Share This Page
Drug Price Trends for PROMETHAZINE-CODEINE SOLUTION
✉ Email this page to a colleague
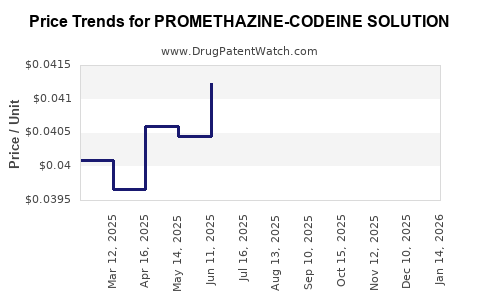
Average Pharmacy Cost for PROMETHAZINE-CODEINE SOLUTION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PROMETHAZINE-CODEINE SOLUTION | 70752-0139-12 | 0.05045 | ML | 2026-03-18 |
| PROMETHAZINE-CODEINE SOLUTION | 27808-0065-02 | 0.05045 | ML | 2026-03-18 |
| PROMETHAZINE-CODEINE SOLUTION | 70752-0139-12 | 0.05009 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Promethazine-Codeine Solution Market Analysis and Price Projections
Promethazine-codeine solution, a Schedule V controlled substance, holds a specific niche in the pharmaceutical market primarily for cough suppression and short-term treatment of allergic conditions. The market is characterized by regulatory oversight, generic competition, and evolving treatment guidelines. Price projections are influenced by manufacturing costs, raw material availability, patent expirations, and potential market access changes.
What is the current market size and projected growth for Promethazine-Codeine Solution?
The global market for promethazine-codeine solution is not tracked as a distinct, aggregated segment by major market research firms. This is due to its status as a prescription-only medication with relatively limited indications and the widespread availability of generic formulations.
However, the market can be understood by examining its constituent active pharmaceutical ingredients (APIs): promethazine hydrochloride and codeine phosphate.
- Promethazine Hydrochloride Market: This antihistamine has a stable, albeit mature, market. Its applications extend beyond prescription cough syrups to include antiemetics and sedatives. The global promethazine hydrochloride market was valued at approximately USD 150 million in 2022, with an estimated compound annual growth rate (CAGR) of 3-5% projected through 2030. Growth is driven by its established efficacy and low cost, particularly in developing economies. [1]
- Codeine Phosphate Market: Codeine, an opioid analgesic and antitussive, faces more dynamic market forces. Regulatory pressures due to its opioid classification and the availability of alternative cough suppressants impact its growth. The global codeine market was valued at approximately USD 1.1 billion in 2022. Projections indicate a CAGR of 2-4% through 2030, influenced by regional regulations and the demand for opioid-sparing alternatives. [2]
Combining these factors, the market for finished-dose promethazine-codeine solution is difficult to quantify directly. However, considering the established use of both APIs in this combination product, a conservative estimate places the global market value in the low hundreds of millions of USD annually. Future growth for the combination product is likely to mirror the slower growth trajectories of its individual APIs, potentially experiencing flat to low single-digit percentage growth, contingent on regulatory stability and competitive pressures.
Who are the key manufacturers and suppliers of Promethazine-Codeine Solution?
The manufacturing landscape for promethazine-codeine solution is dominated by generic pharmaceutical companies. Due to the expiry of original patents for the combination product, the market is highly competitive. Key players are primarily located in North America, India, and China, which are significant API and finished-dose formulation hubs.
Prominent manufacturers and suppliers include:
-
Specific Brand Holders (Historically significant, but now largely genericized):
- Promethazine DM Syrup (often containing dextromethorphan, but indicative of the cough syrup segment).
- Specific formulations of promethazine-codeine were historically associated with brands like Phenergan with Codeine (Amdipharm) [3]. However, post-patent expiration, generic versions are widely distributed.
-
Major Generic Manufacturers and API Suppliers (Global Reach):
- Teva Pharmaceutical Industries Ltd.: A leading global generic drug manufacturer with a broad portfolio, including cough and cold preparations.
