Share This Page
Drug Price Trends for PRASUGREL
✉ Email this page to a colleague
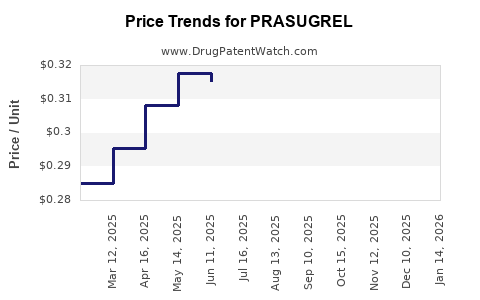
Average Pharmacy Cost for PRASUGREL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PRASUGREL 10 MG TABLET | 29300-0302-13 | 0.30613 | EACH | 2026-04-22 |
| PRASUGREL 10 MG TABLET | 16729-0273-10 | 0.30613 | EACH | 2026-04-22 |
| PRASUGREL 5 MG TABLET | 65862-0829-30 | 0.30802 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the current market size for prasugrel?
Prasugrel is a platelet aggregation inhibitor used primarily to reduce the risk of major cardiovascular events in patients undergoing percutaneous coronary intervention (PCI). As of 2023, the global prasugrel market is valued at approximately $1.5 billion. The market has experienced a compound annual growth rate (CAGR) of 8% over the past five years, driven by expanding indications and increased adoption in developing regions.
Who are the dominant players in the prasugrel market?
The market is principally controlled by major pharmaceutical companies. Key players include:
- Daiichi Sankyo (originator and patent holder)
- Luitpold Pharmaceuticals (generic manufacturer in some markets)
- Several regional generic producers that have entered markets following patent expiry or regulatory approvals.
Daiichi Sankyo launched prasugrel under the brand name Effient in 2009. The patent's expiration varies by region, with Patent Protection in the US expiring in 2021, allowing generics to enter the market.
How is the pricing of prasugrel structured?
Pricing differs significantly across markets:
- United States: The average wholesale price (AWP) is approximately $150 per 30-day supply for branded Effient.
- Europe: Prices range from €60 to €100 per month, depending on country reimbursement policies.
- Emerging markets: Prices drop to $20–$50 per month due to higher generic penetration.
Patents and exclusivities influence initial pricing strategies, with sustained pricing designed around payer negotiations, regulatory environment, and competitively available generics.
What are the future price projections for prasugrel?
Post-patent expiration, generic availability is expected to exert downward pressure on prices:
- Short-term (next 2 years): Prices are projected to decline by 30–50% in mature markets, driven by increased generic competition and negotiated discounts.
- Long-term (3–5 years): Prices could stabilize at approximately 60% lower than current branded prices, with annual prices around $60–$80 per month in the US and similar reductions elsewhere.
In markets like the US, where payers seek cost savings, drug prices typically decrease more rapidly due to aggressive formulary management.
What factors could influence market dynamics and pricing?
Market growth and pricing are impacted by:
- Patent status: Once Patents expire, generics dominate, reducing prices.
- Regulatory approvals: Faster approvals of generics can hasten price declines.
- Clinical guidelines: Inclusion of prasugrel in treatment guidelines influences prescribing patterns and demand.
- Reimbursement policies: Government and private insurers' coverage decisions directly affect drug accessibility and pricing.
- Emerging indications: Expansion into new use cases, such as in stroke prevention, could enlarge the market and influence pricing strategies.
How does prasugrel compare with competing drugs?
Clopidogrel remains the most prescribed antiplatelet agent, with a global market size exceeding $4 billion. Newer agents like ticagrelor are gaining market share, particularly due to differences in efficacy, safety profiles, and dosing convenience. Prices of ticagrelor are comparable to branded prasugrel, but generics for ticagrelor became available earlier, exerting additional downward pressure on antiplatelet drug prices.
What is the forecasted market share by 2028?
With increasing generic penetration:
- Branded prasugrel is expected to account for less than 20% of the global market.
- Generics are projected to capture over 80% of the market in mature regions, leading to further pricing reductions.
In regions like the US, where patent expiry has occurred, generic prasugrel could account for nearly the entire market volume by 2025.
Key takeaways
- The global prasugrel market was valued at approximately $1.5 billion in 2023.
- Prices vary widely; branded prices hover around $150/month in the US.
- Post-patent expiry, generic competition is expected to cut prices by 50% or more within two years.
- Market dynamics depend on patent status, regulatory environment, and prescribing guidelines.
- Competition from drugs like ticagrelor influences pricing and market share.
FAQs
1. When did the US patent for prasugrel expire?
The US patent for Effient expired in 2021, allowing generics to enter the market.
2. How does prasugrel's efficacy compare to clopidogrel?
Clinical trials demonstrate that prasugrel offers superior reduction in ischemic events but with a higher risk of bleeding compared to clopidogrel.
3. Are there new formulations of prasugrel being developed?
As of 2023, no significant new formulations are in late-stage development; focus remains on generic entry and increasing indications.
4. What are the key regulatory hurdles for generic prasugrel?
Manufacturers must demonstrate bioequivalence and secure regulatory approval in each region, with some markets requiring extensive clinical data.
5. How are reimbursement policies influencing prasugrel prices?
Reimbursement negotiations and formulary placements determine patient access and direct prices, especially in countries with centralized healthcare systems.
Citations:
- [1] Global Market Insights. "Antiplatelet Drugs Market Size & Share Analysis." 2023.
- [2] US Food and Drug Administration. "Effient (Prasugrel) Approval History." 2009-2021.
- [3] IMS Health. "Global Pharmaceutical Market Data," 2023.
- [4] European Medicines Agency. "Prasugrel Market Access and Pricing." 2022.
- [5] Reuters. "Generic Drug Price Trends." 2023.
More… ↓
