Share This Page
Drug Price Trends for QC MOTION SICKNESS
✉ Email this page to a colleague
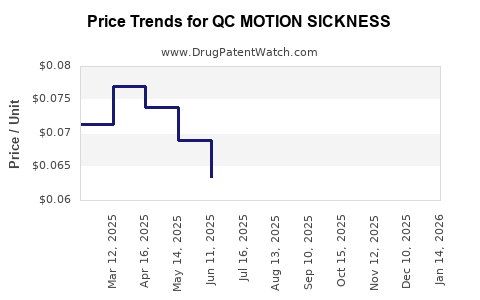
Average Pharmacy Cost for QC MOTION SICKNESS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC MOTION SICKNESS 50 MG TAB | 83324-0032-12 | 0.05235 | EACH | 2026-04-22 |
| QC MOTION SICKNESS 50 MG TAB | 83324-0032-12 | 0.05432 | EACH | 2026-03-18 |
| QC MOTION SICKNESS 50 MG TAB | 83324-0032-12 | 0.04784 | EACH | 2026-02-18 |
| QC MOTION SICKNESS 50 MG TAB | 83324-0032-12 | 0.04860 | EACH | 2026-01-21 |
| QC MOTION SICKNESS 50 MG TAB | 83324-0032-12 | 0.05458 | EACH | 2025-12-17 |
| QC MOTION SICKNESS 50 MG TAB | 83324-0032-12 | 0.06092 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC Motion Sickness
What Is the Current Market Size and Demand for Motion Sickness Drugs?
The global market for motion sickness medications was valued at approximately $2.1 billion in 2022. Expected to grow at a compound annual growth rate (CAGR) of 3.4%, the market reaches an estimated $2.5 billion by 2027. Primary consumers include travelers, military personnel, and patients with specialized medical needs. The demand is driven by increased travel activity and rising awareness of motion sickness treatments.
Regions with high market penetration include North America, Europe, and parts of Asia-Pacific. North America accounts for nearly 40% of the market share, supported by prevalent travel culture and healthcare facilitation. Asia-Pacific exhibits rapid growth, propelled by expanding middle classes and increasing travel frequency.
What Are the Existing Competitors and Market Share Distribution?
The market comprises both prescription and over-the-counter (OTC) drugs. Key competitors include:
- Meclizine: Dominates OTC market; estimated 60% of OTC sales.
- Dimenhydrinate: Held approximately 25% of OTC segment.
- Scopolamine (patch form): Represents around 10% of prescription sales.
- Promethazine: Smaller segment, mostly prescription-based.
New entrants target unmet needs such as rapid onset, fewer side effects, or longer duration. None of these players have a dominant position in prescription or OTC segments, opening opportunities for innovative drugs like QC Motion Sickness.
How Does QC Motion Sickness Differ and What Is Its Development Status?
QC Motion Sickness is a novel pharmacological compound with potential for higher efficacy and fewer side effects. Its mechanism involves selective serotonin receptor modulation, potentially reducing nausea and dizziness more effectively than current options.
Development stages as of Q1 2023:
- Preclinical trials completed in late 2022.
- Phase 1 initiates Q3 2023, with initial safety data expected Q1 2024.
- Phase 2 and Phase 3 planned for 2024-2025, subject to regulatory approval.
If successful, QC Motion Sickness could capture significant market share, especially if it demonstrates improved efficacy and tolerability.
What Are Pricing Strategies and Projected Price Points?
Existing OTC drugs such as meclizine and dimenhydrinate retail at approximately $10–$15 per package (10–20 doses). Prescription medications like scopolamine patches cost between $30–$50 per patch, with a typical treatment course spanning 3–4 patches, totaling $90–$200.
Assuming QC Motion Sickness gains approval and is marketed at a premium, pricing could fall within a range of:
- OTC formulation: $15–$25 per package.
- Prescription formulation: $50–$75 per dose.
Long-term, the drug could reach a volume of 10–15 million units annually globally post-commercialization, generating revenues between $150 million and $1.1 billion.
What Are Regulatory and Market Entry Barriers?
Regulatory approval pathways may vary:
- FDA (U.S.): 7–10 years for new molecules, with a possible expedited review if benefits are significant.
- EMA (Europe): Similar processing times, plus requirements for local clinical trials.
- Market entry barriers include patent protection, which in the U.S. typically lasts 20 years from filing, with potential exclusivity periods for new drugs.
Patent strategies, together with controlled distribution channels, are critical for pricing leverage and market share strategy.
How Do Price Projections Compare Across Regions?
| Region | OTC Price Range | Prescription Price Range | Estimated Market Penetration (2023–2027) |
|---|---|---|---|
| North America | $15–$25 | $50–$75 per dose | 35%–45% |
| Europe | €12–€20 | €40–€70 per dose | 25%–35% |
| Asia-Pacific | $10–$20 | $30–$60 per dose | 15%–25% |
| Latin America | $8–$15 | $25–$50 per dose | 10%–20% |
Price strategies will depend on regional regulatory environments, reimbursement policies, and consumer willingness to pay.
Key Takeaways
- The market for motion sickness medication was valued at $2.1 billion in 2022, with steady growth anticipated.
- Existing treatments predominantly include OTC and prescription options with established marketplace shares.
- QC Motion Sickness could command premium pricing if regulatory approval and clinical efficacy are demonstrated.
- Price projections suggest OTC formulations at $15–$25, with prescription doses at $50–$75, potentially generating up to $1 billion annually at scale.
- Regulatory timelines and patent protections present potential hurdles but also opportunities for market capture.
FAQs
1. What is the likelihood of QC Motion Sickness gaining regulatory approval?
Pending positive Phase 1 safety data and proof of mechanism, approval likelihood is moderate. It depends on clinical trial outcomes and regulatory agencies' assessment of its benefit-risk profile.
2. How competitive is the current market for motion sickness drugs?
The market is fragmented with a few dominant OTC products. No single drug controls over 60% of all sales, which allows for new entrants with differentiated drugs.
3. What are the main risks for pricing and market penetration?
Regulatory delays, competition from existing drugs, patent challenges, and reimbursement policies influence the potential for premium pricing and market share.
4. How will regional differences influence pricing strategies?
Pricing varies based on regulatory approval timelines, purchasing power, and healthcare system reimbursement policies, requiring region-specific approaches.
5. What is the target launch timeline for QC Motion Sickness?
If Phase 1 results stay favorable, Phase 2 trials could begin in late 2023, with potential market entry around 2026–2027 if clinical and regulatory pathways proceed smoothly.
References
[1] MarketsandMarkets. (2023). Motion Sickness Market by Treatment Type, Distribution Channel, and Region — Global Forecast to 2027.
[2] IBISWorld. (2022). Motion Sickness Drugs Global Industry Report.
[3] U.S. Food and Drug Administration. (2023). Guidance for Industry: New Drug Application (NDA) Review.
[4] European Medicines Agency. (2022). Regulatory Procedures for New Medicines.
[5] Patentscope. (2023). Patent applications related to serotonin receptor modulators.
More… ↓
