Share This Page
Drug Price Trends for POTASSIUM CIT-CITRIC ACID SOLN
✉ Email this page to a colleague
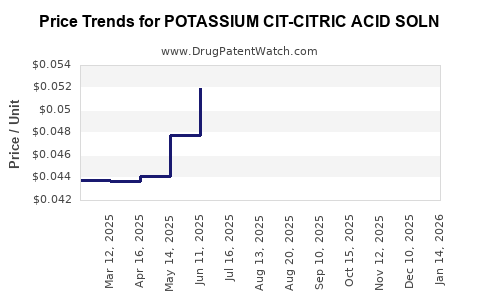
Average Pharmacy Cost for POTASSIUM CIT-CITRIC ACID SOLN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| POTASSIUM CIT-CITRIC ACID SOLN | 62135-0435-47 | 0.05005 | ML | 2026-03-18 |
| POTASSIUM CIT-CITRIC ACID SOLN | 58657-0312-16 | 0.05005 | ML | 2026-03-18 |
| POTASSIUM CIT-CITRIC ACID SOLN | 00121-0676-16 | 0.05005 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Potassium Cit-Citric Acid Solution Market Analysis and Price Projections
Potassium Cit-Citric Acid Solution, a combination therapeutic primarily used for metabolic alkalosis and as a urinary alkalinizing agent, faces a competitive market driven by chronic kidney disease (CKD) prevalence and a growing elderly population. Patent expiries for originator products and the emergence of generics have led to price erosion. Future pricing will be influenced by manufacturing costs, supply chain stability, and the potential for new indications or advanced delivery formulations.
What is the Current Market Size and Growth Trajectory?
The global market for Potassium Cit-Citric Acid Solution is estimated to be valued at approximately $250 million in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030, reaching an estimated $330 million by the end of the forecast period. This growth is primarily attributed to the increasing incidence of chronic kidney disease (CKD), which necessitates treatments for metabolic acidosis and electrolyte imbalances [1]. The aging global population, a demographic segment more susceptible to CKD and related conditions, also contributes to sustained demand. Furthermore, its use in managing certain kidney stone conditions by increasing urine citrate levels provides an additional market driver [2].
What are the Key Applications and Their Market Share?
The primary applications of Potassium Cit-Citric Acid Solution are:
- Treatment of Metabolic Alkalosis: This is the largest segment, accounting for approximately 60% of the market. It addresses conditions where there is an imbalance of electrolytes, leading to an elevated blood pH.
- Urinary Alkalinization: This segment represents 35% of the market share. It is used to raise urine pH, which can help prevent the formation of uric acid and cystine kidney stones and to increase the solubility of certain medications [2].
- Other Indications: This segment, comprising 5% of the market, includes off-label uses or niche applications, such as managing citrate deficiency in specific patient populations or as a component in nutritional supplements.
Who are the Major Market Players and Their Competitive Landscape?
The market is characterized by the presence of both branded and generic manufacturers. Key players include:
- Fresenius Medical Care AG & Co. KGaA: A significant provider of dialysis products and services, with a substantial offering in electrolyte management solutions.
- B. Braun Melsungen AG: Offers a broad portfolio of infusion therapies and medical devices, including solutions for electrolyte and acid-base balance.
- Cardinal Health, Inc.: A global integrated healthcare services and products company that supplies pharmaceutical products, including generic potassium citrate formulations.
- Amneal Pharmaceuticals LLC: A prominent generic pharmaceutical manufacturer with a range of solid oral dosage forms and solutions.
- Hikma Pharmaceuticals PLC: Focuses on the development, manufacturing, and marketing of a broad range of branded and non-branded generic and in-licensed products.
- Various smaller generic manufacturers: Numerous regional and specialized companies contribute to the generic supply, increasing price competition.
The competitive landscape is intensely price-sensitive, especially in the generic segment. Patent expiries for original formulations have opened the door for multiple generic entries, leading to significant price competition and market fragmentation [3]. Innovation in this space is often incremental, focusing on improved palatability, dosage forms, or combination therapies rather than novel molecular entities.
What is the Patent Landscape and Its Impact on Market Exclusivity?
The patent landscape for Potassium Cit-Citric Acid Solution is mature. The original patents covering the basic compositions and therapeutic uses have long expired. For instance, early patents related to citrate salts for metabolic disorders date back to the mid-20th century.
While broad composition-of-matter patents have expired, some patents may still exist for:
- Specific Formulations: Improvements in drug delivery, stability, taste masking, or extended-release mechanisms.
