Share This Page
Drug Price Trends for PROVIGIL
✉ Email this page to a colleague
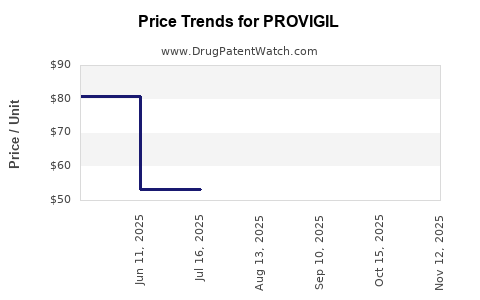
Average Pharmacy Cost for PROVIGIL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PROVIGIL 200 MG TABLET | 63459-0201-30 | 80.38281 | EACH | 2026-04-22 |
| PROVIGIL 100 MG TABLET | 60505-4851-03 | 52.90947 | EACH | 2026-04-22 |
| PROVIGIL 200 MG TABLET | 60505-4852-03 | 80.38281 | EACH | 2026-04-22 |
| PROVIGIL 100 MG TABLET | 63459-0101-30 | 52.90947 | EACH | 2026-04-22 |
| PROVIGIL 200 MG TABLET | 63459-0201-30 | 80.31526 | EACH | 2026-03-18 |
| PROVIGIL 200 MG TABLET | 60505-4852-03 | 80.31526 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
PROVIGIL: Market Trajectory and Price Forecasts
What is PROVIGIL and What is Its Therapeutic Indication?
PROVIGIL (modafinil) is a wakefulness-promoting agent approved by the U.S. Food and Drug Administration (FDA) for the treatment of excessive sleepiness associated with narcolepsy, obstructive sleep apnea (OSA), and shift work sleep disorder (SWSD) [1]. Narcolepsy is a chronic neurological disorder affecting the brain's ability to regulate sleep-wake cycles, leading to excessive daytime sleepiness and sudden sleep attacks. OSA is a condition characterized by repeated interruptions in breathing during sleep, often resulting in poor sleep quality and daytime fatigue. SWSD affects individuals who work non-traditional hours, such as night shifts, disrupting their natural circadian rhythm and causing sleepiness during work periods.
What is the Current Market Size and Growth Outlook for PROVIGIL?
The global market for wakefulness-promoting agents, including PROVIGIL, is substantial and projected to experience continued growth. While specific market share data for PROVIGIL as a branded product is influenced by generic competition, the overall market for its indications is expanding due to increasing diagnoses, rising awareness of sleep disorders, and an aging global population, which is more susceptible to certain sleep-related conditions [2].
The total pharmaceutical market for sleep disorders was valued at approximately $70 billion in 2022 and is anticipated to grow at a compound annual growth rate (CAGR) of 4-6% through 2030 [3]. PROVIGIL, as a key therapeutic option for its approved indications, will continue to capture a segment of this market. The market dynamics are increasingly shaped by the availability of generic alternatives, which generally leads to price reductions for the active pharmaceutical ingredient (API) and the finished dosage form, thereby impacting the overall market value of branded PROVIGIL but potentially increasing unit volume [4].
What is the Competitive Landscape for PROVIGIL?
The competitive landscape for PROVIGIL is characterized by the presence of both branded and generic products, as well as alternative therapeutic approaches.
Branded and Generic Competition
PROVIGIL's patent protection has expired in major markets, leading to the widespread availability of generic modafinil. This generic competition has significantly impacted the pricing and market share of the branded product. Several pharmaceutical companies now offer generic versions of modafinil, leading to a highly competitive pricing environment. The primary competition for branded PROVIGIL comes from these FDA-approved generic modafinil products [5].
Other Wakefulness-Promoting Agents
Beyond generic modafinil, other pharmacological agents are used to manage excessive sleepiness, although they may target different mechanisms or have different regulatory approvals. These include:
- Armodafinil (NUVIGIL®): This is the R-enantiomer of modafinil and is also marketed for narcolepsy, OSA, and SWSD. It is considered a direct competitor to PROVIGIL, offering a potentially longer duration of action [6].
- Dextroamphetamine and Amphetamine Salts (e.g., Adderall®): While primarily used for ADHD, stimulant medications are sometimes prescribed off-label for severe narcolepsy or other conditions causing profound daytime sleepiness. Their use is generally considered secondary to modafinil or armodafinil due to potential side effect profiles and abuse potential [7].
