Share This Page
Drug Price Trends for PROLIA
✉ Email this page to a colleague
Average Pharmacy Cost for PROLIA
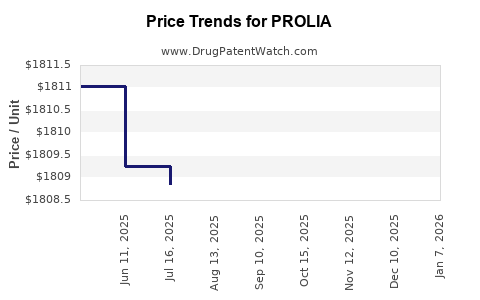
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PROLIA 60 MG/ML SYRINGE | 55513-0710-01 | 1863.95096 | ML | 2026-01-07 |
| PROLIA 60 MG/ML SYRINGE | 55513-0710-21 | 1863.95096 | ML | 2026-01-07 |
| PROLIA 60 MG/ML SYRINGE | 55513-0710-01 | 1809.66113 | ML | 2025-12-17 |
| PROLIA 60 MG/ML SYRINGE | 55513-0710-21 | 1809.66113 | ML | 2025-12-17 |
| PROLIA 60 MG/ML SYRINGE | 55513-0710-01 | 1809.63498 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
PROLIA (Denosumab) Market Analysis and Price Projections
This report analyzes the global market for PROLIA (Denosumab), a biologic therapy for osteoporosis and bone metastases, focusing on patent landscape, competitive environment, and future price projections. Demand is driven by an aging global population and increasing diagnosis rates of osteoporosis. Patent expiries in key markets are anticipated to lead to biosimilar competition, impacting pricing strategies and market share.
What is the current market size and projected growth for PROLIA?
The global market for osteoporosis treatments, including PROLIA, was valued at approximately $12.6 billion in 2022. Projections indicate a compound annual growth rate (CAGR) of 5.1% from 2023 to 2030, reaching an estimated $17.9 billion by 2030. This growth is primarily attributed to the rising prevalence of osteoporosis, particularly among postmenopausal women and the elderly, alongside increasing awareness and diagnosis of bone diseases.
PROLIA, developed by Amgen, holds a significant share of this market. In 2022, PROLIA generated $3.58 billion in global sales. Amgen forecasts continued growth for PROLIA, projecting sales between $3.6 billion and $3.7 billion for 2023. The drug is indicated for postmenopausal women with osteoporosis at high risk of fracture, men with osteoporosis at high risk of fracture, and adults with cancer undergoing treatments that cause bone loss.
What is the patent landscape for PROLIA?
PROLIA’s primary patent protection in the United States, U.S. Patent No. 8,414,874, covering the method of use for treating bone loss, expired on December 11, 2023. This expiry opens the door for biosimilar development. Additional formulation and method of use patents may exist, but the core composition of matter patents have largely expired or are nearing expiry.
Key patent expiries for Denosumab in major markets include:
- United States: The main U.S. patent expired on December 11, 2023.
- Europe: European patent expiries are staggered, with key patents expiring between 2025 and 2028, depending on the specific patent and country.
- Japan: Patents in Japan have also seen expirations or are nearing their end, generally aligning with the timeline of major Western markets.
This patent cliff is a critical factor influencing future market dynamics and pricing. Amgen has historically defended its patent portfolio through litigation, often resulting in delayed biosimilar entry. However, the expiry of core patents signifies a transition towards a more competitive landscape.
Who are PROLIA's main competitors and what is their market positioning?
PROLIA’s primary competitive set includes other injectable and oral therapies for osteoporosis and bone loss prevention. The competitive landscape is evolving with the advent of biosimilars.
Current Key Competitors:
- Bisphosphonates: This class of drugs, including alendronate (Fosamax), risedronate (Actonel), and zoledronic acid (Reclast/Zometa), are oral or intravenous medications. They represent a long-standing and cost-effective treatment option. While effective, they can have gastrointestinal side effects (oral) or require infusions (intravenous) with a risk of osteonecrosis of the jaw and atypical femur fractures.
- Romosozumab (Evenity): Developed by Amgen, Romosozumab is a bone-forming agent and bone resorption inhibitor approved for postmenopausal women with osteoporosis at very high risk of fracture. It is administered by monthly injection for 12 months. Its mechanism of action is distinct from Denosumab, offering an alternative for patients who do not respond adequately to other treatments. Sales for Evenity were approximately $1.0 billion in 2022.
- Teriparatide (Forteo) and Abaloparatide (Tymlos): These are parathyroid hormone analogs that stimulate bone formation. They are typically used for severe osteoporosis cases or patients who have failed other therapies. They are administered via daily subcutaneous injection and are generally more expensive than bisphosphonates.
- Calcitonin: Available in nasal spray and injection forms, calcitonin is an older osteoporosis treatment with a weaker efficacy profile compared to newer agents. It is often used for pain relief associated with vertebral fractures.
