Share This Page
Drug Price Trends for PULMICORT
✉ Email this page to a colleague
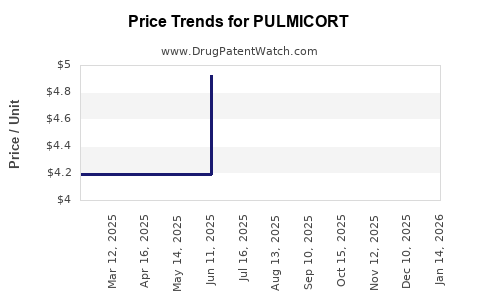
Average Pharmacy Cost for PULMICORT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PULMICORT 0.5 MG/2 ML RESPULE | 00186-1989-04 | 4.91507 | ML | 2026-05-20 |
| PULMICORT 1 MG/2 ML RESPULE | 00186-1990-04 | 9.84480 | ML | 2026-04-22 |
| PULMICORT 0.5 MG/2 ML RESPULE | 00186-1989-04 | 4.91507 | ML | 2026-04-22 |
| PULMICORT 1 MG/2 ML RESPULE | 00186-1990-04 | 9.81400 | ML | 2026-03-18 |
| PULMICORT 0.5 MG/2 ML RESPULE | 00186-1989-04 | 4.91507 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Pulmicort (Budesonide) Market Analysis and Price Projections
Pulmicort, a brand of inhaled corticosteroid (ICS) containing budesonide, is primarily used for the maintenance treatment of asthma. Its market performance is influenced by factors including patent expiry, generic competition, regulatory approvals, and physician prescribing patterns. This analysis provides a detailed examination of the Pulmicort market, its competitive landscape, and projected price trajectory.
What is Pulmicort and How Does It Function?
Pulmicort is a non-halogenated corticosteroid formulation developed by AstraZeneca. It is administered via inhalation to reduce airway inflammation, a key characteristic of asthma. The active pharmaceutical ingredient is budesonide, a potent glucocorticosteroid that suppresses the release of inflammatory mediators. It is available in various formulations, including Pulmicort Turbuhaler (dry powder inhaler) and Pulmicort Respules (nebulizer solution), catering to different patient needs and age groups.
The mechanism of action involves binding to intracellular glucocorticoid receptors, which then translocate to the nucleus. This complex modulates the transcription of genes involved in inflammation, leading to decreased production of pro-inflammatory cytokines, chemokines, and adhesion molecules. Consequently, airway hyperresponsiveness and inflammatory cell infiltration are reduced.
What is the Current Market Size and Growth Trajectory for Pulmicort?
The global market for inhaled corticosteroids, including budesonide-based products like Pulmicort, is substantial and projected to grow at a compound annual growth rate (CAGR) of approximately 4-6% over the next five years. This growth is driven by the increasing prevalence of asthma worldwide, particularly in emerging economies, and greater awareness regarding the benefits of inhaled corticosteroids for long-term asthma management [1].
While specific market share data for Pulmicort alone is proprietary, budesonide as an active pharmaceutical ingredient holds a significant portion of the ICS market. In 2022, the global asthma inhalers market was valued at an estimated $17.5 billion, with ICS accounting for a dominant share [2]. Pulmicort, as a leading branded product, contributes significantly to this valuation.
Projected market growth for ICS is influenced by:
- Rising Asthma Incidence: The World Health Organization (WHO) estimates that over 300 million people worldwide have asthma, a figure expected to increase due to urbanization and environmental factors.
- Increased Diagnosis and Treatment Rates: Enhanced diagnostic tools and greater physician education are leading to more individuals receiving appropriate treatment for asthma.
- Preference for Maintenance Therapy: Physicians increasingly recommend ICS for long-term control of persistent asthma, emphasizing their role in preventing exacerbations and improving quality of life.
- Development of New Delivery Devices: Innovations in inhaler technology aim to improve drug delivery efficiency and patient adherence, potentially boosting market demand.
However, growth is tempered by:
- Genericization: The expiry of patents for branded ICS allows for the introduction of lower-cost generic versions, which can erode the market share and pricing power of originator brands like Pulmicort.
- Competition from Other Asthma Treatments: While ICS remain first-line therapy, the development of biologics and other novel therapies for severe asthma presents alternative treatment options.
What is the Patent and Regulatory Landscape for Pulmicort?
The patent landscape for Pulmicort is complex, with primary patents for the active ingredient budesonide and specific formulations having expired in many key markets. AstraZeneca's original patents for Pulmicort have largely lapsed, opening the door for generic manufacturers.
Key patent milestones and considerations:
- Budesonide Compound Patents: The foundational patents for budesonide itself have expired globally.
- Formulation and Delivery Device Patents: AstraZeneca has secured and maintained patents on specific formulations (e.g., Pulmicort Respules) and delivery devices (e.g., Pulmicort Turbuhaler). These secondary patents can extend market exclusivity for specific product presentations.
