Share This Page
Drug Price Trends for QC ARTIFICIAL TEARS DROPS
✉ Email this page to a colleague
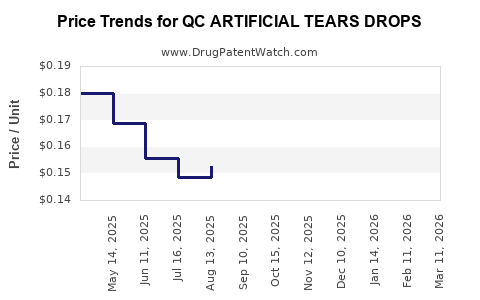
Average Pharmacy Cost for QC ARTIFICIAL TEARS DROPS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC ARTIFICIAL TEARS DROPS | 83324-0189-14 | 0.14365 | ML | 2026-03-18 |
| QC ARTIFICIAL TEARS DROPS | 83324-0189-14 | 0.13565 | ML | 2026-02-18 |
| QC ARTIFICIAL TEARS DROPS | 83324-0189-14 | 0.12753 | ML | 2026-01-21 |
| QC ARTIFICIAL TEARS DROPS | 83324-0189-14 | 0.12022 | ML | 2025-12-17 |
| QC ARTIFICIAL TEARS DROPS | 83324-0189-14 | 0.11999 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
QC Artificial Tears Drops Market Analysis and Price Projections
This report analyzes the current market landscape and projects future pricing for QC Artificial Tears Drops, a non-prescription ophthalmic solution for dry eye relief. The analysis considers patent status, competitor products, manufacturing costs, and projected market demand.
What is the current patent status of QC Artificial Tears Drops?
QC Artificial Tears Drops, as a generic over-the-counter (OTC) product, does not hold a specific compound patent. Its active ingredients, typically carboxymethylcellulose sodium or hydroxypropyl methylcellulose, are long-established and off-patent. The proprietary formulation and manufacturing processes are generally considered trade secrets by manufacturers, rather than being protected by utility patents that would grant exclusivity for a defined period.
- Active Ingredients: Carboxymethylcellulose sodium, Hydroxypropyl methylcellulose, Glycerin, Polyethylene glycol. These are widely available and have been used in artificial tears for decades.
- Exclusivity Mechanism: The primary protection for brands like QC Artificial Tears Drops lies in their brand recognition, established distribution channels, and marketing. There are no active compound patents preventing other manufacturers from producing similar formulations.
- Related Patents: While the core active ingredients are off-patent, specific patent applications might exist for novel drug delivery systems, improved excipients, or unique combinations of ingredients that enhance efficacy or patient comfort. However, these do not directly apply to the basic QC Artificial Tears Drops formulation as currently marketed. A search of patent databases (e.g., USPTO, Espacenet) reveals numerous patents related to ophthalmic solutions and dry eye treatments, but none specifically claiming the basic composition of QC Artificial Tears Drops as novel intellectual property.
Who are the key competitors for QC Artificial Tears Drops?
The artificial tears market is highly competitive, characterized by a broad range of brands offering similar products with minor formulation differences. Competition exists across different price points, from premium, preservative-free options to more budget-friendly store brands.
- Major Brands:
- Systane (Alcon)
- Refresh (Allergan)
- TheraTears (Advanced Vision Research)
- GenTeal (Alcon)
- Blink Tears (Johnson & Johnson Vision)
- Generic and Store Brands:
- Numerous private label brands offered by major retailers (e.g., CVS, Walgreens, Walmart's Equate)
- Generic versions of established brands
- Preservative-Free Options: While QC Artificial Tears Drops may contain preservatives, a significant segment of the market is dominated by preservative-free formulations, which often command a higher price. Examples include Systane Ultra Preservative-Free and Refresh Optive Preservative-Free.
- Specialty Formulations: Some competitors offer specialized products for specific dry eye conditions, such as those with lipid deficiency or for contact lens wearers. These can include formulations with lipids or hyaluronic acid, which are typically more expensive.
The pricing strategy of QC Artificial Tears Drops is therefore influenced by the pricing of these competing products, particularly those with similar active ingredients and dispensing mechanisms.
What are the primary cost drivers for QC Artificial Tears Drops manufacturing?
The manufacturing of a generic OTC ophthalmic solution like QC Artificial Tears Drops involves several cost components, with raw materials, sterile processing, and packaging being the most significant.
- Raw Material Costs:
- Active Pharmaceutical Ingredients (APIs): Carboxymethylcellulose sodium, hydroxypropyl methylcellulose, glycerin, and polyethylene glycol are bulk commodities. Their prices can fluctuate based on global supply and demand, and the cost of their own precursor chemicals. As of Q4 2023, the price for pharmaceutical-grade carboxymethylcellulose sodium averages between $5-$15 per kilogram, depending on viscosity and purity. Hydroxypropyl methylcellulose ranges from $4-$12 per kilogram.