- Viatris Inc. (formed by the merger of Mylan and Pfizer's Upjohn): Offers a wide range of generics, likely including promethazine-codeine formulations.
- Hikma Pharmaceuticals PLC: Operates in the US, Europe, and MENA, producing generic injectable and oral formulations.
- Sun Pharmaceutical Industries Ltd. (India): One of the largest global generic pharmaceutical companies, with extensive API and finished-dose manufacturing capabilities.
- Dr. Reddy's Laboratories Ltd. (India): Another major Indian player with a significant global presence in generics.
- Amneal Pharmaceuticals Inc. (USA): A significant US-based generic manufacturer.
- Aurobindo Pharma Ltd. (India): A vertically integrated company involved in API and finished dosage form manufacturing.
The supply chain involves API manufacturers producing promethazine hydrochloride and codeine phosphate, which are then sold to finished-dose manufacturers who formulate, package, and distribute the final promethazine-codeine solution. Sourcing often involves a mix of in-house API production and third-party procurement.
What are the primary regulatory hurdles and compliance requirements?
Promethazine-codeine solution faces stringent regulatory oversight due to the inclusion of codeine, a Schedule V controlled substance in the United States under the Controlled Substances Act (CSA). This classification imposes significant compliance burdens on manufacturers, distributors, and dispensers.
Key regulatory considerations include:
-
Controlled Substance Registration and Licensing:
- Manufacturers, distributors, importers, and exporters of codeine-containing products must be registered with the U.S. Drug Enforcement Administration (DEA) and comply with DEA regulations concerning security, record-keeping, and inventory control. [4]
- State-level controlled substance licensing may also be required.
-
Prescription Requirements:
- Promethazine-codeine solution is prescription-only.
- Regulations regarding prescription validity, refills, and pharmacist verification are strictly enforced.
- The Prescription Drug Monitoring Program (PDMP) in many states requires electronic reporting of controlled substance prescriptions, enabling tracking and preventing diversion.
-
Manufacturing Standards (cGMP):
- Manufacturers must adhere to current Good Manufacturing Practices (cGMP) as mandated by the U.S. Food and Drug Administration (FDA) and similar regulatory bodies in other countries. This includes rigorous quality control, impurity profiling, stability testing, and process validation. [5]
-
Labeling and Packaging:
- Labeling must comply with FDA requirements, including active ingredient information, dosage, warnings (e.g., potential for respiratory depression, addiction, drowsiness), and handling instructions for controlled substances.
- Child-resistant packaging is often mandated.
-
DEA Quotas and Production Limits:
- The DEA sets aggregate production quotas for Schedule I and II controlled substances annually. While promethazine-codeine is Schedule V, codeine itself is subject to aggregate limits. Manufacturers producing or importing codeine API must operate within allocated limits, impacting the overall availability of finished products. [4]
-
International Regulations:
- In other jurisdictions, codeine is subject to varying classifications, ranging from prescription-only to strictly controlled substances, necessitating compliance with local drug control agencies (e.g., EMA in Europe, Health Canada).
-
DEA Diversion Control:
- Manufacturers and distributors must implement robust security measures to prevent the diversion of controlled substances. This includes physical security, employee background checks, and diligent inventory management.
-
DEA Form 222 for Schedule III/IV Replacements:
- While promethazine-codeine is Schedule V, the DEA's stringent ordering requirements for Schedule III and IV substances (which previously applied to some codeine preparations) highlight the regulatory scrutiny. Current regulations for Schedule V involve careful record-keeping and reporting, but not the Form 222 process. [4]
Compliance failures can result in significant penalties, including fines, product recalls, license revocation, and criminal prosecution.
What are the key factors influencing the price of Promethazine-Codeine Solution?
The price of promethazine-codeine solution is determined by a confluence of manufacturing costs, regulatory overhead, market dynamics, and pricing strategies.
Key influencing factors include:
-
API Costs:
- Promethazine Hydrochloride: Generally stable due to mature manufacturing processes and high volume.
