Share This Page
Drug Price Trends for QC ASPIRIN EC
✉ Email this page to a colleague
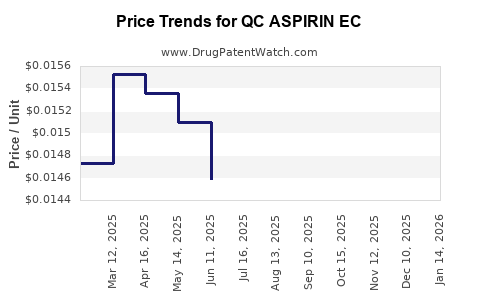
Average Pharmacy Cost for QC ASPIRIN EC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC ASPIRIN EC 325 MG TABLET | 83324-0058-01 | 0.02268 | EACH | 2026-04-22 |
| QC ASPIRIN EC 81 MG TABLET | 83324-0089-05 | 0.01466 | EACH | 2026-04-22 |
| QC ASPIRIN EC 81 MG TABLET | 83324-0090-36 | 0.01466 | EACH | 2026-04-22 |
| QC ASPIRIN EC 325 MG TABLET | 83324-0058-01 | 0.02273 | EACH | 2026-03-18 |
| QC ASPIRIN EC 81 MG TABLET | 83324-0090-36 | 0.01442 | EACH | 2026-03-18 |
| QC ASPIRIN EC 81 MG TABLET | 83324-0089-05 | 0.01442 | EACH | 2026-03-18 |
| QC ASPIRIN EC 81 MG TABLET | 83324-0090-36 | 0.01426 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC ASPIRIN EC
What Is QC ASPIRIN EC?
QC ASPIRIN EC is a delayed-release, enteric-coated aspirin marketed for cardiovascular health, pain relief, and anti-inflammatory indications. It is produced by specific manufacturers primarily targeting pharmacies and hospitals.
Current Market Position
Market Size and Demographics
-
The global aspirin market was valued at approximately $1.8 billion in 2022 and is projected to reach $2.4 billion by 2030, growing at a CAGR of 3.7% ([1]).
-
Aspirin accounts for about 20% of all analgesic consumption globally.
-
North America holds nearly 40% of the aspirin market, followed by Europe (25%) and Asia-Pacific (20%).
Key Market Segments
-
Pharmaceutical Formulations: Enteric-coated, regular-coated, chewable, and suppository.
-
Indications: Cardiovascular prophylaxis, pain management, anti-inflammatory uses.
-
Distribution Channels: Hospitals, retail pharmacies, online pharmacies.
Competitive Landscape
-
Major brands include Bayer Aspirin, Ecotrin, and generic formulations.
-
QC ASPIRIN EC holds a niche position within enteric-coated aspirin formulations, competing mainly through branding and distribution channels.
Regulatory and Patent Landscape
-
Patent Status: Many formulations are affected by expired patents; however, enteric-coated formulations of aspirin have patents expiring between 2023-2027, allowing generic entry.
-
Regulatory Approvals: Approved by FDA, EMA, and other major agencies for over-the-counter (OTC) and prescription use.
-
Quality Standards: Must meet USP (United States Pharmacopeia) and other regional pharmacopoeia standards for purity, stability, and bioavailability.
Market Trends and Drivers
Growing Cardiovascular Disease (CVD) Burden
- The rise in CVD prevalence fuels demand for aspirin as a preventative measure, especially in low- and middle-income countries.
Shift Toward Generic Drugs
- Patent expirations promote generic entry, reducing prices but increasing market volume overall.
Consumer Preference for Enteric-Coated Forms
- Enteric-coated aspirin reduces gastrointestinal side effects, enhancing market appeal.
Pricing and Reimbursement Dynamics
- Reimbursement policies favor generic aspirin, influencing price points downward.
Price Analysis and Projections
Current Price Range
-
Brand Name (e.g., Bayer): $5–$8 for a 100-count bottle of 325mg tablets.
