Share This Page
Drug Price Trends for PROPYLTHIOURACIL
✉ Email this page to a colleague
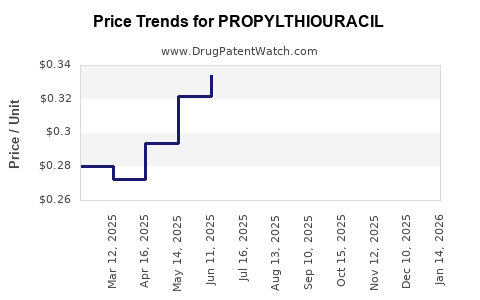
Average Pharmacy Cost for PROPYLTHIOURACIL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PROPYLTHIOURACIL 50 MG TABLET | 00480-9242-01 | 0.27237 | EACH | 2026-04-22 |
| PROPYLTHIOURACIL 50 MG TABLET | 62135-0606-90 | 0.27237 | EACH | 2026-04-22 |
| PROPYLTHIOURACIL 50 MG TABLET | 67253-0651-10 | 0.27237 | EACH | 2026-04-22 |
| PROPYLTHIOURACIL 50 MG TABLET | 33342-0314-11 | 0.27237 | EACH | 2026-04-22 |
| PROPYLTHIOURACIL 50 MG TABLET | 70752-0171-10 | 0.27237 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for PROPYLTHIOURACIL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| PROPYLTHIOURACIL 50MG TAB | Amerisource Health Services LLC dba American Health Packaging | 68084-0964-25 | 5X6 | 48.92 | 2023-09-15 - 2028-09-14 | FSS | ||
| PROPYLTHIOURACIL 50MG TAB | Amerisource Health Services LLC dba American Health Packaging | 68084-0964-25 | 5X6 | 47.99 | 2024-01-01 - 2028-09-14 | Big4 | ||
| PROPYLTHIOURACIL 50MG TAB | Amerisource Health Services LLC dba American Health Packaging | 68084-0964-25 | 5X6 | 48.63 | 2024-01-01 - 2028-09-14 | FSS | ||
| PROPYLTHIOURACIL 50MG TAB | Amerisource Health Services LLC dba American Health Packaging | 68084-0964-25 | 5X6 | 47.56 | 2023-09-15 - 2028-09-14 | Big4 | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Propylthiouracil Market Analysis and Price Projections
Propylthiouracil (PTU) is an antithyroid medication used to treat hyperthyroidism, particularly Graves' disease. The market for PTU is influenced by the prevalence of thyroid disorders, competitive landscape, regulatory environment, and manufacturing costs. Price projections are contingent on these factors, with potential impacts from generic competition and advancements in alternative treatments.
What is the Current Market Size and Growth Rate for Propylthiouracil?
The global market for propylthiouracil is a niche segment within the broader antithyroid drug market. Precise, up-to-date market size figures for PTU alone are often aggregated within larger categories due to its status as a mature generic drug. However, industry reports tracking the hyperthyroidism treatment market, where PTU plays a role, indicate a steady demand.
The prevalence of hyperthyroidism, a primary driver for PTU demand, is estimated to affect approximately 1-2% of the global population, with Graves' disease being the most common cause [1]. This consistent patient pool underpins the sustained demand for effective, affordable treatments like PTU.
While growth rates for individual generic drugs are rarely published with granular detail, the overall antithyroid market, including PTU, is projected to experience a Compound Annual Growth Rate (CAGR) of approximately 3-5% over the next five to seven years. This growth is attributed to an increasing awareness of thyroid disorders, improving diagnostic capabilities, and a rising incidence of autoimmune diseases [2].
Key Market Drivers:
- Prevalence of Hyperthyroidism: The sustained occurrence of conditions like Graves' disease directly translates to a consistent patient base requiring antithyroid therapy.
- Generic Drug Status: As an established generic, PTU offers a cost-effective treatment option, making it accessible in both developed and developing economies.
- Cost-Effectiveness: Compared to newer, branded therapies or surgical interventions, PTU's lower cost profile remains a significant advantage.
- Limited New Entrants in Generics: While the generic market can be competitive, the development and approval pathways for older drugs like PTU may deter new entrants unless significant cost efficiencies or manufacturing advantages are achievable.