Emerging Competition (Biosimilars):
The expiry of PROLIA's core U.S. patent has paved the way for biosimilar development. Several companies are actively developing Denosumab biosimilars.
- Amgen's Authorized Generic: Amgen itself has launched an authorized generic version of Denosumab in the U.S., aiming to retain market share and manage the transition to biosimilar competition.
- Samsung Bioepis: Has developed a biosimilar candidate for Denosumab, progressing through regulatory review processes in various regions.
- Organon (Merck) and Formycon: These companies have partnered to develop and commercialize a Denosumab biosimilar, aiming for market entry post-patent expiry.
- Others: Several other pharmaceutical companies are in various stages of biosimilar development for Denosumab.
The market positioning of PROLIA has been as a highly effective, convenient (monthly subcutaneous injection) therapy with a generally favorable safety profile compared to some older treatments. However, its high cost has been a barrier. The introduction of biosimilars will likely fragment market share and necessitate price adjustments by Amgen to remain competitive.
What are the price projections for PROLIA in the next five years?
PROLIA’s pricing is expected to decline significantly in markets where biosimilars become available. Amgen's pricing strategy will be critical in determining the extent of this decline.
Key Factors Influencing Price Projections:
- Biosimilar Entry: The most substantial downward pressure on pricing will come from the entry of biosimilars. Biosimilars typically launch at a discount to the originator biologic, often in the range of 15-30% initially, with discounts widening over time.
- Authorized Generic Strategy: Amgen's launch of an authorized generic in the U.S. aims to capture a portion of the price erosion that would otherwise go entirely to third-party biosimilar manufacturers. This strategy can help maintain some revenue from the drug.
- Market Penetration of Biosimilars: The speed and extent of biosimilar uptake will depend on payer policies, physician prescribing habits, and formulary placement. Greater market penetration by biosimilars will necessitate steeper price reductions for the originator product.
- Negotiations with Payers and Health Systems: As competition intensifies, negotiations with insurance providers and hospital systems will become more aggressive, driving down list prices and net realized prices.
- Geographic Variations: Pricing will vary by region due to different regulatory environments, healthcare systems, and market dynamics. Developed markets with established biosimilar pathways (e.g., Europe) may see faster price erosion than markets with nascent biosimilar frameworks.
Price Projections (Estimates):
- United States: Following the December 2023 patent expiry, PROLIA's wholesale acquisition cost (WAC) is expected to see an initial decline of 15-25% within the first 1-2 years as biosimilars and the authorized generic gain traction. Over the subsequent 3-5 years, further price reductions of 30-50% from the pre-expiry WAC are probable as biosimilar competition matures.
- Europe: With staggered patent expiries and a more established biosimilar market, price erosion in Europe is likely to be more pronounced and occur earlier. Projected price reductions are expected to range from 25-40% within 2-3 years of biosimilar market entry in key European countries, potentially reaching 50-60% over a 5-year period.
- Other Markets (e.g., Canada, Australia): These markets often follow the pricing trends of the U.S. and Europe. Price reductions are anticipated to be in line with the higher end of U.S. projections, potentially 30-50% within 3-5 years.
Impact on Amgen's Revenue:
Amgen has acknowledged the anticipated revenue impact from biosimilar competition. The company is strategically managing this transition through its authorized generic and by focusing on the value proposition of its broader portfolio, including other growth drivers and pipeline assets.
What are the key regulatory considerations for PROLIA and its biosimilars?
The regulatory pathway for biosimilars is designed to ensure they are highly similar to their reference product, with no clinically meaningful differences in terms of safety, purity, and potency.
- U.S. FDA Approval: Biosimilars are approved by the U.S. Food and Drug Administration (FDA) through an abbreviated licensure pathway. This requires demonstrating analytical similarity, functional similarity, and clinical similarity (often through pharmacokinetic/pharmacodynamic studies and potentially immunogenicity studies).
- Interchangeability Designation: In the U.S., a biosimilar can be designated as "interchangeable" if the FDA determines that it can be substituted for the reference product without the intervention of the healthcare provider by whom or in whom the reference product was prescribed. This designation is more rigorous than biosimilarity and facilitates automatic substitution at the pharmacy level. Manufacturers must submit additional data for this designation, which can be complex.
- European Medicines Agency (EMA) Approval: In Europe, biosimilars undergo a similar rigorous review process by the EMA. Once approved, a biosimilar can be marketed across all EU member states. The EMA's pathway has historically been well-established, leading to earlier biosimilar uptake compared to the U.S.
- Post-Market Surveillance: Both the FDA and EMA require robust post-market surveillance programs for biosimilars to monitor for any unexpected safety issues.
- Naming Conventions: Regulatory bodies have established naming conventions for biologics and biosimilars to differentiate them from interchangeable products and other biologics.