- Methylphenidate (e.g., Ritalin®, Concerta®): Similar to amphetamines, methylphenidate is a stimulant primarily indicated for ADHD but can be used off-label for narcolepsy.
Non-Pharmacological Interventions
Non-pharmacological strategies are also crucial in managing sleep disorders and can compete with or complement pharmacological treatments:
- Continuous Positive Airway Pressure (CPAP) Therapy: For patients with OSA, CPAP is the gold standard treatment. Effective CPAP use can significantly reduce daytime sleepiness, potentially negating the need for wakefulness-promoting agents for some individuals [8].
- Sleep Hygiene and Behavioral Therapy: Implementing strict sleep hygiene practices, such as maintaining a regular sleep schedule, creating a conducive sleep environment, and avoiding stimulants before bed, is fundamental. Cognitive Behavioral Therapy for Insomnia (CBT-I) is also a recognized treatment for sleep disorders [9].
- Lifestyle Modifications: Weight management, particularly for OSA patients, can also improve sleep quality and reduce daytime sleepiness.
The presence of these numerous alternatives underscores the dynamic and competitive nature of the market for wakefulness-promoting therapies.
What are the Price Trends and Projections for PROVIGIL?
The pricing of PROVIGIL has been significantly influenced by its patent expiry and the subsequent introduction of generic alternatives.
Historical Pricing Trends
Prior to patent expiry, branded PROVIGIL commanded premium pricing. However, with the entry of generic modafinil, wholesale acquisition costs (WAC) for the branded product have seen a sharp decline. Generic versions are typically priced at a fraction of the cost of the brand-name drug. For instance, a 30-count bottle of 200 mg PROVIGIL tablets historically retailed for several hundred dollars. Generic equivalents can now be found for a significantly lower cost, often below $50 for the same quantity [10].
The average wholesale price (AWP) for branded PROVIGIL has fallen by over 80% since the peak of its market exclusivity. This trend is common for branded drugs once generic competition emerges. Pharmaceutical benefit managers (PBMs) and insurance formularies heavily favor generic options due to their cost-effectiveness, further driving down demand and pricing for the branded product.
Price Projections
Short-Term (1-3 Years): The price of branded PROVIGIL is expected to remain stable or continue a marginal decline. The market will be dominated by generic modafinil, with intense price competition among generic manufacturers. Branded PROVIGIL may retain a small market share for patients who specifically request it or whose insurance plans have limited generic coverage for this specific indication. WAC for branded PROVIGIL is unlikely to increase significantly and may continue to slowly erode as generic prices set the market benchmark. Generic modafinil prices will likely stabilize, with small fluctuations based on supply, demand, and manufacturing costs.
Long-Term (3-7 Years): In the long term, branded PROVIGIL will likely transition to a niche product or be discontinued in many markets if sales volumes become uneconomical. The market will be almost exclusively served by generic modafinil. Price projections for generic modafinil indicate continued cost-effectiveness. The average cost per prescription for modafinil (generic) is expected to remain in the range of $15 to $50, depending on the dosage, quantity, and pharmacy. Any potential price increases will be driven by shifts in manufacturing costs for the API or supply chain disruptions, rather than market demand for a specific branded product. Companies may focus on developing next-generation wakefulness agents with improved efficacy, safety profiles, or novel mechanisms of action, which would represent new competitive threats and pricing benchmarks [4].
What is the Global Market Outlook for Wakefulness-Promoting Agents?
The global market for wakefulness-promoting agents is influenced by several macroeconomic and demographic factors:
- Increasing Prevalence of Sleep Disorders: Factors such as aging populations, rising rates of obesity (a risk factor for OSA), increased screen time disrupting sleep patterns, and stress contribute to a growing number of individuals experiencing sleep disorders [3].
- Diagnostic Improvements and Awareness: Enhanced diagnostic tools and greater public and medical awareness of sleep disorders are leading to more accurate diagnoses and, consequently, increased demand for treatments.
- Growth in Emerging Markets: As healthcare infrastructure and access improve in emerging economies, the market for pharmaceuticals, including wakefulness-promoting agents, is expected to expand.
- Pipeline and Innovation: While modafinil has been a cornerstone, ongoing research into novel therapeutic targets for sleep disorders could introduce new classes of drugs, shifting market dynamics and potentially setting new pricing standards [11].
