Share This Page
Drug Price Trends for QC OMEPRAZOLE MAG DR
✉ Email this page to a colleague
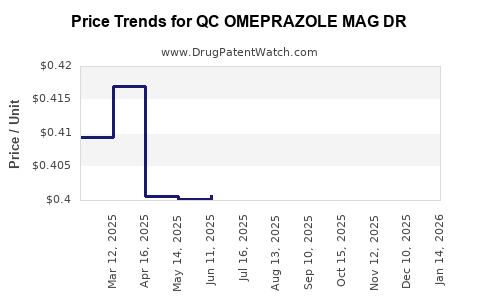
Average Pharmacy Cost for QC OMEPRAZOLE MAG DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC OMEPRAZOLE MAG DR 20 MG TAB | 83324-0011-14 | 0.42190 | EACH | 2026-03-18 |
| QC OMEPRAZOLE MAG DR 20 MG TAB | 83324-0011-42 | 0.42190 | EACH | 2026-03-18 |
| QC OMEPRAZOLE MAG DR 20 MG TAB | 83324-0011-14 | 0.40896 | EACH | 2026-02-18 |
| QC OMEPRAZOLE MAG DR 20 MG TAB | 83324-0011-42 | 0.40896 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC OMEPRAZOLE MAG DR
What is QC OMEPRAZOLE MAG DR?
QC OMEPRAZOLE MAG DR is a delayed-release formulation of omeprazole, a proton pump inhibitor (PPI) used for treating gastroesophageal reflux disease (GERD), Zollinger-Ellison syndrome, and peptic ulcers. It is typically marketed as a prescription medication, with specific regulatory approvals depending on the country.
Market Landscape
Current Market Size
The global PPI market, valued at approximately USD 8.5 billion in 2022, is projected to reach USD 11.3 billion by 2028, growing at a Compound Annual Growth Rate (CAGR) of 4.9% [1].
Key Competition
The market features several established brands, including:
- Nexium (esomeprazole)
- Prilosec (omeprazole)
- Prevacid (lansoprazole)
Biotech and generic manufacturers are actively expanding access through patent expirations.
Regional Insights
- North America accounts for roughly 40% of the global PPI market.
- Europe contributes 25%–30%, driven by high prevalence of GERD.
- Asia-Pacific exhibits rapid growth, with a CAGR of approximately 6%, fueled by increasing urbanization and healthcare access.
Patent and Regulatory Status
- Original patents for omeprazole expired in 2009 in most regions, leading to broad generic availability.
- Specialty formulations like delayed-release (DR) versions are often protected by new patents or exclusivities, influencing market entry barriers.
Price Trends and Projections
Current Pricing Dynamics
- Generic omeprazole: USD 0.10–0.20 per pill retail.
- Brand-name equivalents: USD 1.50–2.50 per pill.
- Specialized formulations like QC OMEPRAZOLE MAG DR: Estimated at USD 3.00–4.50 per tablet, reflecting patent protections and manufacturing costs.
Influencing Factors
- Introduction of generics reduces prices by 60–80% within two years of patent expiry.
- Regulatory exclusivity prolongs higher pricing for branded drugs.
- Manufacturing costs influence retail prices; delayed-release formulations tend to be more expensive due to complex manufacturing processes.
Price Projections (Next 5 Years)
| Year | Estimated Price Range (USD per tablet) | Notes |
|---|---|---|
| 2023 | USD 3.00–4.50 | Current market price |
| 2024 | USD 2.80–4.20 | Increased generic competition begins |
| 2025 | USD 2.40–3.80 | Patent protection challenges financial stability |
| 2026 | USD 2.00–3.50 | Entry of new generics in key markets |
| 2027 | USD 1.80–3.00 | Mature generic market with price erosion |
Factors That Could Alter Projections
- Regulatory changes: Fast-track approvals or extended exclusivity can sustain higher prices.
- Market entry delays: Manufacturing hurdles or patent litigations could prolong branded pricing.
- Disease prevalence: Rising GERD and ulcer incidences escalate demand, supporting prices.
- Emergence of substitutes: New drug classes could erode PPI market share and prices.
Market Drivers and Risks
Key Drivers
- Increasing prevalence of GERD, obesity, and NSAID use.
- Expanding aging populations.
- Growing awareness and diagnosis rates, especially in emerging markets.
- Prescriber preference for branded formulations in certain regions.
Risks
- Patent expirations enabling generic entry.
- Regulatory approvals for alternative therapies, such as H2-receptor antagonists or novel acid blockers.
- Pricing pressures from healthcare systems aiming to reduce costs.
Strategic Considerations
- Securing patent protection or exclusivities can sustain higher prices.
- Geographic expansion into emerging markets enhances revenue prospects.
- Developing combination therapies or innovative delivery forms can differentiate offerings.
Summary
QC OMEPRAZOLE MAG DR faces mounting competition from generics, which are expected to rapidly drive prices downward over the next five years. Near-term prices remain elevated due to patent protections and formulation complexity. Market growth hinges on increasing disease burden and regional healthcare expenditure, particularly in Asia-Pacific.
Key Takeaways
- The global PPI market is expanding at approximately 4.9% annually, with omeprazole remaining a core segment.
- Current prescription prices for QC OMEPRAZOLE MAG DR are around USD 3.00–4.50 per tablet.
- Prices are projected to decline to approximately USD 1.80–3.00 by 2027, primarily due to generic competition.
- Patent status, regulatory policies, and regional disease prevalence heavily influence pricing dynamics.
- Market entry barriers include manufacturing complexity and patent protections, which sustain higher prices temporarily.
FAQs
Q1: How long do patent protections last for formulations like QC OMEPRAZOLE MAG DR?
A1: Patents typically last 20 years from filing, but extension or exclusivity periods vary and can be extended through legal strategies or regulatory incentives.
Q2: What factors most influence the price decline of branded omeprazole formulations?
A2: Patent expiry and subsequent generic entry are primary drivers, along with healthcare policy pressures and market competition.
Q3: Are new delivery methods affecting the market for delayed-release PPIs?
A3: Yes, innovations such as granules, powders, or combination pills can provide competitive advantages but typically take time to penetrate the market.
Q4: Which regions have the highest growth potential for QC OMEPRAZOLE MAG DR?
A4: Asia-Pacific exhibits the most rapid growth, driven by rising GERD prevalence and expanding healthcare infrastructure.
Q5: How do regulatory changes impact future pricing?
A5: Regulations that streamline approval or extend exclusivity can sustain higher prices, while policies favoring generics accelerate price declines.
References
[1] MarketsandMarkets. (2022). Proton Pump Inhibitors Market by Compound, Drug Class, Distribution Channel, and Region. Retrieved from https://www.marketsandmarkets.com
More… ↓