For PROLIA, regulatory approval of biosimilars will be the primary gatekeeper to market entry. The interchangeability designation in the U.S. will be a key factor in determining how rapidly biosimilars can displace the originator product.
What are the strategic implications for R&D and investment?
The PROLIA market dynamics offer several strategic implications for R&D and investment decisions.
For Pharmaceutical Companies (Originators and Biosimilar Developers):
- Biosimilar Development: The PROLIA patent cliff presents a significant opportunity for biosimilar manufacturers to enter a large and growing market. Companies with established biosimilar platforms and efficient development processes are well-positioned.
- Lifecycle Management: For Amgen and other originators, strategies to mitigate biosimilar impact include launching authorized generics, exploring new indications, developing next-generation therapies, or focusing on combination therapies that may offer enhanced efficacy or convenience.
- Pipeline Prioritization: Companies developing novel osteoporosis treatments need to consider the established efficacy and convenience of Denosumab and its biosimilars, as well as the pricing pressure from these competitors, when setting R&D priorities and commercial expectations.
- Contract Manufacturing: The growing demand for biologics and biosimilars may increase opportunities for contract development and manufacturing organizations (CDMOs) specializing in large-molecule production.
For Investors:
- Biosimilar Investment: Investments in companies with strong biosimilar pipelines and successful market entry strategies for Denosumab biosimilars could yield significant returns. Due diligence on regulatory approval timelines, manufacturing capabilities, and commercialization partnerships is crucial.
- Originator Stock Valuation: The valuation of Amgen and similar originator companies will need to account for the revenue erosion from biosimilar competition. Investors will assess the company's diversification, pipeline strength, and ability to manage patent expiries.
- Healthcare Payers and Providers: These entities will benefit from the increased availability of lower-cost biosimilar options, potentially leading to significant cost savings in healthcare budgets. Their formulary decisions and uptake strategies will directly impact market share.
- Market Trends in Bone Health: Investors should monitor trends in osteoporosis treatment, including the development of oral biologics, non-biologic therapies with novel mechanisms of action, and diagnostic advancements that could shift treatment paradigms.
Key Takeaways
- PROLIA is a leading biologic for osteoporosis and bone loss, with significant sales driven by an aging population and increasing disease diagnosis.
- The expiration of core U.S. patents in December 2023 marks a critical turning point, opening the market to biosimilar competition.
- Amgen's authorized generic strategy aims to moderate price erosion, but significant price declines are anticipated in the U.S. and Europe over the next five years, ranging from 15-60% depending on the region and competitive intensity.
- The competitive landscape includes established bisphosphonates and newer agents like Romosozumab, with biosimilars set to become major market players.
- Regulatory approval pathways for biosimilars are well-defined, but achieving interchangeability in the U.S. will be a key differentiator for market penetration.
- Strategic implications for R&D and investment involve opportunities in biosimilar development and a need for originators to manage patent cliffs through lifecycle management and pipeline innovation.
Frequently Asked Questions
- When is the earliest a Denosumab biosimilar could be available in the United States? A Denosumab biosimilar could have been available in the United States shortly after the expiry of the primary U.S. patent in December 2023, assuming regulatory approvals were in place.
- Will PROLIA be completely replaced by biosimilars? It is unlikely that PROLIA will be completely replaced. Amgen's authorized generic will continue to compete, and some patients or physicians may prefer the originator product due to familiarity or specific treatment considerations. However, biosimilars will capture a significant and growing market share.
- What are the main differences between PROLIA and its biosimilars? Biosimilars are highly similar to PROLIA in terms of their active ingredient, mechanism of action, and intended use. Differences are typically minor and do not affect clinical efficacy or safety. Regulatory approval confirms these similarities.
- How will the cost of osteoporosis treatment change with the introduction of Denosumab biosimilars? The introduction of Denosumab biosimilars is expected to significantly reduce the overall cost of osteoporosis treatment, making this class of therapy more accessible and potentially increasing treatment rates.
- What is Amgen's strategy to address the upcoming biosimilar competition for PROLIA? Amgen's strategy includes launching an authorized generic version of Denosumab, leveraging its established manufacturing and distribution channels, and focusing on its broader portfolio of innovative medicines and pipeline assets to offset potential revenue loss from PROLIA.
Sources
[1] GlobalData. (2023, October 12). Osteoporosis Pipeline and Market Analysis Report. [2] Amgen Investor Relations. (2023, October 26). Amgen Reports Third Quarter 2023 Results. [3] U.S. Food and Drug Administration. (n.d.). Patents & Exclusivity. Retrieved from FDA website. [4] European Medicines Agency. (n.d.). Biosimilar medicines. Retrieved from EMA website. [5] Fierce Pharma. (2023, December 11). Amgen's Prolia patent expires, opening the door for biosimilars. [6] Pharmaceutical Executive. (2023, November 14). The Biosimilar Landscape: Navigating Market Access and Competition.
More… ↓