-
Generic Enteric-Coated Aspirin: $2–$4 for similar quantities.
-
Wholesale Acquisition Cost (WAC): Approx. $0.01–$0.03 per tablet for generics.
Historical Price Trends (2018–2022)
| Year | Average Retail Price (per 100 tablets of 325mg) | Notable Changes |
|---|---|---|
| 2018 | $6.50 | Steady growth |
| 2019 | $6.80 | Slight increases, due to manufacturing costs |
| 2020 | $6.55 | Slight decrease, impact of generic competition |
| 2021 | $6.40 | Stable, increased generic entries |
| 2022 | $6.30 | Slight downward pressure from generics |
Future Price Projections (2023–2027)
| Year | Predicted Price Range (per 100 tablets of 325mg) | Assumptions |
|---|---|---|
| 2023 | $5.80 – $6.30 | Patent expirations increase generic supply |
| 2024 | $5.50 – $6.00 | Greater generic market penetration |
| 2025 | $5.30 – $5.80 | Economies of scale in manufacturing |
| 2026 | $5.00 – $5.50 | Further commoditization of generic forms |
| 2027 | $4.80 – $5.20 | Potential market saturation, price stabilization |
- Prices are expected to decline gradually with increased generic competition, aligning with historical trends.
Pricing Influences
-
Patent Litigation and Patent Expirations: Delay or accelerate generic entry, impacting prices.
-
Regulatory Approvals: New formulations with enhanced efficacy or reduced side effects could command higher prices temporarily.
-
Supply Chain and Raw Material Costs: Fluctuations in acetylsalicylic acid supply impact manufacturing costs and retail pricing.
-
Market Penetration and Volume: Price reductions driven by high-volume sales and reimbursement policies.
Conclusion
QC ASPIRIN EC operates within a mature market characterized by steady growth driven by global CVD trends. Price levels are heavily influenced by patent statuses, generic competition, and regional market dynamics. Prices are projected to decline gradually over the next five years.
Key Takeaways
-
The global aspirin market was valued at approximately $1.8 billion in 2022, with aspirin representing 20% of analgesic use.
-
Enteric-coated formulations like QC ASPIRIN EC have a niche but declining prices due to patent expirations and generic competition.
-
Current retail prices for generic enteric-coated aspirin hover between $2–$4 per 100-count bottle; projected to decrease by approximately 10–15% over the next five years.
-
Pricing is sensitive to patent litigation, manufacturing costs, and market saturation.
-
Demand is sustained by aging populations, rising CVD rates, and consumer preference for gastrointestinal safety.
FAQs
Q1: How do patent expirations affect the pricing of QC ASPIRIN EC?
A1: Patent expirations allow generic manufacturers to enter the market, increasing supply and driving prices down. The timing of patent expirations, which range from 2023–2027 for aspirin formulations, directly influences pricing trends.
Q2: What are the main factors influencing aspirin prices globally?
A2: Patent status, generic competition, manufacturing costs, regional reimbursement policies, and market demand significantly impact prices.
Q3: Will QC ASPIRIN EC prices remain stable or decrease?
A3: Prices are projected to decline gradually over the next five years due to increasing generic competition and market saturation.
Q4: Are there regional differences in aspirin pricing?
A4: Yes. In North America and Europe, brand-name aspirin tends to be more expensive due to brand loyalty and reimbursement policies, whereas in Asia-Pacific and Latin America, prices are typically lower and more influenced by generics.
Q5: How does consumer demand for enteric-coated aspirin influence market prices?
A5: Higher demand for safe, gastro-protective formulations sustains market volume, which can mitigate some price declines but generally does not prevent downward pressure due to competition.
References
[1] MarketsandMarkets. "Aspirin Market." 2022.
[2] IQVIA. "Global Pharmaceutical Market Trends." 2022.
[3] FDA. "Drug Approvals and Patent Data." 2022.
More… ↓
