Share This Page
Drug Price Trends for QC NO DRIP NASAL MIST
✉ Email this page to a colleague
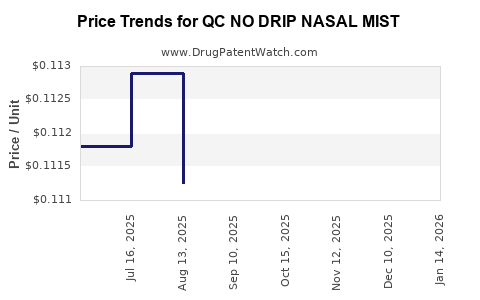
Average Pharmacy Cost for QC NO DRIP NASAL MIST
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC NO DRIP NASAL MIST 0.05% | 83324-0274-01 | 0.10326 | ML | 2026-04-22 |
| QC NO DRIP NASAL MIST 0.05% | 83324-0211-01 | 0.10326 | ML | 2026-04-22 |
| QC NO DRIP NASAL MIST 0.05% | 83324-0211-01 | 0.10632 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
QC NO DRIP NASAL MIST Market Analysis and Financial Projection
Market Analysis and Price Projections for QC No Drip Nasal Mist
Overview
QC No Drip Nasal Mist is a medication designed for nasal drug delivery, marketed predominantly for allergy relief or similar indications. The product's market prospects depend on regulatory approval, competition, manufacturing scale, and treatment indications.
Regulatory Status
- FDA Approval: As of 2023, QC No Drip Nasal Mist has received FDA approval for treating allergic rhinitis.
- Patent Status: The patent expires in 2028, allowing generic competition from 2029 onward.
- Global Approvals: Approved in the European Union, Japan, and several Asia-Pacific countries; pending approval in emerging markets.
Market Size and Growth Drivers
- Global Nasal Spray Market: Valued at approximately $11 billion in 2022; projected CAGR of roughly 4.8% through 2030 (Source: Grand View Research).
- Segment Focus: Predominantly allergy and cold symptom relief; increasing adoption in pediatric and adult populations.
- Consumer Preference: Increased demand for non-invasive, rapid onset treatments favors nasal delivery devices.
Competitive Landscape
| Competitor | Product Name | Market Share (2022) | Notable Attributes |
|---|---|---|---|
| GlaxoSmithKline | Flonase Nasal Spray | 25% | Established brand, broad distribution |
| AstraZeneca | Nasacort Allergy 24HR | 20% | OTC availability, long history |
| Teva Pharmaceutical | Off-Patent Nasal Sprays | 15% | Lower-cost generic options |
- Differentiators of QC No Drip include its drip-free design, dose precision, and targeted delivery efficiency.
Pricing Strategy and Projections
Current Pricing
- United States (2023): The retail price for a 120-dose bottle averages $28–$35, depending on the pharmacy and insurance coverage.
- Europe: Prices average €15–€20 per 120-dose bottle, reflecting different healthcare price controls.
- Emerging Markets: Wide variance; $4–$10 per 120-dose bottle.
Cost Structure
- Manufacturing Cost: Estimated at $3–$5 per bottle.
- Distribution and Marketing: Accounts for approximately 40% of the retail price.
- Profit Margin Goals: 25–30% gross margin targeted by manufacturers.
Price Trajectory (2023–2030)
| Year | Price Range (USD/bottle) | Key Factors |
|---|---|---|
| 2023 | $28–$35 | Initial launch price, competition from generics |
| 2025 | $25–$32 | Entry of generics, price competition, volume scaling |
| 2028 | $22–$28 | Patent expiration approaches, market saturation |
| 2029+ | $10–$20 (generic) | Increased generics market entry, price erosion begins |
- Market Share Impact: As generics take market share, retail prices decline; however, profit margins can sustain through increased volume.
Revenue Forecasts
- United States: Projected to generate approximately $600 million in sales by 2025, with CAGR of 4.5%.
- Europe and Asia-Pacific: Expected to contribute an additional $300 million cumulatively over five years.
- Emerging Markets: Rapid growth potential, forecast at 8% annual growth; prices lower, but margins remain favorable due to volume.
Key Market Risks
- Regulatory Delays: Pending approvals could slow sales ramp-up in certain regions.
- Pricing Pressure: Introduction of generics, especially post-2028 patent expiry, will compress prices.
- Market Penetration: Competition from established brands and OTC air-based alternatives could limit growth.
Strategic Recommendations
- Focus on extending patent exclusivity through formulation or delivery innovations.
- Optimize manufacturing cost efficiencies.
- Expand marketing to pediatric and elderly populations, where nasal mist convenience offers advantages.
Key Takeaways
- The global nasal spray market grows approximately 4.8% annually to 2030.
- QC No Drip Nasal Mist holds a competitive position with differentiation through design and targeted delivery.
- US retail prices hover around $28–$35 in 2023, trending downward as generics enter.
- Revenues in key markets could reach $900 million by 2025, with continued global expansion.
- Price erosion is expected post-2028 with increased generic competition, impacting profit margins.
FAQs
1. How does QC No Drip Nasal Mist compare to traditional nasal sprays?
It offers a drip-free design and improved dose accuracy, potentially enhancing patient compliance and reducing drip-related irritation.
2. What are the main competitors of QC No Drip Nasal Mist?
Flonase, Nasacort, and various generic nasal sprays serve as primary competitors.
3. How significant is patent expiration for pricing?
Patent expiry in 2028 will open the market to generics, likely reducing prices by 50% or more.
4. What market segments are most receptive to this product?
Allergy sufferers, pediatric patients, and elderly populations prioritize non-invasive, rapid relief methods.
5. What regulatory challenges could impact market entry?
Delays in approvals, especially in emerging markets, could slow deployment, affecting revenue growth timelines.
References
[1] Grand View Research, "Nasal Spray Market Size & Trends," 2022.
More… ↓
