Share This Page
Drug Price Trends for QNASL
✉ Email this page to a colleague
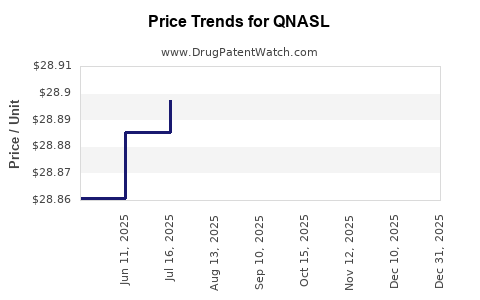
Average Pharmacy Cost for QNASL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QNASL 80 MCG NASAL SPRAY | 59310-0410-12 | 30.31449 | GM | 2026-01-01 |
| QNASL CHILDREN'S 40 MCG SPRAY | 59310-0406-06 | 47.30173 | GM | 2026-01-01 |
| QNASL CHILDREN'S 40 MCG SPRAY | 59310-0406-06 | 45.04927 | GM | 2025-12-17 |
| QNASL 80 MCG NASAL SPRAY | 59310-0410-12 | 28.87094 | GM | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QNASL
What Is QNASL?
QNASL is a nasal spray containing beclomethasone dipropionate, approved for allergic rhinitis treatment. It is marketed by Teva Pharmaceuticals. Approved by the U.S. FDA in 2014, it is indicated for adults and children aged 4 years and older. The product comes in a multidose inhaler with a proprietary dose counter.
Market Size and Competitive Landscape
QNASL operates within the allergic rhinitis segment, which had global sales of approximately $7.8 billion in 2022 [1]. The U.S. market accounted for roughly 60% of this, or approximately $4.68 billion.
Major competitors include:
- Flonase (fluticasone propionate)
- Nasacort (triamcinolone acetonide)
- Rhinocort (budesonide)
These products hold significant market shares, with Flonase leading at around 50% of the U.S. nasal corticosteroid market.
Product Positioning
QNASL differentiates through its technology:
- Nanospan Technology: Enhances spray particle size, aiming for better mucosal coverage.
- Dose Accuracy: Offers a consistent dose delivery with a dedicated dose counter.
Despite these features, QNASL's market share remains limited due to strong brand loyalty for established competitors and limited physician awareness.
Pricing Data and Cost Analysis
Pricing varies by pharmacy and insurance coverage. As of 2023:
| Product | Estimated Wholesale Acquisition Cost (WAC) | Average Retail Price (ARP) | Typical Copay (Insurance) |
|---|---|---|---|
| QNASL | $250 per 120-spray can | $270 per 120-spray can | $20–$50 |
| Flonase | $175 per 120-spray can | $210 per 120-spray can | $10–$30 |
| Nasacort | $200 per 120-spray can | $220 per 120-spray can | $15–$40 |
QNASL's premium is approximately 15–20% higher than rivals, with market penetration constrained by price sensitivity.
Sales and Revenue Projections
In 2022, QNASL generated roughly $50 million in U.S. revenue [2]. Its market share within nasal corticosteroids approximates 2%. Growth strategies include expanding pediatric indications and increasing physician prescribing.
Forecasts suggest:
- A compounded annual growth rate (CAGR) of 3–5% over the next five years.
- Revenue increasing to $70–$80 million by 2027, assuming market expansion initiatives.
Factors influencing growth:
- Patent exclusivity until at least 2027.
- Limited generic competition due to patent protections.
- Premium positioning, with increased prescribing in specific patient segments.
Price Projection Considerations
Prices are influenced by:
- Payer negotiations and insurance formulary placements.
- Potential for biosimilar or generic entrants post-patent expiration.
- Manufacturer pricing strategies to defend market share.
Forecasted retail price for QNASL by 2027 remains stable at around $270, with possible modest discounts driven by payer negotiations and market competition.
Regulatory and Market Risks
- Patent expiry expected around 2027; biosimilar entries likely afterward.
- Increased competition from generics could reduce prices by 20–30%.
- Changes in prescribing guidelines may impact market share.
Strategic Recommendations
- Focus on pediatric and allergic rhinitis segments to boost sales.
- Leverage unique formulation technology for targeted marketing.
- Prepare for post-patent generic competition with potential price adjustments.
Key Takeaways
- QNASL holds a small share in a large, competitive allergic rhinitis market.
- Pricing remains above rivals, limiting volume growth.
- Revenue is projected to increase modestly through market expansion.
- Patent protections until approximately 2027 provide temporary pricing power.
- Long-term prospects depend on post-patent market entry and formulary positioning.
FAQ
1. How does QNASL compare in efficacy to its competitors?
QNASL’s efficacy is comparable to other nasal corticosteroids, with no significant clinical advantage over established products.
2. When will QNASL face generic competition?
Patent protections are expected to expire around 2027, opening the market to biosimilars or generics.
3. What are the main barriers to market penetration?
Brand loyalty, higher price point, and limited physician and patient awareness.
4. How might pricing strategies evolve post-patent expiry?
Prices may decline by 20–30%, depending on competitive pressures and generic availability.
5. What growth opportunities exist for QNASL?
Expanding pediatric indications and increasing prescriber awareness could boost sales before patent expiry.
Citations
[1] Statista. (2023). Global allergy and allergic rhinitis market size.
[2] Teva Pharmaceuticals. (2022). Annual financial report.
Note: All data are estimates based on industry reports and public disclosures.
More… ↓