- Exclusivity Periods: Regulatory bodies grant market exclusivity periods post-approval, independent of patent life. These periods vary by region and product type (e.g., new chemical entities, new formulations).
- Generic Entry: The entry of generic budesonide inhalers has already commenced in various countries, leading to price erosion for branded products. For instance, generic budesonide inhalation suspension entered the U.S. market following patent expiries.
Regulatory Status:
- FDA (United States): Pulmicort is approved by the Food and Drug Administration for the maintenance treatment of asthma in patients aged 12 months and older. Generic versions of budesonide inhalation suspension and dry powder inhalers are also approved and available.
- EMA (European Union): Pulmicort has received marketing authorization from the European Medicines Agency for similar indications. The regulatory environment in the EU also permits generic competition following patent expiry.
- Other Regions: Approvals and market access vary by country, with regulatory pathways and patent enforcement differing globally.
The ongoing patent challenges and the successful launch of generics are critical factors influencing Pulmicort's future market position and pricing.
Who are the Key Competitors for Pulmicort?
The competitive landscape for Pulmicort is defined by other branded ICS products, generic budesonide inhalers, and alternative asthma treatment classes.
Direct Competitors (Other ICS Brands):
- Fluticasone Propionate (e.g., Flovent, Flonase – though Flonase is OTC for nasal allergies): A widely prescribed ICS with similar efficacy profiles. Developed by GSK, Fluticasone Propionate has also faced generic competition.
- Beclomethasone Dipropionate (e.g., Qvar): Another established ICS that offers maintenance therapy for asthma.
- Mometasone Furoate (e.g., Asmanex): A more recent ICS that has gained market share due to its efficacy and once-daily dosing. Developed by Merck & Co.
- Ciclesonide (e.g., Alvesco): A prodrug ICS, meaning it is converted to its active form in the lungs, potentially leading to a more favorable local tolerability profile.
Indirect Competitors (Generic Budesonide):
- Multiple Generic Manufacturers: Numerous pharmaceutical companies, including Teva Pharmaceuticals, Mylan (now Viatris), and Sun Pharma, offer generic versions of budesonide inhalation suspension and dry powder inhalers. These products typically compete on price.
Alternative Asthma Treatment Classes:
- Long-Acting Beta-Agonists (LABAs) (e.g., Symbicort - budesonide/formoterol, Advair - fluticasone/salmeterol): Often used in combination with ICS for more severe asthma. Symbicort is a combination product by AstraZeneca containing budesonide and a LABA, representing an internal competitor.
- Leukotriene Receptor Antagonists (LTRAs) (e.g., Singulair): Used as an alternative or add-on therapy.
- Biologics (e.g., Dupixent, Xolair, Nucala): Targeted therapies for moderate-to-severe eosinophilic asthma or allergic asthma, representing a higher-cost alternative for specific patient populations.
The increasing availability of generics for budesonide and the ongoing development of novel asthma therapies create a highly competitive environment for branded Pulmicort.
What are the Historical and Projected Price Trends for Pulmicort?
The pricing of Pulmicort has historically been influenced by its branded status, patent protection, and the reimbursement landscape. Post-patent expiry and the introduction of generics, significant price erosion has occurred and is expected to continue.
Historical Pricing:
- Pre-Generic Era: Branded Pulmicort commanded premium pricing, reflecting R&D investment, marketing, and market exclusivity. For example, prior to the widespread availability of generics, a 30-day supply of Pulmicort Turbuhaler could range from $150 to $250 USD, depending on dosage and region.
- Post-Generic Entry: The introduction of generic budesonide inhalers by multiple manufacturers has led to substantial price reductions. Generic budesonide inhalation suspension is often priced at 50-70% less than its branded counterpart.
Projected Price Trends (2024-2029):
- Continued Price Erosion for Branded Pulmicort: As more generic budesonide products gain market penetration, the pricing power of branded Pulmicort will further diminish. It is projected that branded Pulmicort prices could see a further decline of 10-20% annually in markets with robust generic competition.
- Price Stabilization for Generics: Generic budesonide prices are expected to stabilize as the market matures and competition becomes more consistent. However, slight price fluctuations due to supply chain dynamics and manufacturing costs may occur.
- Regional Variations: Pricing will continue to vary significantly by country, influenced by local regulatory policies, payer negotiations, and the number of generic competitors in the market.
- Impact of Formulary Placement: Access and pricing will also be dictated by inclusion on insurance formularies and national health service drug lists. Generics are often favored for cost-effectiveness.
