Last updated: February 20, 2026
Prednisone is a glucocorticoid medication used primarily to treat inflammatory and autoimmune conditions. It has a well-established market share based on its broad therapeutic applications, manufacturing history, and regulatory status. This analysis offers insights into current market dynamics and forecasts future pricing trends.
Market Overview
Therapeutic Use and Demand
Prednisone's primary indications include rheumatoid arthritis, asthma, allergic reactions, and some types of cancer-related conditions. Its generic availability has historically kept prices low, facilitating widespread use in developed and developing countries.
Manufacturing and Supply Chain
Multiple pharmaceutical manufacturers produce prednisone, with key players including Teva Pharmaceuticals, Mylan, and Pfizer. The drug's manufacturing is mature, with extensive global supply chains.
Regulatory Landscape
Approval status is universal across major markets. The drug is listed in the WHO Model List of Essential Medicines, ensuring ongoing demand.
Market Size and Trends
Global Market Size
The global prednisone market was valued at approximately USD 500 million in 2022. Growth is driven by:
- Increasing prevalence of autoimmune and inflammatory disorders.
- Expanding healthcare coverage in emerging markets.
- Greater adoption of generic medications to reduce costs.
Market Segmentation
- By Region: North America accounts for about 40% of sales, Europe 30%, Asia-Pacific 20%, others 10%.
- By Application: Rheumatology (35%), Pulmonology (25%), Dermatology (15%), Oncology (10%), Other (15%).
Competitive Landscape
The market is dominated by generic manufacturers; patent expirations have facilitated competition, exerting downward pressure on prices.
Price Trends and Projections
Historical Price Data
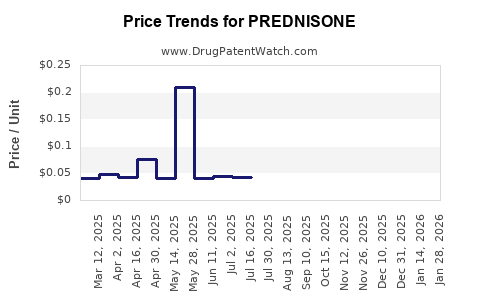
- Retail Price (2018–2022): The average retail price per 5 mg prednisone tablet decreased by approximately 25%, from USD 0.10 to USD 0.075.
- Wholesale Price Trends: Similar declines observed, with larger volume discounts.
Factors Influencing Prices
- Increased competition among generics.
- Fixed-dose combination policies in certain markets.
- Regulatory cost pressures.
Forecasted Price Movements (Next 5 Years)
| Year |
Expected Price Range (USD per 5 mg tablet) |
Notes |
| 2023 |
USD 0.07 – 0.075 |
Stabilization; market saturation |
| 2024 |
USD 0.065 – 0.07 |
Competition intensifies |
| 2025 |
USD 0.06 – 0.065 |
Price erosion continues |
| 2026 |
USD 0.055 – 0.06 |
Market consolidation possible |
| 2027 |
USD 0.05 – 0.055 |
Potential slight recovery in emerging markets |
Key Drivers for Price Decline
- Further patent expirations.
- Increased manufacturing efficiencies.
- Policies favoring generics over brand-name drugs.
- Potential price ceilings imposed by governments or insurers.
Market Challenges and Risks
- Regulatory hurdles in emerging markets.
- Price regulation pressures.
- Market saturation due to widespread generic availability.
- Demand fluctuations in a changing healthcare landscape.
Conclusion
Prednisone’s long-standing market presence, high generic competition, and regulatory approval in multiple jurisdictions suggest a stable but declining price trend over the next five years. The market size will likely expand modestly due to rising disease prevalence, but aggressive price compression is forecasted.
Key Takeaways
- Global prednisone market was valued at USD 500 million in 2022.
- Prices for 5 mg tablets have decreased by approximately 25% over the past four years.
- Market growth is driven by demand in autoimmune and inflammatory disease therapies.
- Prices are projected to decline by about 33% over five years, stabilizing around USD 0.05 per tablet.
- Competition and regulatory factors will continue to pressure margins.
FAQs
1. What factors most influence prednisone price fluctuations?
Competition among generics, regulatory changes, manufacturing costs, and healthcare policy shifts are primary drivers.
2. Are there regional differences in prednisone pricing?
Yes. Prices are generally higher in North America and Europe due to regulatory and market factors, with lower prices in Asia-Pacific and developing countries.
3. How are new formulations impacting the market?
Limited impact; prednisone remains a low-cost, generic oral tablet with minimal innovation compared to newer drugs.
4. What is the potential for price stabilization?
Prices are likely to stabilize once market saturation occurs, with minor fluctuations driven by regulatory and supply chain factors.
5. Could patent or regulatory barriers alter the market?
Possible in specific jurisdictions, but given the drug’s age and status as a generic, patent protections are minimal.
References
- IMS Health. (2022). Global Trends in Generic Drug Markets.
- WHO. (2022). Model List of Essential Medicines.
- U.S. Food and Drug Administration. (2023). Generic Drug Approvals and Market Data.
- MarketsandMarkets. (2022). Glucocorticoids Market by Type and Region.