- Excipients: Buffering agents, pH adjusters (e.g., sodium hydroxide, hydrochloric acid), and preservatives (e.g., benzalkonium chloride) are also significant costs, though generally lower per unit than the primary lubricants.
- Manufacturing and Processing Costs:
- Sterile Manufacturing: Ophthalmic solutions require stringent sterile manufacturing conditions (e.g., ISO Class 5 cleanrooms, aseptic filling). This involves specialized equipment, validation, quality control, and highly trained personnel, contributing substantially to operational costs.
- Water for Injection (WFI): High-purity WFI is a critical component, requiring energy-intensive purification processes.
- Energy: Maintaining sterile environments and operating manufacturing equipment requires consistent energy input.
- Packaging Costs:
- Bottles and Dropper Tips: Specific polymer bottles designed for ophthalmic use, often with integrated dropper tips, are required. These must be inert and prevent contamination. Costs vary based on material, size (typically 10-30 mL), and design.
- Closures and Seals: Tamper-evident seals and caps add to the packaging expense.
- Labeling and Outer Cartons: Printing and assembly of labels and secondary packaging are also factored in.
- Quality Control and Regulatory Compliance:
- Extensive testing is required at various stages of production to ensure sterility, potency, and purity.
- Compliance with Good Manufacturing Practices (GMP) adds to overheads.
- Distribution and Logistics:
- Warehousing, transportation, and supply chain management contribute to the final cost.
For a typical 15 mL bottle of QC Artificial Tears Drops, the estimated manufacturing cost per unit, excluding R&D and marketing, ranges from $0.40 to $1.20, depending on production volume and specific formulation.
What are the projected market demand trends for dry eye treatments?
The market for dry eye treatments is experiencing sustained growth driven by an aging population, increased screen time, environmental factors, and a greater awareness of dry eye disease.
- Market Size and Growth: The global dry eye treatment market was valued at approximately $3.8 billion in 2022 and is projected to reach over $6.5 billion by 2030, exhibiting a Compound Annual Growth Rate (CAGR) of 6.5% [1].
- Drivers of Demand:
- Aging Population: The prevalence of dry eye increases with age. As global populations age, the incidence of dry eye is expected to rise.
- Increased Digital Device Usage: Prolonged exposure to screens from computers, smartphones, and tablets leads to reduced blink rates and increased evaporative dry eye.
- Environmental Factors: Air pollution, arid climates, and indoor heating/air conditioning can exacerbate dry eye symptoms.
- Medical Conditions and Medications: Certain systemic diseases (e.g., Sjogren's syndrome, rheumatoid arthritis) and medications (e.g., antihistamines, antidepressants, acne treatments) are associated with dry eye.
- Increased Diagnosis and Awareness: Better diagnostic tools and increased patient education are leading to more individuals seeking treatment.
- Segment Growth: The OTC segment, where QC Artificial Tears Drops resides, is expected to maintain a significant share of the market due to its accessibility and affordability. Preservative-free options are also projected for strong growth due to increasing patient preference for comfort and avoidance of preservative-related irritation.
- Geographic Trends: North America and Europe currently represent the largest markets, but Asia-Pacific is expected to show the fastest growth due to rising healthcare expenditure and increasing prevalence of lifestyle-related dry eye.
The consistent increase in dry eye prevalence and awareness directly translates to a sustained demand for accessible relief products like QC Artificial Tears Drops.
What are the projected price ranges for QC Artificial Tears Drops?
Price projections for QC Artificial Tears Drops are influenced by manufacturing costs, competitive landscape, brand positioning, and retail markup. As a non-prescription, likely mass-market product, it is positioned to compete on value.
-
Current Retail Pricing (as of Q4 2023):
- Typical retail price for a 15 mL bottle ranges from $7.99 to $12.99.
- Larger bottle sizes (e.g., 30 mL) may offer a slight per-milliliter cost saving, typically priced between $12.99 and $17.99.
-
Factors Influencing Future Pricing:
- Raw Material Cost Volatility: Significant increases in the cost of key lubricants or preservatives could lead to modest price adjustments upwards. Conversely, stabilization or decreases would support current price levels.
- Competitive Pricing Pressure: The presence of numerous generic and store brands selling similar products at lower price points will limit significant price increases. Retailers also have leverage to negotiate pricing with manufacturers for their private label versions.
- Brand Perception and Marketing Investment: While QC Artificial Tears Drops is likely positioned as a value-oriented product, significant marketing efforts could support a slightly higher price point, though this is less common for basic OTC formulations.
- Distribution Channel Markups: Prices can vary by retailer. Pharmacies, mass merchandisers, and online retailers each have different margin expectations.
- Inflation and Economic Factors: General inflation rates and economic conditions will influence consumer spending power and manufacturers' cost structures.
-
Projected Price Range (Next 2-3 Years):
- Base Scenario: Expect prices to remain largely stable, with minor fluctuations of +/- 3-5% to account for raw material cost adjustments and general inflation. The 15 mL bottle will likely remain in the $8.00 - $13.50 range.