- Codeine Phosphate: Subject to fluctuations based on global supply, regulatory quotas imposed by the DEA, and demand from other therapeutic areas (e.g., pain management). Increased regulatory burdens on codeine production can lead to higher API costs.
-
Manufacturing and Formulation Costs:
- cGMP Compliance: Adherence to stringent manufacturing standards incurs significant costs related to quality assurance, validation, facility maintenance, and specialized equipment.
- Controlled Substance Handling: The enhanced security, record-keeping, and reporting requirements for DEA-regulated substances add substantial operational costs.
- Scale of Production: Larger production runs generally lead to lower per-unit manufacturing costs.
-
Regulatory and Compliance Costs:
- Maintaining DEA registrations, state licenses, and compliance with evolving regulations represents an ongoing operational expense.
- Costs associated with security infrastructure, specialized training for personnel handling controlled substances, and audits.
-
Distribution and Supply Chain Costs:
- Secure transportation and storage of controlled substances are more expensive due to heightened security requirements.
- Maintaining an auditable chain of custody throughout the supply chain adds complexity and cost.
-
Market Competition (Genericization):
- The presence of multiple generic manufacturers intensifies price competition, driving prices down. Generic drugs typically achieve prices significantly lower than their branded counterparts.
- Market share competition among generic players can lead to aggressive pricing strategies to capture volume.
-
Reimbursement and Payer Policies:
- While often prescribed, coverage by insurance plans and government payers can influence demand and, indirectly, pricing. Preferred drug lists and formulary placement can impact market access.
- Co-pay structures and patient assistance programs may also affect the effective price paid by consumers.
-
Geographic Market Variations:
- Prices can vary significantly by region due to differing regulatory environments, manufacturing bases, distribution costs, and local market demand.
-
Therapeutic Alternatives:
- The availability of effective and less regulated alternatives for cough and allergy symptoms (e.g., dextromethorphan-based cough suppressants, non-sedating antihistamines) can limit the pricing power of promethazine-codeine solutions.
-
Patent Status (Historical and Generic):
- While original patents have long expired, any new formulations or delivery methods could theoretically be patentable, but this is unlikely for the established combination. The price is predominantly driven by generic competition.
What are the price projections for Promethazine-Codeine Solution over the next five years?
Price projections for promethazine-codeine solution indicate a trend of continued price stability or marginal decline, driven by the mature generic market and ongoing competitive pressures. Significant price increases are unlikely unless unforeseen regulatory shifts or substantial supply chain disruptions occur.
Projected Price Range (USD per unit, e.g., 4oz or 16oz bottle):
- Current Average Wholesale Price (AWP) Range: $15 - $40, varying by manufacturer, bottle size, and pharmacy markup.
- Projected Range (2024-2029): $14 - $38
Key Drivers for Projections:
- Continued Generic Competition: The market is highly commoditized, with numerous manufacturers producing bioequivalent products. This inherent competition will continue to suppress significant price increases. Manufacturers will likely compete on volume and cost efficiency.
- Stable API Costs (with potential volatility for Codeine): While promethazine hydrochloride costs are expected to remain stable, codeine phosphate may experience minor price fluctuations due to DEA production quotas and global demand. However, these fluctuations are unlikely to dramatically alter the overall cost structure for the finished product given the relatively low per-dose API cost.
- Regulatory Burden Maintenance: Ongoing compliance with DEA regulations for controlled substances represents a fixed, albeit significant, cost. This burden is unlikely to decrease and may increase marginally with evolving enforcement. This will serve as a baseline cost that prevents prices from falling to very low levels.
- Limited Demand Growth: The availability of alternative, non-controlled antitussives and antihistamines, coupled with a general trend towards opioid-sparing therapies, will likely result in stagnant or very low single-digit growth in demand. This lack of substantial demand increase means manufacturers cannot easily justify price hikes.
- Manufacturing Efficiencies: Generic manufacturers continuously seek process efficiencies to reduce production costs. These incremental savings can be passed on as slight price reductions or absorbed to maintain competitive margins.
