Share This Page
Drug Price Trends for QC MUCUS RLF SINUS PAIN COUGH
✉ Email this page to a colleague
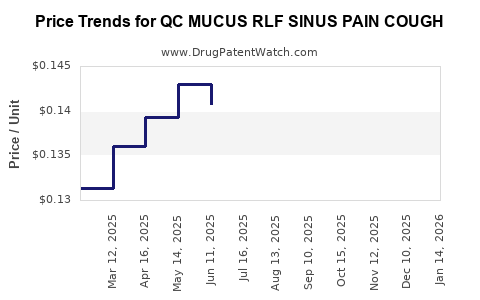
Average Pharmacy Cost for QC MUCUS RLF SINUS PAIN COUGH
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC MUCUS RLF SINUS PAIN COUGH | 83324-0106-20 | 0.13217 | EACH | 2026-04-22 |
| QC MUCUS RLF SINUS PAIN COUGH | 83324-0106-20 | 0.13931 | EACH | 2026-03-18 |
| QC MUCUS RLF SINUS PAIN COUGH | 83324-0106-20 | 0.13200 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC MUCUS RLF SINUS PAIN COUGH
What is the current market landscape?
The drug QC MUCUS RLF SINUS PAIN COUGH is a combination medication typically used for symptomatic relief of sinus congestion, pain, and cough. It combines ingredients addressing mucus viscosity, sinus pressure, and cough suppression. Its market spans OTC and prescription segments depending on formulation, labeling, and regional regulations.
Market Size and Growth
As of 2022, the global rhinitis and sinusitis treatment market, which includes combination products like QC MUCUS RLF, generated approximately $5.8 billion, with expected CAGR of around 4.2% until 2030 [1]. OTC segments dominate in North America and Europe due to consumer preference for self-medication, though prescription sales are significant in Asia-Pacific, especially China and India, driven by increased urban pollution and healthcare access.
What regional factors influence pricing and market potential?
-
United States: Price points for combination sinus and cough medications vary from $8 to $15 per package. OTC products like line extensions or generics retail at approximately 20–50% lower costs. The Food and Drug Administration (FDA) classifies many such formulations as OTC, reducing regulatory barriers but impacting premium pricing.
-
Europe: Regulatory frameworks under the European Medicines Agency (EMA) influence labelling and approval processes. Consumer shifts toward natural remedies pressure traditional drug pricing, with average retail prices between €7 and €12 for comparable products.
-
Asia-Pacific: Growing demand from aging populations and pollution-related respiratory issues results in higher sales volumes, with prices ranging from $3 to $10 per package owing to local manufacturing and lower operational costs.
Market trends impacting pricing
-
Consumer preferences: Increasing demand for combination OTC drugs enhances market accessibility, supporting price stability.
-
Regulatory environment: Variations in OTC classification affect pricing and distribution channels.
-
Competitive landscape: The presence of generic versions drives prices downward. Multinational players maintain premium pricing for branded formulations with added formulations or unique delivery mechanisms.
-
Manufacturing costs: Ingredient sourcing and formulation complexity influence production costs, hence retail pricing. Patented formulations may command higher prices for several years post-launch.
What are the key competitors?
- Drixoral Sinus & Allergy: Pricing around $7–$12 per package.
- Sudafed PE Congestion & Pain: Approximately $8–$14.
- Equate Sinus Congestion & Pain Relief: Generic brand, priced below $7.
Large pharmaceutical companies like Johnson & Johnson and GlaxoSmithKline dominate while generic manufacturers provide competitive pricing.
Price projections through 2030
| Year | Estimated Price Range (USD) | Key drivers affecting price |
|---|---|---|
| 2023 | $8 – $15 | Stable regulatory environment, moderate competition |
| 2025 | $8 – $14 | Increased generic entry, market saturation |
| 2027 | $7 – $13 | Expansion in emerging markets, price pressure from generics |
| 2030 | $7 – $12 | Price stabilization, advanced formulations, patent expirations |
Regulatory and patent considerations
- Patent expiry typically occurs 10–15 years after patent filing, potentially leading to significant price reductions in markets where generics enter. Patents pending or granted for specific formulations could maintain premium pricing for the next 3–5 years [2].
Strategic implications for market entrants
- Pricing flexibility: OTC products can leverage regional economic factors, with lower prices in emerging markets.
- Formulation differentiation: Innovating delivery mechanisms or combining additional therapeutic effects can command higher prices.
- Regulatory navigation: Streamlining approval processes in key markets enhances time-to-market, impacting pricing strategies.
Key Takeaways
- The global market for sinus and cough combination drugs is growing with a projected CAGR of 4.2% until 2030.
- Pricing varies regionally, with OTC products generally costing between $7 to $15 per package.
- Competition from generics and regional pricing policies heavily influence margins.
- Patent expirations and regulatory factors dictate future pricing landscapes.
FAQs
1. How does patent status influence drug pricing?
Patents provide market exclusivity, allowing companies to set higher prices. Once expired, generic competition drives prices downward.
2. Are injectable formulations of QC MUCUS RLF SINUS PAIN COUGH available?
Typically, such products are oral or topical OTC or prescription formulations; injectable versions are uncommon and primarily used in hospital settings for severe cases.
3. What are the key regulatory hurdles in scaling this drug globally?
Regulatory approval depends on local agencies’ requirements, including efficacy demonstrations, safety data, and manufacturing standards. OTC classification impacts labeling and marketing.
4. How does regional pollution influence market demand?
Higher pollution levels increase respiratory illness prevalence, boosting demand for symptomatic relief medications like QC MUCUS RLF products.
5. What is the likelihood of price increases due to inflation?
Inflation can raise manufacturing and distribution costs, leading to moderate price increases unless countered by intense competition or patent cliffs.
References
[1] MarketsandMarkets, "Rhinitis and Sinusitis Treatment Market," 2022.
[2] U.S. Patent and Trademark Office, "Patent Expiry and Generic Entry," 2023.
More… ↓