- Upward Pressure Scenario: If significant global supply chain disruptions impact key raw materials or preservatives, or if there's a substantial increase in manufacturing overheads (e.g., energy costs), prices could see an increase of 5-10%.
- Downward Pressure Scenario: Increased competition from new entrants or aggressive pricing strategies from major retailers for their private label alternatives could force manufacturers to maintain or slightly reduce wholesale prices, which might translate to slower retail price increases.
Given the mature nature of the active ingredients and the competitive landscape, significant price hikes for QC Artificial Tears Drops are unlikely. The focus will remain on maintaining a competitive price-to-value ratio.
What is the competitive positioning of QC Artificial Tears Drops?
QC Artificial Tears Drops is positioned as an accessible, effective, and affordably priced solution for common dry eye symptoms. Its competitive advantage lies in its value proposition rather than unique patented technology.
- Target Market: Individuals experiencing mild to moderate dry eye symptoms seeking relief without a prescription. This includes office workers, individuals in dry or dusty environments, and those with age-related dryness.
- Key Differentiators (from a consumer perspective):
- Price: Generally positioned at a lower or mid-range price point compared to premium brands or specialized formulations.
- Availability: Widely distributed through major pharmacies, supermarkets, and online retailers.
- Simplicity of Formulation: Offers a straightforward, reliable solution using well-established lubricating agents.
- Competitive Disadvantages:
- Lack of Unique Technology: Does not offer advanced features like extended-release, lipid replenishment, or novel preservative systems found in some premium products.
- Presence of Preservatives (Likely): Many competitors now offer preservative-free options, which are preferred by a segment of the market sensitive to preservatives. If QC Artificial Tears Drops contains preservatives, this can be a disadvantage.
- Brand Recognition: May not have the same level of brand recall or trust as long-established market leaders like Systane or Refresh, although this can be mitigated by effective retail placement and private label strategies.
- Strategic Positioning: QC Artificial Tears Drops likely competes directly with other generic or value-oriented brands and private label offerings. Its success depends on efficient manufacturing, strong distribution partnerships, and a pricing strategy that emphasizes affordability and accessibility. It aims to capture market share from consumers who prioritize cost-effectiveness for daily dry eye management.
Key Takeaways
- QC Artificial Tears Drops operates in a market with no active compound patent protection, allowing for broad generic competition.
- The market is intensely competitive, featuring numerous established brands, generic alternatives, and private label offerings.
- Manufacturing costs are driven by raw materials, stringent sterile processing, and packaging, with unit production costs estimated between $0.40 and $1.20 for a 15 mL bottle.
- Demand for dry eye treatments is growing robustly, fueled by an aging population, increased screen time, and environmental factors.
- Projected retail prices for a 15 mL bottle are expected to range between $8.00 and $13.50 over the next 2-3 years, with limited potential for significant increases due to competitive pressures.
- The product's competitive positioning is based on value, affordability, and accessibility, rather than proprietary innovation.
Frequently Asked Questions
-
Will QC Artificial Tears Drops introduce a preservative-free version? While not publicly announced, the market trend towards preservative-free formulations makes this a plausible future product extension. Manufacturers often develop variations to meet evolving consumer preferences and address concerns about preservative sensitivity.
-
How does the cost of raw materials impact the final price of QC Artificial Tears Drops? Fluctuations in the cost of key lubricants (e.g., carboxymethylcellulose sodium) and preservatives can necessitate price adjustments. Significant increases in these commodity prices are the primary driver for potential upward revisions in the product's retail price, though competitive pressures will moderate the extent of such increases.
-
What is the typical profit margin for a product like QC Artificial Tears Drops for the manufacturer? For generic OTC pharmaceuticals, manufacturers often operate on lower margins per unit compared to patented drugs. Profitability relies on high sales volumes. Margins can range from 15% to 30% for manufacturers, with a larger portion of the final retail price attributable to distribution and retail markup.
-
Are there any upcoming regulatory changes that could affect the production or pricing of QC Artificial Tears Drops? Current regulatory frameworks for OTC ophthalmic solutions are well-established (e.g., FDA guidelines in the U.S.). Significant new regulations that would drastically alter manufacturing costs or pricing are not anticipated in the short term, though ongoing pharmacovigilance and quality standards must be maintained.
-
How does the market share of QC Artificial Tears Drops compare to market leaders like Systane? As a specific brand of QC Artificial Tears Drops is not explicitly detailed, it's assumed to be a generic or private label. Market share for such products is fragmented. Established brands like Systane and Refresh hold significant market share (often 10-20% individually or collectively), while the aggregate share of numerous generic and private label products is substantial, exceeding 50% of the overall OTC dry eye market by volume.
Citations
[1] Grand View Research. (2023). Dry Eye Treatment Market Size, Share & Trends Analysis Report By Product Type (Tear Substitutes, Anti-Inflammatory Drugs, Punctal Plugs), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online), By Region, And Segment Forecasts, 2023 - 2030.
More… ↓
