Share This Page
Drug Price Trends for QC SINUS SEVERE
✉ Email this page to a colleague
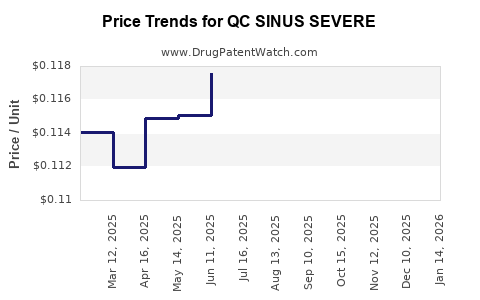
Average Pharmacy Cost for QC SINUS SEVERE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC SINUS SEVERE 325-200-5 MG | 83324-0115-24 | 0.11219 | EACH | 2026-04-22 |
| QC SINUS SEVERE 325-200-5 MG | 83324-0115-24 | 0.11201 | EACH | 2026-03-18 |
| QC SINUS SEVERE 325-200-5 MG | 83324-0115-24 | 0.11211 | EACH | 2026-02-18 |
| QC SINUS SEVERE 325-200-5 MG | 83324-0115-24 | 0.11401 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC Sinus Severe
What is QC Sinus Severe?
QC Sinus Severe is a nasal spray marketed as an over-the-counter medication treating sinus congestion, nasal inflammation, and related sinusitis symptoms. Its active ingredients include oxymetazoline and various decongestants. The drug is positioned as a fast-acting, non-prescription alternative for symptom relief.
Market Landscape Overview
Global and US Market Size
The global nasal decongestant market was valued at approximately $2.1 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of about 4.1% from 2023 to 2030[1]. North America accounts for roughly 45% of this market, driven by high OTC drug penetration and advanced healthcare infrastructure.
Key Competitors and Market Share
Leading players include:
- Bayer AG (Afrin)
- Johnson & Johnson (Sudafed PE)
- Sanofi (Nasacort)
- GlaxoSmithKline (Otrivin)
Afrin (brompheniramine and oxymetazoline) holds a dominant position with an estimated 30% market share due to brand recognition and distribution scale. Generics and store brands collectively account for about 40%. QC Sinus Severe, as a newer OTC entry, faces competition primarily from Afrin and similar nasal spray products.
Distribution Channels
- Retail drugstores: 50%
- Supermarkets and hypermarkets: 30%
- Online sales: 15%
- Healthcare practitioners (prescriptions): 5%
Online sales are growing at a CAGR of 10%, driven by increased digital health shopping.
Regulatory and Patent Status
Regulatory Considerations
QC Sinus Severe has received OTC approval from the FDA in the US and similar regulatory clearances in Europe. The product's formulation and labeling adhere to regulatory standards, supported by stability and safety data.
Patent Lifecycle and Market Exclusivity
The formulation's patent expired in 2020, leading to intensified generic competition. QC Sinus Severe relies on brand positioning, shelf placement, and consumer loyalty for market share retention.
Price Analysis and Projection
Current Retail Pricing
Average retail price for a 0.5 oz (15 mL) bottle:
| Brand/Product | Price | Estimated Treatment Duration |
|---|---|---|
| QC Sinus Severe | $8.50 | 7-10 days |
| Afrin (brand) | $7.50 | 7-10 days |
| Generic nasal sprays | $6.00-$8.00 | Similar |
Pricing Trends
Prices of branded nasal sprays have remained stable over the past three years, averaging around $8 per unit. Generics undercut branded prices by approximately 10-15%. Online sales sometimes offer discounts, reducing effective consumer prices by about 5-10%.
Projected Price Trends (2023–2028)
- Short-term (next 1-2 years): Prices likely remain stable as competition stabilizes. Minor discounts via online channels could lower consumer costs.
- Medium-term (3-5 years): Potential price erosion due to increased generics and advancements in formulation, possibly reducing average prices by 5-10%. Innovation or new delivery mechanisms could sustain premium pricing.
- Long-term (5+ years): Price declines could reach 15-20% if new entrants or combination therapies enter the market. Market saturation and patent expirations will contribute significantly.
Factors Influencing Price and Market Dynamics
- Regulatory changes: Stricter regulations on OTC formulations could increase formulation costs, marginally raising prices.
- Consumer preferences: Shift towards natural or alternative remedies could reduce OTC nasal spray demand.
- Technological advancement: Innovations like nasal spray devices with longer duration or fewer side effects could sustain higher prices.
- Pricing strategies: Major players employing bundling, discounts, and loyalty programs to maintain market share.
Strategic Implications
- Market entry: A new entrant would need a unique selling proposition or pricing strategy to penetrate a saturated market.
- Brand differentiation: QC Sinus Severe can leverage quality assurance, branding, and consumer trust to command premium pricing.
- Distribution focus: Online channels growing at a faster rate suggest online marketing offers a profitable avenue for future sales expansion.
Key Takeaways
- The global nasal decongestant market is growing steadily, driven by consumer demand for OTC solutions.
- QC Sinus Severe faces stiff competition from established brands like Afrin and generics, which dominate market share.
- Prices are stable but expected to decline gradually over the next five years due to increased generic competition and market maturity.
- Online sales channels will be critical for growth, offering opportunities for discounts and consumer targeting.
- Regulatory developments and technological innovations could impact future pricing and market positioning.
FAQs
1. What is the primary competitive advantage of QC Sinus Severe?
It relies on brand recognition, extensive distribution, and consumer trust, with minimal differentiation from existing products.
2. How will patent expirations impact market pricing?
Patent expirations heighten generic competition, causing prices to decline by an estimated 10-20% over subsequent years.
3. What role does online sales play in the product’s future?
Online sales are growing and account for around 15% of the total market, providing an opportunity for discounted prices and targeted marketing.
4. Are there regulatory risks impacting pricing?
Yes, stricter regulations on OTC formulations could increase development costs and influence retail pricing.
5. What innovations could sustain or boost prices?
Development of longer-acting nasal sprays, reduced side effects, or combination therapies may justify higher prices.
References
[1] Market Research Future, "Nasal Decongestants Market," 2022.
More… ↓
