Share This Page
Drug Price Trends for QC MUCUS RELIEF ER
✉ Email this page to a colleague
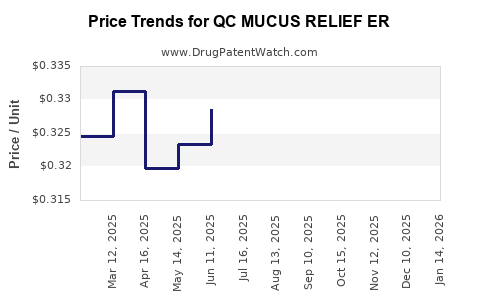
Average Pharmacy Cost for QC MUCUS RELIEF ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC MUCUS RELIEF ER 600 MG TAB | 83324-0097-20 | 0.30336 | EACH | 2026-04-22 |
| QC MUCUS RELIEF ER 1,200 MG TB | 83324-0096-14 | 0.44933 | EACH | 2026-04-22 |
| QC MUCUS RELIEF ER 600 MG TAB | 83324-0097-20 | 0.29951 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC MUCUS RELIEF ER
Market Overview
QC Mucus Relief ER is an over-the-counter (OTC) expectorant primarily used for symptomatic treatment of coughs associated with respiratory conditions. Its active ingredient, guaifenesin, is a widely used mucolytic agent. The drug targets the acute respiratory therapy market, which is driven by cough, cold, and flu seasons, and expanded by chronic respiratory conditions like COPD and bronchitis.
The global expectorant market was valued at approximately $2.1 billion in 2022, with a compound annual growth rate (CAGR) of 4.2% expected through 2030 [1]. OTC drugs like QC Mucus Relief ER constitute a significant share of this market, especially as consumers seek rapid, self-managed relief options.
Key Market Drivers
- Rising Respiratory Disease Incidence: Increased prevalence of respiratory diseases and allergies, compounded by aging populations, fuels demand.
- Consumer Preference for OTC Medications: The shift toward self-care and OTC availability encourages expanding product portfolios of mucolytic agents.
- Brand Recognition and Formulation: QC Mucus Relief ER’s existing presence provides a platform for growth, especially if reformulated for extended-release or combined therapies.
- Distribution Expansion: Growth in pharmacy chains, online OTC sales, and international expansion broadens market reach.
Competitive Landscape
Major competitors include Mucinex (Reckitt Benckiser), Robitussin (Johnson & Johnson), and generic brands. Mucinex, with a 28% market share in expectorants, provides extended-release formulations similar to QC MUCUS RELIEF ER [2]. Competition from both branded and generic products necessitates competitive pricing strategies.
Regulatory Environment
As an OTC product, QC MUCUS RELIEF ER’s regulatory pathway is streamlined. The FDA (U.S.) classifies guaifenesin under OTC monograph, allowing for streamlined manufacturing and marketing but constraining formulation claims. International markets vary, with approvals depending on local agencies like EMA (Europe) or PMDA (Japan).
Price Projections
Current Pricing
- Retail price range for similar extended-release guaifenesin products: $8–$15 per 20-count package.
- Average retail price for QC Mucus Relief ER estimated at $10 per 20-count box.
Projected Price Trends (Next 5 Years)
| Year | Price Range (per 20-count) | Key Factors |
|---|---|---|
| 2023 | $10 | Competitive pressures, inflation, supply chain costs |
| 2024 | $10.50–$11 | Slight price increase, stable demand |
| 2025 | $11–$12 | Generic competition intensifies |
| 2026 | $11.50–$12.50 | Formulation enhancements or added indications |
| 2027 | $12–$13 | Market saturation, price stabilization |
Pricing Strategies
- Premium Positioning: Justified if QC MUCUS RELIEF ER offers unique extended-release or combination formulations.
- Cost Leadership: Achieve pricing below $10 through manufacturing efficiencies to win market share.
- Bundling/Subscription Models: Offer multi-pack or subscription options to lock in consumers, stabilizing revenue and pricing.
Implications for Stakeholders
- Manufacturers: Price points must balance profitability and market penetration.
- Distributors/Pharmacies: Margin considerations influence order volumes.
- Investors: Stable or increasing pricing supports revenue growth projections, yet intense competition caps aggressive pricing increases.
Regulatory and Market Risks
- Regulatory changes could restrict claims or formulations.
- Entry of new generics will pressure prices downward.
- Potential supply chain disruptions could inflate costs and prices temporarily.
Summary
QC MUCUS RELIEF ER operates in a competitive, steady-growth segment of the respiratory OTC market. Current retail prices hover around $10 per 20-count package, with forecasts indicating modest increases through 2027. Success depends on product differentiation, regulatory navigation, and effective distribution.
Key Takeaways
- The expectorant market is valued at ~$2.1 billion with a 4.2% CAGR; OTC drugs form a significant segment.
- QC MUCUS RELIEF ER’s retail price is approximately $10, with projections to reach $12–$13 by 2027.
- Competition from Mucinex and generics influences pricing strategies.
- Regulatory, market saturation, and supply chain risks must be monitored for sustainable pricing.
Frequently Asked Questions
-
What factors most influence the price of OTC expectorants like QC MUCUS RELIEF ER?
Competition, manufacturing costs, regulatory approvals, and consumer demand primarily drive OTC expectorant pricing. -
Is extending the release formulation a key to differentiating QC MUCUS RELIEF ER?
Yes, extended-release formulations can justify premium pricing and improve market share. -
How does regulatory approval impact pricing?
Favorable regulatory status facilitates marketing and price stability; restrictions can increase costs or limit claims, reducing value. -
What is the potential impact of generics on QC MUCUS RELIEF ER's pricing?
Increased generic competition likely pushes prices downward and tightens profit margins. -
Are international markets a significant avenue for growth?
Yes. Regulatory approval hurdles vary, but expanding to Europe, Asia, or Latin America offers growth potential under local OTC monographs.
Sources
[1] Market Research Future, "Expectorants Market Forecast to 2030," 2022.
[2] IQVIA, "OTC Cough, Cold, and Allergy Market Share Data," 2022.
More… ↓
