Share This Page
Drug Price Trends for POVIDONE-IODINE
✉ Email this page to a colleague
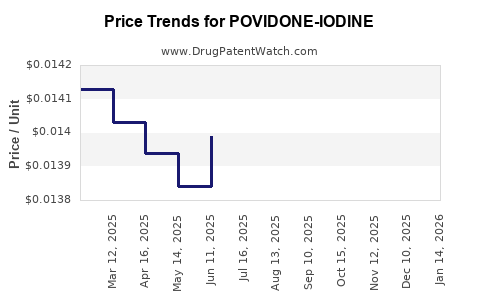
Average Pharmacy Cost for POVIDONE-IODINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| POVIDONE-IODINE 10% SOLUTION | 70000-0060-01 | 0.02025 | ML | 2026-04-22 |
| POVIDONE-IODINE 10% SOLUTION | 00904-1103-09 | 0.01518 | ML | 2026-04-22 |
| POVIDONE-IODINE 10% SOLUTION | 70000-0060-01 | 0.02047 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
POVIDONE-IODINE Market Analysis and Financial Projection
What Is the Global Market Size and Composition for Povidone-Iodine?
The global povidone-iodine market was valued at approximately $250 million in 2022. It is forecast to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030, reaching around $340 million by 2030. This growth stems from increasing demand for antiseptics in medical, surgical, and consumer hygiene markets.
Market segments include:
- Medical and surgical antiseptics: 60%
- Consumer antiseptic products (wound care, first aid): 30%
- Veterinary antiseptics: 10%
Key regions driving growth are North America, Europe, and parts of Asia-Pacific. North America accounts for about 40% of sales, driven by hospital infection control policies, while Asia-Pacific's rapid population growth and aging demographics expand demand.
What Are the Main Drivers of Market Growth?
-
Hospital Infection Prevention: Stringent infection control standards boost antiseptic use. The rise in surgeries and wound management elevates demand.
-
Consumer Awareness: Increased awareness about hygiene elevates retail sales including antiseptic solutions, wipes, and creams.
-
Regulatory Approvals: Approvals from agencies like the FDA, EMA contribute to market confidence and product launches.
-
Increased Use in Veterinary Medicine: Growing pet ownership correlates with higher demand for veterinary antiseptics.
-
COVID-19 Pandemic Impact: Heightened emphasis on disinfectants and antiseptics globally resulted in a demand spike, some of which persists.
What Are Key Product and Pricing Trends?
- Product Forms: Tinctures, solutions, ointments, and swab sticks constitute the primary formats.
- Pricing: The average retail price varies from $2 to $8 per bottle (100 mL), influenced by concentration, brand, and form.
- Market Entry: Generic manufacturers dominate, pushing prices downward. Branded products can command premium prices up to 20% higher.
Who Are Major Industry Players?
Leading companies include:
- Perrigo Company: Focuses on OTC antiseptics and wound care products.
- Reckitt Benckiser: Offers iodine-based antiseptics for consumer markets.
- Johnson & Johnson: Provides povidone-iodine solutions for surgical and wound care.
- Mundipharma: Supplies povidone-iodine swabs and ointments.
- Taylor Pharmaceuticals: Distributes iodine solutions globally.
Market share estimates suggest Perrigo and Reckitt collectively control approximately 55% of the global market.
What Are Price Projections for Povidone-Iodine?
Price projections assume a steady decline aligned with increased generic competition.
| Year | Estimated Average Price per 100 mL (USD) | Notable Factors |
|---|---|---|
| 2023 | $4.50 | Post-pandemic stabilization |
| 2025 | $4.00 | Increased commoditization |
| 2027 | $3.50 | Market saturation in developed economies |
| 2030 | $3.00 | Broader adoption in emerging markets |
The downward trend results from technological advancements lowering manufacturing costs and expanding generic options.
What Are Potential Market Risks and Challenges?
- Regulatory Risks: Stringent standards and approval processes could delay new product launches or restrict formulations.
- Supply Chain Disruptions: Raw material shortages or geopolitical issues affect iodine sourcing and manufacturing.
- Price Pressure: Competition from generics may suppress prices.
- Market Saturation: Mature markets may see limited growth, especially in developed regions.
What Is the Competitive Landscape?
The competitive landscape is highly fragmented with multiple regional players. Innovation is limited due to the mature nature of antiseptic formulations, but companies seek to differentiate via packaging, branding, and extending product lines into combined antiseptics or wound-healing products.
How Do Regulatory and Patent Policies Affect Market Dynamics?
- Regulatory Approvals: Require compliance with local agencies such as the FDA, EMA, and China’s NMPA. Approval timelines can extend 6-12 months.
- Patent Landscape: Key formulations are off-patent, enabling widespread generic manufacturing. Certain formulations or delivery devices may still be under patent, restricting entry.
Key Takeaways
- The povidone-iodine market is valued at approximately $250 million (2022) and expected to reach $340 million by 2030.
- Growth is driven primarily by infection control protocols, consumer hygiene awareness, and COVID-19 relevance.
- Pricing trends show a decline, with prices per 100 mL dropping from about $4.50 in 2023 to an estimated $3.00 in 2030.
- Market penetration is constrained by regulatory hurdles, supply chain risks, and competition.
- Major players include Perrigo, Reckitt, Johnson & Johnson, and Mundipharma, with general market fragmentation.
FAQs
Q1: Which regions will see the fastest growth in povidone-iodine demand?
Asia-Pacific and Latin America will experience faster growth due to rising healthcare infrastructure, increased healthcare spending, and expanding consumer markets.
Q2: How does the rise of alternative antiseptics impact povidone-iodine?
Substitutes such as chlorhexidine and alcohol-based products challenge povidone-iodine in some applications, particularly due to differing spectrum of activity, cost, and user preference.
Q3: Are there emerging formulations or delivery methods?
Yes, new formulations include rapid-dissolving tablets, controlled-release topical patches, and combined antiseptic-antibiotic products, aimed at improving compliance and efficacy.
Q4: What are the patent considerations for new entrants?
Most basic povidone-iodine formulations are generic, but innovations in delivery systems or specific formulations may still be patent-protected, offering entry points for differentiation.
Q5: How do regulatory differences across markets influence market access?
Approval times and requirements differ; for instance, the FDA mandates extensive clinical data, leading to longer approval periods compared to some emerging markets with more streamlined processes.
Sources:
- Market Research Future, “Global Povidone-Iodine Market Forecast,” 2022.
- Grand View Research, “Antiseptic Market Analysis,” 2023.
- FDA and EMA product approval databases.
- Company financial reports and product catalogs.
More… ↓