Factors Limiting Growth:
- Availability of Alternatives: Propylthiouracil faces competition from other antithyroid drugs, notably methimazole, which is often considered a first-line treatment due to a generally more favorable safety profile [3].
- Adverse Event Profile: PTU has a known risk of serious adverse effects, including liver toxicity and agranulocytosis, which can limit its use in certain patient populations and encourage the adoption of alternatives [3, 4].
- Maturity of the Market: As a well-established generic, PTU is unlikely to experience rapid, disruptive growth. Demand is largely driven by existing treatment protocols and patient inertia.
What are the Primary Applications and Patient Populations for Propylthiouracil?
Propylthiouracil is primarily prescribed for the management of hyperthyroidism. Its main applications include:
- Treatment of Graves' Disease: This is the most common indication for PTU. Graves' disease is an autoimmune disorder where the thyroid gland produces excessive amounts of thyroid hormones [1].
- Management of Thyroid Storm: PTU is used in the urgent treatment of thyroid storm, a life-threatening exacerbation of hyperthyroidism, due to its ability to inhibit the peripheral conversion of thyroxine (T4) to triiodothyronine (T3) [5]. This dual action, blocking hormone synthesis and peripheral conversion, makes it valuable in acute settings.
- Pre-operative Preparation: PTU may be used to render a hyperthyroid patient euthyroid prior to thyroid surgery [1].
- Adjunctive Therapy: In some cases, it can be used as adjunctive therapy when radioactive iodine treatment is not immediately effective or is contraindicated.
Target Patient Populations:
- Pregnant Women: PTU is often the preferred antithyroid drug during the first trimester of pregnancy due to concerns about potential teratogenicity associated with methimazole [4, 6]. After the first trimester, methimazole may be substituted.
- Patients with Thyroid Storm: As mentioned, its rapid action makes it critical for managing this acute condition.
- Patients Intolerant to Methimazole: Individuals who experience adverse reactions to methimazole may be switched to PTU.
- Patients with Severe Hyperthyroidism: Its ability to block peripheral T4 to T3 conversion offers an advantage in managing severe cases.
The specific patient population can fluctuate based on clinical guidelines, pregnancy rates, and the incidence of adverse drug reactions to competing therapies.
What is the Competitive Landscape for Propylthiouracil?
The competitive landscape for propylthiouracil is characterized by its generic status and the availability of alternative antithyroid medications.
Direct Competitors (Other Antithyroid Drugs):
- Methimazole (Tapazole): This is the most significant competitor. Methimazole is generally considered the first-line treatment for hyperthyroidism due to its efficacy, longer duration of action (allowing for once-daily dosing in many cases), and a lower risk of certain severe adverse effects compared to PTU [3]. However, concerns regarding teratogenicity in the first trimester of pregnancy can lead to PTU being preferred.
- Radioactive Iodine (I-131): While not a drug in the traditional sense, radioactive iodine therapy is a common and often definitive treatment for hyperthyroidism, particularly Graves' disease. It permanently ablates thyroid tissue, eliminating the need for daily medication. Its use is typically reserved for patients who are not pregnant or breastfeeding and who have failed or are intolerant to antithyroid drugs.
- Surgical Thyroidectomy: In specific cases, surgical removal of the thyroid gland is performed. This is usually reserved for patients with very large goiters, suspected malignancy, or those who cannot tolerate medical therapy.
Indirect Competition:
- Beta-Blockers (e.g., Propranolol): These drugs do not treat the underlying hyperthyroidism but manage the symptoms, such as rapid heart rate, tremors, and anxiety. They are often used as adjunctive therapy or for short-term symptom control while waiting for antithyroid drugs or radioactive iodine to take effect.
Generic Manufacturers:
As a generic drug, PTU is manufactured by multiple pharmaceutical companies. The market is fragmented, with numerous suppliers of the active pharmaceutical ingredient (API) and finished dosage forms. Key factors in this segment include:
- API Sourcing and Cost: The cost of raw materials and the efficiency of API manufacturing significantly impact pricing.
- Regulatory Compliance: Adherence to Good Manufacturing Practices (GMP) and successful regulatory filings (e.g., Abbreviated New Drug Applications - ANDAs in the US) are critical.
- Distribution Networks: Establishing robust supply chains to reach pharmacies and healthcare providers globally.
The generic nature of PTU means that competition primarily occurs on price, supply reliability, and the ability to maintain high quality standards.