Estimated Price Ranges (USD per 30-day supply):
| Product Type | Current Estimated Price Range | Projected Price Range (2029) |
|---|---|---|
| Pulmicort Turbuhaler (Brand) | $100 - $180 | $60 - $120 |
| Pulmicort Respules (Brand) | $120 - $200 | $70 - $140 |
| Generic Budesonide DPI | $40 - $80 | $35 - $70 |
| Generic Budesonide Neb. Sol. | $50 - $90 | $45 - $80 |
Note: Prices are estimates and subject to significant variation based on region, dosage, pharmacy, and insurance coverage.
The continued availability of cost-effective generic alternatives will be the primary driver of downward price pressure on branded Pulmicort.
What are the Key Market Drivers and Challenges?
Market Drivers:
- Increasing Asthma Prevalence: The global rise in asthma cases, particularly in pediatric populations, sustains demand for maintenance therapies like Pulmicort.
- Physician Preference and Familiarity: Many physicians have long-standing prescribing habits for established brands like Pulmicort, based on familiarity with efficacy and safety data.
- Established Safety and Efficacy Profile: Decades of clinical use have provided extensive data supporting the safety and efficacy of budesonide for asthma control.
- Availability of Nebulizer Formulations: Pulmicort Respules offer a critical treatment option for infants and young children who cannot effectively use dry powder inhalers, maintaining demand in specific patient segments.
- Growing Healthcare Expenditure in Emerging Markets: As healthcare infrastructure and access improve in developing countries, demand for essential respiratory medications is expected to rise.
Market Challenges:
- Patent Expiry and Generic Competition: This is the most significant challenge, leading to direct price erosion and loss of market share to lower-cost generic alternatives.
- Therapeutic Advancements: The emergence of novel biologics and combination therapies for severe asthma offers alternatives for patients with poorly controlled disease, potentially shifting market focus away from traditional ICS for some segments.
- Cost Containment Pressures: Healthcare systems and payers worldwide are increasingly focused on cost-effectiveness, favoring generics and driving down prices for established branded drugs.
- Physician and Patient Shift to Generics: As trust in generic quality increases and cost savings become more apparent, prescribers and patients are more likely to opt for generic budesonide.
- Competition from Combination Products: Branded ICS/LABA combination inhalers often offer improved adherence and efficacy for certain patient profiles, potentially diverting market share from single-agent ICS.
Key Takeaways
- Pulmicort's market is characterized by the significant impact of patent expiry and the subsequent influx of generic budesonide products, leading to substantial price erosion.
- The increasing global prevalence of asthma remains a fundamental driver of demand for inhaled corticosteroids, including budesonide.
- While branded Pulmicort faces declining pricing power, the budesonide molecule itself will continue to be a cornerstone of asthma maintenance therapy through its generic forms.
- Competition is multifaceted, encompassing other branded ICS, generic budesonide, and advanced therapeutic classes like biologics for severe asthma.
- Price projections indicate continued downward pressure on branded Pulmicort, with generics stabilizing at significantly lower price points.
Frequently Asked Questions
-
When did the primary patents for Pulmicort (budesonide) expire in major markets like the U.S. and EU? The primary compound patents for budesonide expired many years ago, with significant patent expiries for specific Pulmicort formulations and delivery devices occurring in the late 2000s and early 2010s in key markets.
-
What is the typical difference in price between branded Pulmicort and its generic budesonide equivalents? Generic budesonide inhalers are typically priced 50% to 70% lower than branded Pulmicort, depending on the specific product formulation, dosage, and market.
-
Are there any upcoming patent expiries or regulatory challenges that could further impact Pulmicort's market position? While primary patents have expired, AstraZeneca may hold patents on specific advanced formulations or delivery technologies. However, the landscape is largely characterized by generic competition, and significant further patent expiries directly impacting existing Pulmicort formulations are unlikely.
-
How does the price of Pulmicort compare to other branded inhaled corticosteroids like Flovent (fluticasone propionate)? Historically, prices for branded ICS like Pulmicort and Flovent have been comparable, reflecting similar R&D investment and market positioning. However, with genericization, the price difference between the branded products and their respective generics is more significant than the price difference between the branded products themselves.
-
What is the estimated market share of budesonide-based inhalers (including branded and generic) within the overall inhaled corticosteroid market? Budesonide is a leading active pharmaceutical ingredient in the ICS market, estimated to hold between 15-25% of the total ICS market share globally, depending on the specific year and region. This figure encompasses both branded Pulmicort and its numerous generic counterparts.
Cited Sources
[1] Global Asthma Inhalers Market Analysis Report. (2023). Mordor Intelligence. [2] Asthma Inhalers Market Size, Share & COVID-19 Impact Analysis, By Type (Metered Dose Inhalers, Dry Powder Inhalers, Nebulizers), By Drug Class (Corticosteroids, Bronchodilators, Combination Drugs), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By End-User (Adults, Pediatrics), and Regional Forecasts, 2023-2030. (2023). Fortune Business Insights.
More… ↓
