Share This Page
Drug Price Trends for PREMARIN VAGINAL CREAM-APPL
✉ Email this page to a colleague
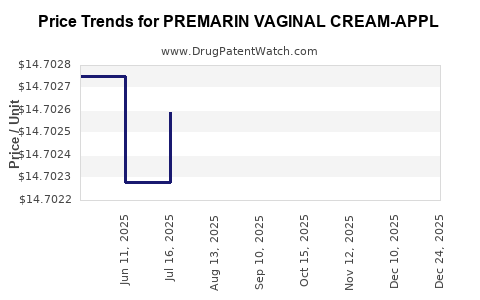
Average Pharmacy Cost for PREMARIN VAGINAL CREAM-APPL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PREMARIN VAGINAL CREAM-APPL | 00046-0872-21 | 15.09907 | GM | 2026-01-01 |
| PREMARIN VAGINAL CREAM-APPL | 00046-0872-21 | 14.70225 | GM | 2025-12-17 |
| PREMARIN VAGINAL CREAM-APPL | 00046-0872-21 | 14.70234 | GM | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for PREMARIN VAGINAL CREAM-APPL
Overview
PREMARIN Vaginal Cream, a hormone replacement therapy (HRT) product containing conjugated estrogens, is used primarily to treat atrophic vaginitis in postmenopausal women. The product is marketed by Novo Nordisk and has been available in the United States since the late 1990s. Its market has persisted despite multiyear declines driven by changing physician prescribing patterns, regulatory guidance, and competition from alternative therapies.
Market Size and Key Drivers
Patient Population
- Estimated postmenopausal women in the U.S.: 45 million (HealthData Research, 2022)
- Women with symptomatic atrophic vaginitis: 10%–20% of postmenopausal women (~4.5–9 million)
- Prescribed hormone therapy: approximately 25%–30% of women with symptoms (~1.1–2.7 million annual users)
Regulatory Environment & Trends
- FDA Guidance (2013): Emphasized risk reduction for hormone therapies
- Decline in prescriptions: 35% over five years ending 2021 (IQVIA data)
- Increase in non-hormonal, local treatments and alternative therapies
Market Revenue Estimates
- Estimated U.S. market for vaginal estrogen products: $200 million (2022)
- PREMARIN's market share: approximately 10–15%, corresponding to $20–30 million annually
Competitive Landscape
Major Competitors
- Estrace Vaginal Cream (almond estrogen, Allergan)
- Osphena (ospemifene, Shionogi)
- Local therapies: vaginal moisturizers, lubricants, or non-estrogen options
Product Differentiators
- Established brand with long safety record
- Prescribed predominantly by gynecologists and primary care physicians
- Availability of compounded formulations offers an alternative but with regulatory and safety concerns
Pricing Analysis
Current Pricing in the U.S.
- Brand: PREMARIN Vaginal Cream (50 mcg/gram)
- Pack Size: Typically 2.5g applicators, with a one-month supply
- Wholesale Acquisition Cost (WAC): ~$30–$40 per applicator as of 2023
- Retail Price: $50–$70 per month, depending on pharmacy
Price Comparison with Competitors
| Product | Average Monthly Price | Active Ingredient & Strength |
|---|---|---|
| PREMARIN Vaginal Cream | $50–$70 | 0.625 mg conjugated estrogens per gram |
| Estrace Vaginal Cream | $45–$60 | 0.1 mg per gram |
| Osphena | $200–$300 | Ospemifene (oral, 60 mg tablets) |
Pricing Trends
- Slight price erosion noted over the past 5 years
- Wholesale discounts and pharmacy benefit manager (PBM) contracts influence net prices
Price Projections
Assumptions for Forecasting
- Minimal change in regulatory status
- Continued decline in overall use due to alternative options
- No major patent expirations or new formulations
- Marginal market share shift from brand to compounded products
Projected Trends (Next 5 Years)
| Year | Estimated Annual Revenue | Commentary |
|---|---|---|
| 2023 | $20–$25 million | Current market size, slight decline due to competition |
| 2024–2025 | $15–$20 million | Market contraction continues, part of shifting preferences |
| 2026–2027 | $10–$15 million | Decline stabilizes as newer therapies gain acceptance |
Factors Impacting Price and Market
- Increased competition from non-estrogen options
- Potential for formulary exclusions affecting pharmacy stocking
- Possible confirmation of safety via ongoing studies; could stabilize demand
- Regulatory restrictions on compounded formulations may influence substitution trends
Regulatory and Policy Impact
- FDA regulations favor branded, approved products
- Legislation (e.g., Drug Price Transparency Act) pressures manufacturers to justify pricing
- Off-label compounded alternatives reduce demand, exerting downward pressure on prices
Key Takeaways
- The market for PREMARIN Vaginal Cream is declining, with a current U.S. revenue estimate of approximately $20–$25 million annually.
- Pricing remains relatively stable, with WAC around $30–$40 per applicator, but discounts and rebates likely lower net prices.
- Competition from other estrogen products and non-estrogen therapies continues to erode market share.
- Future revenue is projected to decline further, stabilizing near $10–$15 million over the next five years.
- Price pressure from regulatory changes and generic/compounded alternatives will influence the product's economic trajectory.
FAQs
1. How does PREMARIN Vaginal Cream compare to alternatives in efficacy?
Efficacy for symptom relief in atrophic vaginitis is similar across estrogen creams. No new head-to-head clinical trials have shown superiority, making choice dependent on physician preference, patient response, and formulary coverage.
2. What are the main factors affecting the product’s pricing?
Market competition, regulatory environment, formulary placement, and rebate strategies primarily influence pricing. Demand decline also pressures price stability.
3. Is there potential for price increases?
Limited potential exists due to decreasing demand, increased competition, and regulatory oversight, which favor price stabilization or slight decreases.
4. How do regulatory policies influence future market prospects?
FDA guidance encouraging safety and efficacy, alongside restrictions on compounded versions, may influence market share and pricing strategies.
5. What is the outlook for new formulations or delivery methods?
No current major pipeline exists. Future developments could either stabilize demand or accelerate decline depending on innovation adoption and regulatory approval.
Sources
[1] IQVIA. (2023). Market Dynamics of Vaginal Estrogen Products.
[2] HealthData Research. (2022). Postmenopausal Women Demographics.
[3] FDA. (2013). Guidance for Industry: Estrogen and Progestin Drug Products.
[4] Shionogi. (2023). Osphena Prescribing Data.
[5] Novartis and other manufacturers' financial reports.
More… ↓