What are the Key Manufacturing and Supply Chain Considerations for Propylthiouracil?
The manufacturing and supply chain for propylthiouracil, like other generic pharmaceuticals, involve several critical considerations that impact availability and cost.
Active Pharmaceutical Ingredient (API) Manufacturing:
- Synthesis Route: PTU is synthesized through chemical processes. Manufacturers focus on optimizing these routes for yield, purity, and cost-effectiveness.
- Raw Material Sourcing: The availability and price volatility of precursor chemicals are significant factors. Supply chain disruptions or geopolitical issues affecting raw material suppliers can impact API production.
- Quality Control: Rigorous testing is required at each stage of API synthesis to ensure purity, potency, and freedom from contaminants, adhering to pharmacopeial standards (e.g., USP, EP).
- Geographic Concentration of API Production: Historically, a significant portion of API manufacturing has been concentrated in certain regions, such as China and India. This concentration can create vulnerabilities in the global supply chain.
Finished Dosage Form (FDF) Manufacturing:
- Formulation: PTU is typically formulated into oral tablets. Manufacturers must ensure consistent dissolution rates, stability, and bioavailability.
- Packaging: Packaging must protect the drug from light, moisture, and physical damage, and comply with regulatory labeling requirements.
- Scale of Production: Manufacturers must balance production capacity with market demand to avoid stockouts or oversupply.
- GMP Compliance: FDF manufacturing facilities must strictly adhere to current Good Manufacturing Practices (cGMP) mandated by regulatory agencies like the FDA and EMA.
Supply Chain Logistics:
- Distribution Channels: PTU reaches patients through wholesale distributors, retail pharmacies, and hospitals. Maintaining efficient logistics is crucial.
- Temperature and Storage Requirements: While PTU tablets are generally stable at room temperature, proper storage conditions must be maintained throughout the supply chain to preserve drug integrity.
- Regulatory Oversight: All stages, from API production to finished product distribution, are subject to regulatory inspections and compliance checks.
- Supply Chain Resilience: Recent global events have highlighted the importance of diversified sourcing and robust contingency plans to mitigate risks from natural disasters, pandemics, or trade disputes.
Key Players in the Supply Chain:
- API Manufacturers: Companies specializing in the bulk production of the active chemical compound.
- Contract Manufacturing Organizations (CMOs): Companies that manufacture finished dosage forms on behalf of marketing authorization holders.
- Finished Dosage Formulators: Companies that develop and manufacture the final drug product.
- Wholesalers and Distributors: Entities that manage the logistics of moving drugs from manufacturers to pharmacies and healthcare facilities.
The supply chain for generic drugs like PTU is highly sensitive to cost pressures and regulatory compliance. Any disruption, from raw material shortages to manufacturing plant shutdowns, can directly impact drug availability and pricing.
What are the Regulatory Considerations Affecting Propylthiouracil?
Propylthiouracil is subject to stringent regulatory oversight by health authorities worldwide, impacting its approval, manufacturing, marketing, and post-market surveillance.
Marketing Authorization and Approval:
- Generic Drug Approval Pathways: In most major markets (e.g., the U.S. with the FDA's ANDA pathway, Europe with national or centralized procedures), PTU as a generic drug requires demonstration of bioequivalence to an already approved reference listed drug.
- Pharmacopeial Standards: PTU must meet the quality standards set forth in national and international pharmacopeias, such as the United States Pharmacopeia (USP) and the European Pharmacopoeia (EP). These standards specify purity, assay, and limits for impurities.
- Labeling Requirements: Prescribing information, patient information leaflets, and packaging labels must comply with specific regulations regarding drug name, strength, dosage, indications, contraindications, warnings, precautions, adverse reactions, and storage.
Manufacturing and Quality Control:
- Good Manufacturing Practices (GMP): All manufacturing facilities (API and finished product) must comply with cGMP regulations. Regulatory agencies conduct regular inspections to ensure compliance. Any deviation can lead to product recalls, manufacturing halts, or import alerts.
- Drug Master Files (DMFs): API manufacturers typically maintain DMFs with regulatory agencies, detailing the manufacturing process, facilities, and quality controls. Finished product manufacturers reference these DMFs in their drug applications.
