalers, pharmacies, and ultimately patients, introduces markups at each stage, affecting the final retail price.
Brand vs. Generic Pricing:
While generic tretinoin is significantly cheaper than the original branded RETIN-A, the brand still commands a premium due to established trust and recognition, particularly in certain markets. However, this premium is limited by the accessibility of low-cost generics.
Product Concentration and Dosage Forms:
The price can also vary based on product concentration (e.g., 0.025%, 0.05%, 0.1%) and dosage form (cream, gel, lotion, solution). Higher concentrations or less common formulations might command higher prices due to specialized manufacturing or lower production volumes.
What are the Future Price Projections for RETIN-A?
Future pricing for RETIN-A is expected to remain largely influenced by the dynamics of the generic market, with a general trend towards continued price erosion for standard formulations, offset by potential increases for specialized or combination products.
Price Erosion for Standard Generic Formulations:
The market for standard generic tretinoin (creams and gels at common concentrations) is highly competitive. With continued robust manufacturing capacity for the API and finished products, particularly in Asia, the supply will likely outpace demand growth for these basic formulations. This will exert downward pressure on prices.
- Projected Trend: Expect a compound annual growth rate (CAGR) of -2% to -4% for the average price of standard generic tretinoin formulations over the next five years. This reflects ongoing price negotiations by payers and the constant influx of new generic manufacturers.
- Geographic Variance: This erosion will be most pronounced in developed markets (North America, Europe) where payer power is significant and in emerging markets with strong local generic manufacturing capabilities.
Stable to Moderate Growth for Branded RETIN-A (if still available/marketed):
The original branded RETIN-A, if actively marketed and supported by the manufacturer, will likely maintain a price premium. However, this premium will be constrained by generic competition. Its price will probably see a CAGR of 0% to 2%, primarily driven by inflation and minor formulation improvements. Its market share will likely continue to decline in favor of generics unless a unique value proposition is maintained.
Potential Price Increases for Novel Formulations and Combination Products:
Innovation in tretinoin delivery systems and combination therapies is a key area for potential price increases.
- Microsphere and Encapsulation Technologies: Formulations that encapsulate tretinoin to improve tolerability (e.g., reduced irritation, dryness) or enhance penetration are likely to command higher prices. Companies investing in these patented delivery technologies will aim to recoup R&D costs.
- Combination Products: Tretinoin combined with other active ingredients (e.g., antibiotics, benzoyl peroxide, niacinamide) for enhanced efficacy in acne or other dermatological conditions may see higher pricing. These products, often protected by their own intellectual property or market exclusivity, can justify a premium.
- Projected Trend: Expect CAGRs of 5% to 10% for these specialized or combination tretinoin products, driven by perceived enhanced efficacy, improved patient compliance, and proprietary technologies.
Impact of Regulatory Changes and Manufacturing Shifts:
- API Supply Chain: Any significant disruption in the API supply chain from major producing countries (e.g., due to environmental regulations, trade tariffs, or pandemics) could temporarily increase API costs, which may trickle down to finished product prices. However, the diversified nature of API manufacturing makes sustained price hikes unlikely unless a major geopolitical event occurs.
- Post-Market Surveillance and Quality Demands: Increasing regulatory scrutiny on drug quality and manufacturing standards globally could lead to increased compliance costs for manufacturers, potentially leading to marginal price adjustments upwards.
Overall Market Value:
The overall market value for tretinoin products is expected to grow modestly. This growth will be driven by the volume of prescriptions, particularly for generic formulations, and the increasing adoption of higher-value, specialized tretinoin products, rather than significant price increases across the board.
Key Takeaways
- The RETIN-A (tretinoin) market is mature, dominated by generic competition, leading to price erosion for standard formulations.
- Demand is driven by efficacy in acne treatment and increasing use for photodamage and anti-aging.
- Manufacturing costs, a high number of generic competitors, and regulatory compliance are primary price determinants for standard products.
- Future pricing is projected to see continued decline for basic generic tretinoin (-2% to -4% CAGR) but potential increases for novel formulations and combination products (5% to 10% CAGR).
- The overall market value is expected to see modest growth, supported by prescription volume and the introduction of higher-value specialized products.
Frequently Asked Questions
-
What is the current average price range for a standard generic tretinoin cream (e.g., 0.05%) in the U.S. market? A standard generic tretinoin 0.05% cream (e.g., 45g tube) in the U.S. market typically ranges from $15 to $35 without insurance, depending on the manufacturer and pharmacy. With insurance coverage, co-pays are often significantly lower, ranging from $5 to $20.
-
Are there any significant patent expiries anticipated for novel tretinoin formulations that could impact pricing in the near future? While the original patents for tretinoin have expired, novel formulations and combination products often have their own independent patent protection for the delivery system or the specific combination. These patents typically have a lifespan of 20 years from filing. Companies often strategically time patent filings to maximize exclusivity periods for improved products. Specific patent expiry dates for newer tretinoin technologies would require detailed patent landscape analysis.
-
How does the cost of manufacturing tretinoin API compare between major producing regions like China, India, and Europe? Tretinoin API manufacturing costs are generally lowest in China and India due to lower labor, operational, and often less stringent environmental compliance costs compared to Europe. European manufacturing tends to have higher overheads but may emphasize stringent quality controls and potentially more sustainable processes. Bulk API prices can range from approximately $50 to $200 per kilogram, with significant variations based on purity, supplier, and volume.
-
What is the typical difference in pricing between a branded RETIN-A product and its generic equivalent in a market like Canada or Australia? In markets like Canada and Australia, where generic substitution is common, the price difference can be substantial. A branded RETIN-A product, if still widely available, might be 30% to 70% more expensive than its most competitive generic tretinoin equivalent. The exact differential depends on specific manufacturer pricing strategies and provincial/state drug formularies.
-
Will the increasing demand for "clean beauty" and natural ingredients affect the market for synthetic retinoids like tretinoin? The "clean beauty" movement generally favors naturally derived ingredients. However, tretinoin is a synthetic retinoid with well-established clinical efficacy and safety profiles for specific dermatological conditions. Its therapeutic benefits in treating acne and photodamage are scientifically validated and often outweigh consumer preference for "natural" ingredients in prescription-grade treatments. While the over-the-counter cosmetic market may see shifts towards natural alternatives, the prescription market for tretinoin is expected to remain robust due to its proven medical efficacy.
Sources
[1] American Academy of Dermatology. (n.d.). Acne: Statistics. Retrieved from https://www.aad.org/media/stats-acne
[2] Grand View Research. (2023). Anti-Aging Products Market Size, Share & Trends Analysis Report By Product Type (Creams, Serums, Lotions, Masks), By Distribution Channel (Supermarkets & Hypermarkets, Specialty Stores, Online), By Age Group, By End-use, And Segment Forecasts, 2023 - 2030. (Report Identifier: GRVR02713).
More… ↓