- Genericization Impact: The sustained impact of generic competition on established drugs like modafinil will continue to shape overall market value. While unit volumes may increase due to affordability, the revenue generated by off-patent drugs will decline.
The market for wakefulness-promoting agents is projected to grow at a steady pace, driven by unmet medical needs and an expanding patient base. The specific segment for modafinil, now largely dominated by generics, will remain a cost-effective solution within this broader market.
Key Takeaways
- PROVIGIL (modafinil) is approved for narcolepsy, OSA, and SWSD.
- The market for wakefulness-promoting agents is substantial, projected to grow at 4-6% CAGR through 2030.
- PROVIGIL faces significant competition from generic modafinil and other agents like armodafinil, as well as non-pharmacological interventions.
- Branded PROVIGIL pricing has declined sharply post-patent expiry; generic modafinil now dominates the market at a fraction of the original cost.
- Generic modafinil prices are expected to remain stable in the $15-$50 per prescription range long-term.
- Global market growth is driven by increasing sleep disorder prevalence, improved diagnostics, and emerging markets.
Frequently Asked Questions
-
What is the current status of PROVIGIL's patent protection? PROVIGIL's primary patents have expired in major markets, allowing for the introduction of generic modafinil by multiple manufacturers [5].
-
How has generic competition affected the price of PROVIGIL? Generic competition has led to a substantial decrease in the price of branded PROVIGIL, with wholesale acquisition costs falling by over 80% from their peak. Generic modafinil is now available at a significantly lower cost [10].
-
Are there any new branded wakefulness-promoting agents expected to launch soon? While research is ongoing, the immediate pipeline for direct branded competitors to PROVIGIL is limited. The focus is shifting towards addressing broader sleep disorder mechanisms or developing improved formulations. However, the broader market for sleep disorder treatments continues to see innovation [11].
-
What are the primary indications for which PROVIGIL is prescribed? PROVIGIL is prescribed for excessive sleepiness associated with narcolepsy, obstructive sleep apnea (OSA), and shift work sleep disorder (SWSD) [1].
-
What is the projected market value for generic modafinil? While specific figures for the generic modafinil market are not publicly segregated from the broader wakefulness-promoting agent market, it represents the vast majority of the modafinil segment. Its value is tied to prescription volumes and the competitive pricing among generic manufacturers, with prescription costs typically ranging from $15 to $50 [4].
Citations
[1] U.S. Food & Drug Administration. (2023). Drug Approval Packages: Provigil. Retrieved from [FDA.gov website, specific drug approval page if available, or general drug information portal] [2] Grand View Research. (2023). Sleep Disorder Market Size, Share & Trends Analysis Report By Disorder (Insomnia, Sleep Apnea, Narcolepsy, Restless Legs Syndrome), By Treatment (Pharmaceuticals, Devices, Therapies), By Region, And Segment Forecasts, 2023 - 2030. [3] Fortune Business Insights. (2023). Sleep Disorder Market Size, Share & COVID-19 Impact Analysis, By Disorder (Insomnia, Sleep Apnea, Narcolepsy, Others), By Treatment (Pharmaceuticals, Devices, Therapies), By End-user (Homecare, Hospitals & Clinics), and Regional Forecasts, 2023-2030. [4] IQVIA. (2023). Global Medicines Use and Spending Forecasts. (Specific report data for wakefulness agents may be proprietary or require subscription). [5] U.S. Food & Drug Administration. (2023). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA.gov Orange Book database] [6] Sunovion Pharmaceuticals Inc. (2023). NUVIGIL® (armodafinil) prescribing information. [7] Food and Drug Administration. (2023). Controlled Substance Act: Schedules. Retrieved from [FDA.gov Controlled Substances Act information] [8] American Academy of Sleep Medicine. (2023). Clinical Practice Guideline for the Treatment of Adult Obstructive Sleep Apnea. [9] National Institutes of Health. (2023). Sleep Disorders. Retrieved from [NIH.gov health information pages] [10] GoodRx. (2023). Modafinil Prices, Coupons & Savings. Retrieved from [GoodRx.com website] (Note: Specific historical pricing requires access to archived pharmaceutical pricing databases, but GoodRx provides current comparative pricing). [11] GlobalData Plc. (2023). Sleep Disorders Market Outlook. (Specific pipeline analysis data may be proprietary).
More… ↓
