Share This Page
Drug Price Trends for POTASSIUM CL ER
✉ Email this page to a colleague
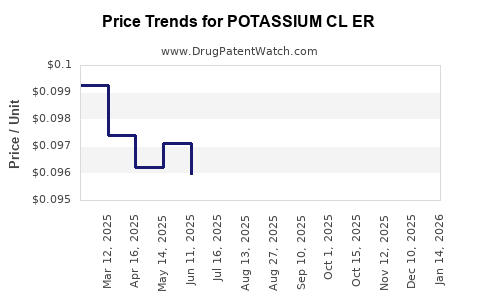
Average Pharmacy Cost for POTASSIUM CL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| POTASSIUM CL ER 20 MEQ TABLET | 72888-0076-05 | 0.16490 | EACH | 2026-04-22 |
| POTASSIUM CL ER 20 MEQ TABLET | 72888-0076-01 | 0.16490 | EACH | 2026-04-22 |
| POTASSIUM CL ER 10 MEQ TABLET | 72888-0075-05 | 0.08864 | EACH | 2026-04-22 |
| POTASSIUM CL ER 10 MEQ TABLET | 72888-0075-01 | 0.08864 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is the Market Scope for Potassium Chloride ER?
Potassium chloride extended-release (ER) is primarily used for potassium supplementation in patients with hypokalemia, chronic kidney disease, and certain cardiovascular conditions. Market demand correlates with the prevalence of these conditions and the adoption of potassium management protocols.
The global potassium chloride market was valued at approximately $3.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 5.8% from 2023 to 2028, driven by increasing chronic disease incidence and aging populations.
Key markets include:
- North America: leading due to high chronic kidney disease prevalence.
- Europe: stable growth, driven by aging demographics.
- Asia-Pacific: fastest growth, due to expanding healthcare infrastructure.
What Are the Regulatory and Patent Statuses?
Potassium chloride ER products are generally off-patent in major markets. Several formulations are marketed as generics, which limits exclusivity and affects pricing strategies.
In the US, the FDA has approved multiple potassium chloride ER products, including K-Tab ER and slow-release formulations by various manufacturers. No recent patent protections are in effect for the primary formulations, although some proprietary delivery mechanisms could be under patent.
How Are Pricing Trends Shaping Up?
Pricing for potassium chloride ER remains competitive with notable variations due to brand versus generic, formulation differences, and distribution channels.
- Generic Prices: Typically range from $0.10 to $0.25 per 10 mEq tablet.
- Brand-Name Products: Can reach up to $0.50 per 10 mEq tablet.
- Market Average: About $0.15 per tablet in the US, with minor fluctuations based on pharmacy and insurance terms.
Pricing pressures caused by generics have pushed margins downward, encouraging manufacturers to differentiate products via delivery mechanisms or extended-release technologies.
What Factors Influence Future Price Trajectories?
1. Patent Expirations and Generic Competition
The expiration of key patents in the late 2010s increased generic availability, inducing price compression. Future patent filings, if any, are unlikely to significantly alter the market unless linked to novel formulations offering improved tolerability or efficacy.
2. Development of Novel Formulations
Innovations like sustained-release delivery mechanisms that reduce gastrointestinal irritation could command premium prices, but such products require regulatory approval and clinical validation.
3. Regulatory Policies and Reimbursement
Pricing strategies are sensitive to reimbursement policies, especially in markets with government-controlled drug pricing, like Canada and parts of Europe. Policy shifts favoring biosimilars and generics can depress prices further.
4. Market Demand and Disease Trends
An aging global population and rising chronic disease rates sustain demand. Increases in conditions like heart failure may boost usage, though this effect might be mitigated by newer treatment guidelines favoring alternative therapies or combination treatments.
5. Supply Chain Disruptions
Raw material availability and manufacturing capacity can impact pricing. Recent supply chain issues in active pharmaceutical ingredient (API) sourcing have caused temporary price spikes for some generic medicines, including potassium chloride.
Competitive Landscape and Price Projection
Major players include Mylan, Teva, and Hikma Pharmaceuticals. The market exhibits high fragmentation, with over 50 generic manufacturers globally.
Projected price trend (2023-2028):
- Slight decline in tablet prices due to competition.
- Minimal chance of significant price increases unless novel formulations or delivery methods enter the market.
- Expected to stabilize around $0.10–$0.20 per 10 mEq tablet in mature markets.
Conclusion
Potassium chloride ER faces a mature, highly competitive market with limited potential for price increases. The primary drivers of future pricing are innovation and regulatory changes. Current prices are influenced heavily by generic competition, with future trends likely to remain within a narrow range unless disruptive technologies emerge.
Key Takeaways
- The global market is valued at over $3 billion, with steady growth driven by aging populations and chronic disease prevalence.
- Most formulations are off-patent, leading to significant generic competition and downward price pressure.
- Innovations in delivery mechanisms, if approved, could shift pricing dynamics.
- Regulatory policies and supply chain factors significantly influence prices.
- Future prices are expected to stabilize around current generic levels unless novel technologies or formulations are introduced.
FAQs
1. Are there any patents still in effect for potassium chloride ER?
Most patents have expired; ongoing patents are limited to specific delivery mechanisms or formulations and are unlikely to significantly impact prices.
2. How does the use of potassium chloride ER differ between countries?
Usage rates are higher in countries with greater prevalence of chronic kidney disease and heart conditions. Regulatory approvals and reimbursement policies also influence prescribing patterns.
3. Will the emergence of biosimilars affect potassium chloride ER pricing?
Biosimilars are not applicable to potassium chloride ER, which is a small-molecule drug. However, generic competition already impacts pricing.
4. What innovations could impact future market dynamics?
Proprietary extended-release formulations with improved safety or tolerability profiles could command premium pricing, provided they gain regulatory approval.
5. How do supply chain issues impact prices?
Disruptions in raw material sourcing or manufacturing capacity can temporarily increase prices but are unlikely to cause long-term shifts unless pervasive.
Sources:
- Market Research Future, "Potassium Chloride Market Analysis," 2023.
- US Food and Drug Administration, "Approved Potassium Chloride Products," 2022.
- IQVIA Institute, "Global Pharmaceutical Market Trends," 2022.
- EvaluatePharma, "Generic Drug Pricing Trends," 2022.
- Statista, "Chronic Disease Prevalence Worldwide," 2023.
More… ↓