Post-Market Surveillance and Safety Monitoring:
- Adverse Event Reporting: Pharmaceutical companies are required to monitor and report adverse events associated with PTU to regulatory authorities. This forms the basis for pharmacovigilance.
- Risk Management Plans (RMPs) / Risk Evaluation and Mitigation Strategies (REMS): Due to the known risks of liver toxicity and agranulocytosis, PTU may be subject to specific safety monitoring programs or restrictions, particularly regarding its use in certain patient groups or for prolonged durations. The FDA, for example, has issued warnings and recommendations regarding liver injury associated with PTU [4].
- Product Recalls: Regulatory agencies can mandate recalls if safety, quality, or efficacy issues are identified.
International Harmonization:
- While efforts are made towards international harmonization (e.g., through the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use - ICH), specific regulatory requirements and interpretations can vary between countries, adding complexity for global manufacturers.
The regulatory environment plays a crucial role in ensuring the safety and efficacy of PTU. Compliance adds significant cost and time to development and manufacturing, but it is essential for market access and patient protection.
What are the Price Drivers and Projections for Propylthiouracil?
The pricing of propylthiouracil is primarily driven by its status as a mature generic drug. Projections are influenced by manufacturing costs, generic competition, and payer dynamics.
Current Pricing Factors:
- Manufacturing Costs: The cost of API synthesis, formulation, packaging, and regulatory compliance are fundamental cost drivers. Fluctuations in raw material prices and energy costs can impact overall manufacturing expenses.
- Generic Competition: The presence of multiple generic manufacturers creates downward pressure on prices. Competition is often intense, leading to narrow profit margins for individual products.
- Payer Influence (Reimbursement Policies): Pharmacy benefit managers (PBMs) and insurance companies negotiate prices with manufacturers and distributors. Their formulary decisions and preferred drug lists significantly influence market access and effective pricing. As a low-cost generic, PTU is generally well-covered, but its placement on formularies can be affected by the availability of even lower-cost alternatives or rebates.
- Wholesale Acquisition Cost (WAC) vs. Net Price: WAC is the list price. The actual net price received by manufacturers after rebates, discounts, and chargebacks is substantially lower.
- Supply Chain Markups: Wholesalers and distributors add their own margins as the drug moves through the supply chain.
Price Projections:
Given its market maturity and generic status, significant price increases for propylthiouracil are unlikely.
- Short to Medium Term (1-3 years): Prices are expected to remain stable or experience marginal declines. Continued generic competition and the pursuit of cost efficiencies by manufacturers will likely offset any inflationary pressures. Price adjustments might occur due to changes in raw material costs or minor manufacturing process improvements.
- Long Term (3-7 years): Little significant price appreciation is anticipated. The market dynamics for PTU are unlikely to change dramatically unless there is a major shift in treatment guidelines favoring PTU, or a substantial disruption in the supply of its main competitor, methimazole. Conversely, the development of novel therapies for hyperthyroidism that significantly displace existing treatments could lead to a decline in PTU demand and potentially further price erosion due to reduced sales volumes.
Factors that Could Influence Price Fluctuations:
- API Supply Disruptions: If a major API supplier faces production issues or is forced to cease operations, it could lead to temporary shortages and price spikes, particularly if alternative suppliers cannot quickly ramp up production.
- Regulatory Actions: New safety warnings or restrictions imposed on PTU could decrease demand, potentially leading to price reductions to incentivize use or clear existing inventory. Conversely, if PTU were found to have superior efficacy or safety in a specific niche patient population not previously recognized, this could marginally bolster demand, but unlikely lead to significant price increases given its generic nature.
- Withdrawal of Competitors: While unlikely for methimazole, if other antithyroid drugs were to be withdrawn from the market due to safety concerns, this could increase demand for PTU, potentially leading to slight price stabilization or a very modest increase, but still constrained by its generic status.
Estimated Price Range (Wholesale Acquisition Cost - WAC, US Market as Example):
As of late 2023/early 2024, the WAC for a bottle of 100 tablets (50 mg) of propylthiouracil can range from approximately $10 to $30 USD, depending on the manufacturer and distributor. The net price after discounts and rebates is considerably lower. These figures represent list prices and actual transaction prices will vary significantly.
The overall trend for established generics like PTU is one of price containment, driven by market forces and payer pressure.