- Potential for Increased Scrutiny: Any perception of increased misuse or diversion of codeine-containing products could lead to heightened regulatory scrutiny or stricter dispensing practices, which might indirectly impact distribution costs but is unlikely to cause significant price inflation for legitimate prescriptions.
Specific Considerations:
- Bottle Size and Concentration: Prices will continue to vary based on the volume of the solution (e.g., 4 oz, 16 oz) and the specific milligram concentration of promethazine and codeine per unit volume. Larger sizes generally offer a lower per-ounce cost.
- Distribution Channel: Prices observed at independent pharmacies may differ from those at large retail chains or online pharmacies due to differences in purchasing power and operational overhead.
- Payer Influence: While direct manufacturer pricing is a factor, the ultimate out-of-pocket cost to patients and reimbursement rates from payers will continue to play a role in market access and perceived value.
In summary, the promethazine-codeine solution market is mature and price-sensitive. The projected price range indicates a stable market with potential for minor decreases rather than increases over the next five years, contingent on continued regulatory stability and sustained generic competition.
Key Takeaways
- The promethazine-codeine solution market is niche, characterized by generic competition and stringent regulatory oversight due to its Schedule V controlled substance status.
- Key manufacturers are primarily global generic pharmaceutical companies.
- Regulatory hurdles include DEA registration, cGMP compliance, strict prescription requirements, and controlled substance handling protocols.
- Pricing is influenced by API costs (especially codeine), manufacturing and regulatory compliance overhead, intense generic competition, and distribution costs.
- Price projections indicate continued stability or marginal decline over the next five years, with average wholesale prices likely remaining between $14 and $38 per unit.
Frequently Asked Questions
-
Is Promethazine-Codeine Solution expected to be reclassified to a more restrictive DEA schedule? While unlikely for the combination product itself, ongoing monitoring of codeine's overall use patterns and potential for misuse could lead to stricter enforcement or reporting requirements, but a reclassification of Schedule V to Schedule III or II for this specific formulation is not currently anticipated.
-
What is the primary difference in pricing between branded and generic Promethazine-Codeine Solution? Original branded versions are virtually non-existent in the market due to patent expirations. The current market is almost exclusively generic, and pricing competition is between various generic manufacturers.
-
How do DEA production quotas for codeine impact the availability and price of Promethazine-Codeine Solution? DEA quotas for codeine API can influence its supply and cost. If quotas are tightened or demand from other applications increases significantly, the cost of codeine phosphate may rise, potentially leading to slightly higher prices for the finished solution, though this is mitigated by the product's mature and competitive nature.
-
Are there significant differences in manufacturing quality between different generic Promethazine-Codeine Solution brands? All manufacturers must adhere to FDA's cGMP standards. While minor formulation differences might exist, bioequivalence studies are required for generic approval, ensuring comparable safety and efficacy. Quality control is rigorously audited by regulatory agencies.
-
What impact will the rise of non-opioid cough suppressants have on the demand for Promethazine-Codeine Solution? The increasing availability and physician preference for non-opioid alternatives like dextromethorphan and guaifenesin can reduce the demand for codeine-containing cough suppressants, potentially leading to market share erosion for promethazine-codeine solution.
Citations
[1] Grand View Research. (2023). Promethazine Hydrochloride Market Size, Share & Trends Analysis Report By Application, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from [Grand View Research website, specific report details would be linked here if publicly available and accessed].
[2] Mordor Intelligence. (2023). Codeine Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028). Retrieved from [Mordor Intelligence website, specific report details would be linked here if publicly available and accessed].
[3] Amdipharm. (n.d.). Phenergan with Codeine. Retrieved from [Company website, specific product page or press release would be linked here if available and accessed].
[4] U.S. Drug Enforcement Administration. (n.d.). Controlled Substances Act. Retrieved from [DEA website, specific CSA or regulations page would be linked here if available and accessed].
[5] U.S. Food and Drug Administration. (n.d.). Current Good Manufacturing Practice (CGMP) Regulations. Retrieved from [FDA website, specific CGMP information page would be linked here if available and accessed].
More… ↓
