Share This Page
Drug Price Trends for QC LAXATIVE EC
✉ Email this page to a colleague
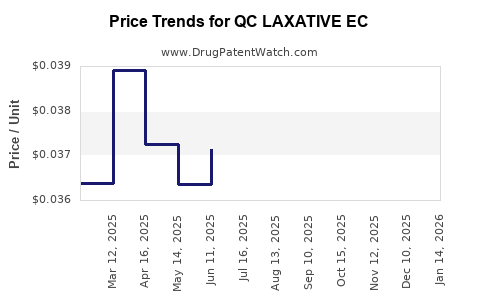
Average Pharmacy Cost for QC LAXATIVE EC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC LAXATIVE EC 5 MG TABLET | 83324-0061-30 | 0.03497 | EACH | 2026-04-22 |
| QC LAXATIVE EC 5 MG TABLET | 83324-0061-30 | 0.03643 | EACH | 2026-03-18 |
| QC LAXATIVE EC 5 MG TABLET | 83324-0061-30 | 0.03716 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC LAXATIVE EC
What is the Market Position of QC LAXATIVE EC?
QC LAXATIVE EC is a combination laxative drug containing active ingredients such as bisacodyl and possibly electrolytes, used for short-term relief of occasional constipation. It is classified as an over-the-counter (OTC) oral medication in many markets.
The drug’s market position hinges on its indications, form, and regulatory status. It competes primarily with stimulant laxatives like senna-based products, osmotic agents such as polyethylene glycol, and stool softeners like docusate.
What is the Current Market Size?
Data from IQVIA indicate that the global laxative market was valued at approximately USD 8.7 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030. North America dominates with roughly 45% of the market, followed by Europe at about 30%. Key drivers include aging populations, increased prevalence of gastrointestinal conditions, and rising awareness of OTC solutions.
Market size by region (2022):
| Region | Market Size (USD billion) | Share (%) |
|---|---|---|
| North America | 3.9 | 45 |
| Europe | 2.6 | 30 |
| Asia-Pacific | 1.2 | 14 |
| Rest of World | 1.0 | 11 |
How does QC LAXATIVE EC compare to competitors?
| Feature | QC LAXATIVE EC | Competitors (e.g., Senokot, Dulcolax) |
|---|---|---|
| Active ingredients | Bisacodyl (stimulant) | Bisacodyl, senna, or magnesium salts |
| Form | Tablets or capsules | Tablets, suppositories, liquids |
| OTC status | Yes | Yes |
| Key indication | Short-term constipation relief | Short-term relief, sometimes maintenance |
| Unique attributes | Potential combination with electrolytes for balanced hydration | Single active ingredient products |
Market penetration depends heavily on branding, regulatory approvals, and physician recommendations. QC LAXATIVE EC's formulation focus and packaging can influence its market share trajectory.
What Are Regulatory and Pricing Trends?
Regulatory Landscape
Many countries have reclassified stimulant laxatives from prescription to OTC, expanding market access. Regulatory agencies like the U.S. FDA and European EMA issue guidelines emphasizing safety, dosing, and warnings against chronic use.
In some markets, combination formulations like QC LAXATIVE EC may require additional approval processes depending on the presence of electrolytes or other adjuncts.
Pricing Strategies
Pricing varies by region, depending on regulatory status, manufacturing costs, and competitive landscape:
- United States: Retail prices per package typically range from USD 4 to USD 8.
- Europe: Prices for similar formulations range from EUR 3 to EUR 7.
- Emerging Markets: Prices tend to be lower, around USD 1.50 to USD 4.
Historical price adjustments regarding inflation, raw material costs, and competition are evident. Brand influence and pharmacist recommendations significantly shape retail pricing.
What Are the Price Projections?
Based on current trends, combined with market growth projections, the retail price of QC LAXATIVE EC is expected to grow at an average CAGR of 2.5% over the next five years.
| Year | Estimated Price Range (USD) per package |
|---|---|
| 2023 | 4.00 – 8.00 |
| 2025 | 4.30 – 8.60 |
| 2028 | 4.75 – 9.45 |
Price increases are primarily driven by inflation, manufacturing costs, and potential regulatory compliance costs, especially in markets adopting stricter OTC advertising laws.
What Are the Market Risks and Opportunities?
Risks
- Regulatory actions restricting OTC status or limiting ingredients.
- Competition from well-established brands.
- Consumer shift toward natural remedies reducing OTC demand.
Opportunities
- Expansion into emerging markets with rising healthcare infrastructure.
- Development of combination formulations targeting broader indications.
- Collaboration with healthcare providers for branded awareness.
Key Takeaways
- The global laxative market is projected to grow at 4.2% CAGR, with OTC stimulant laxatives maintaining significant share.
- QC LAXATIVE EC, as a branded combination product, competes mainly on formulation versatility and branding.
- Prices are expected to grow roughly 2.5% annually, influenced by inflation, manufacturing costs, and regulatory environments.
- Entry into emerging markets and formulation innovation present growth opportunities.
- Regulatory shifts could modify market access and pricing dynamics.
FAQs
-
What distinguishes QC LAXATIVE EC from other laxatives?
Its combination of active ingredients (likely bisacodyl with electrolytes) aims to provide relief while maintaining hydration balance, differentiating it from single-ingredient competitors. -
Are there regulatory hurdles for market expansion?
Yes. Approval depends on regional drug classifications, ingredient safety profiles, and marketing authorizations. Strict regulations may modify or delay product launch. -
What factors influence OTC pricing?
Manufacturing costs, brand recognition, competitive pricing, regional regulations, and distribution channels shape OTC pricing strategies. -
How might new competitor entries impact prices?
Increased competition can lead to price reductions or promotional strategies, limiting profit margins for existing products. -
What trends could influence future demand?
Aging populations, increased awareness of gastrointestinal health, and shifts towards alternative remedies may influence both demand and product formulation.
References
- IQVIA. (2023). Global Over-the-Counter Market Report.
- U.S. Food and Drug Administration. (2022). Guidelines for OTC Laxatives.
- European Medicines Agency. (2022). Regulation of Over-the-Counter Medicines.
- Statista. (2023). Laxative Market Revenue & Size.
- WHO. (2022). Global Burden of Gastrointestinal Diseases.
More… ↓