Key Takeaways
Propylthiouracil (PTU) occupies a stable but niche segment of the antithyroid drug market, primarily driven by the consistent prevalence of hyperthyroidism and Graves' disease. Its cost-effectiveness as a generic medication ensures sustained demand, particularly in cost-sensitive markets and for specific patient populations like pregnant women. However, its competitive landscape is dominated by methimazole, which is often the first-line treatment, and the long-term curative option of radioactive iodine therapy. PTU’s known risk of serious adverse effects also constrains its widespread adoption.
Manufacturing and supply chains are characterized by generic production models, emphasizing cost efficiency, stringent GMP compliance, and global API sourcing. Regulatory oversight is critical, covering approval, manufacturing quality, and ongoing pharmacovigilance, with notable attention to PTU's safety profile regarding liver toxicity and agranulocytosis.
Price projections indicate continued stability or marginal decline for PTU due to intense generic competition and payer negotiations. Significant price increases are improbable; any fluctuations will likely stem from raw material costs, supply chain disruptions, or regulatory actions impacting demand. The market for PTU is mature, with its future trajectory closely tied to the management of thyroid disorders and the competitive dynamics of available treatment options.
Frequently Asked Questions
-
What is the primary difference in clinical use between propylthiouracil (PTU) and methimazole? Methimazole is generally the first-line treatment for hyperthyroidism due to its efficacy and once-daily dosing. PTU is often preferred during the first trimester of pregnancy and for managing thyroid storm due to its rapid action and ability to inhibit peripheral T4 to T3 conversion.
-
Are there significant long-term side effects associated with propylthiouracil? Yes, propylthiouracil carries risks of serious adverse effects, including liver toxicity and agranulocytosis (a severe drop in white blood cells). Regular monitoring is crucial when patients are treated with PTU.
-
How does the price of propylthiouracil compare to other antithyroid medications? As a generic drug, propylthiouracil is significantly less expensive than branded medications and often less costly than newer treatment modalities. Its price is comparable to other generic antithyroid drugs like methimazole.
-
What factors are most likely to cause a shortage of propylthiouracil? Shortages could arise from disruptions in the supply of active pharmaceutical ingredients (APIs), manufacturing plant issues, or increased demand that outstrips production capacity, especially if there are limited alternative suppliers.
-
Can propylthiouracil be used for all types of hyperthyroidism? While effective for hyperthyroidism generally, its use is guided by clinical guidelines and patient-specific factors, including pregnancy status, co-morbidities, and tolerance to other medications. It is particularly indicated for Graves' disease, thyroid storm, and pre-operative preparation.
Cited Sources
[1] Chaker, L., Lee, S. R., & Goldman, M. B. (2023). Graves’ disease in adults: Epidemiology, pathophysiology, and clinical manifestations. In M. M. Garg, S. S. V. Ramachandran, S. R. Lee, K. L. Mulay, & S. D. Singh (Eds.), UpToDate. Retrieved from https://www.uptodate.com (Subscription required)
[2] Grand View Research. (2023). Thyroid Disorder Treatment Market Size, Share & Trends Analysis Report By Treatment Type (Medications, Radioactive Iodine Therapy, Surgery), By Drug Class (Antithyroid Drugs, Beta-blockers), By Condition (Hyperthyroidism, Hypothyroidism), By Region, And Segment Forecasts, 2024 - 2030.
[3] Smith, T. J., & Hegedus, L. (2023). Treatment of hyperthyroidism in adults. In M. M. Garg, S. S. V. Ramachandran, S. R. Lee, K. L. Mulay, & S. D. Singh (Eds.), UpToDate. Retrieved from https://www.uptodate.com (Subscription required)
[4] U.S. Food & Drug Administration. (2023, April 26). FDA Drug Safety Communication: FDA recommends not using propulthiouracil (PTU) in patients with other medication for hyperthyroidism unless no other options are available. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-recommends-not-using-propylthiouracil-ptu-patients-other-medication
[5] Hage, M. P., & Azizi, F. (2018). Thyroid storm. In S. Melmed, K. S. Polonsky, P. R. Larsen, & H. M. Kronenberg (Eds.), Williams Textbook of Endocrinology (14th ed.). Elsevier.
[6] American Thyroid Association. (n.d.). Hyperthyroidism. Retrieved from https://www.thyroid.org/hyperthyroidism/
More… ↓