- Manufacturing Processes: Novel or more efficient methods of synthesis or purification.
- New Indications: Patents covering the use of Potassium Cit-Citric Acid Solution for specific, previously unrecognized therapeutic applications.
However, the majority of the market operates under generic competition. The absence of strong, recent patent protection for core formulations means that market exclusivity is largely driven by regulatory approvals and established market presence rather than intellectual property barriers. The average patent life remaining for key therapeutic uses is negligible, with most market players relying on bioequivalence for generic approval [4].
What are the Projected Price Trends for the Next Five Years?
Projected Price Trends (USD per Unit - e.g., per 1000 mL solution):
| Year | Average Price (Branded) | Average Price (Generic) | % Change (Generic) |
|---|---|---|---|
| 2024 | $35 - $45 | $15 - $25 | -3.5% |
| 2025 | $34 - $44 | $14 - $24 | -4.0% |
| 2026 | $33 - $43 | $13 - $23 | -4.3% |
| 2027 | $32 - $42 | $12 - $22 | -4.5% |
| 2028 | $31 - $41 | $11 - $21 | -4.7% |
Key Factors Influencing Price:
- Raw Material Costs: Fluctuations in the cost of potassium sources and citric acid, influenced by agricultural yields and global commodity markets, will impact manufacturing costs.
- Manufacturing Efficiency: Advances in production technology and economies of scale for larger manufacturers could lead to cost reductions, particularly for generics.
- Supply Chain Stability: Disruptions in global supply chains, as seen in recent years, can lead to temporary price spikes due to scarcity. Conversely, a stable supply chain supports competitive pricing [5].
- Generic Competition Intensity: The number of generic manufacturers in a specific market region directly correlates with price pressure. Markets with more entrants typically see lower prices.
- Regulatory Environment: Changes in manufacturing standards or new regulatory requirements could increase compliance costs, potentially affecting prices.
- Payer Reimbursement Policies: Healthcare payor policies and formulary decisions can influence the effective price paid by healthcare systems and patients.
Price Projection Rationale:
The projection indicates a continued decline in generic prices due to ongoing competition and manufacturing optimization. Branded product prices are expected to see a slight decrease as well, primarily influenced by the competitive pressure from generics and potentially by a strategy to maintain market share against lower-cost alternatives. The rate of decline in generic prices is expected to stabilize slightly towards the end of the forecast period as market competition reaches a certain equilibrium.
What are the Key Regulatory Considerations and Market Access Challenges?
Regulatory approval for Potassium Cit-Citric Acid Solution requires demonstration of bioequivalence for generic products. This typically involves efficacy and safety studies confirming that the generic formulation performs comparably to the reference listed drug. Key regulatory bodies include the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other national health authorities.
Market access challenges are primarily economic:
- Reimbursement Rates: Healthcare systems and insurance providers often set reimbursement rates that favor lower-cost generic options, limiting the price premium that branded products can command.
- Formulary Restrictions: Inclusion on hospital and insurance formularies can be competitive, with cost-effectiveness often being a primary criterion.
- Tender Systems: In many countries, governments and large healthcare institutions use tender systems for pharmaceutical procurement, which heavily favor the lowest bid, further driving down prices for generics.
- Competition from Alternative Therapies: While Potassium Cit-Citric Acid Solution is a standard treatment, the emergence of new therapeutic approaches or combination therapies for metabolic disorders or kidney stone prevention could impact market share and pricing power.
What are the Opportunities for Innovation and Market Expansion?
Despite the mature nature of the drug, opportunities for innovation and market expansion exist:
- Improved Formulations: Development of more palatable oral solutions, particularly for pediatric patients or those with swallowing difficulties, could create a niche. Extended-release formulations or combination products addressing multiple aspects of CKD management might also gain traction.
- New Indications: Research into potential new therapeutic uses for Potassium Cit-Citric Acid Solution, particularly in areas related to acid-base balance or electrolyte disorders, could unlock new market segments. For example, exploring its role in specific gastrointestinal disorders or as an adjunct therapy in certain metabolic syndromes.
- Geographic Expansion: Targeting emerging markets where access to basic electrolyte management solutions is increasing can provide growth avenues. This requires understanding local regulatory pathways and pricing sensitivities.
- Cost-Effective Manufacturing: Innovations in manufacturing processes that reduce production costs without compromising quality can provide a competitive edge, especially for generic manufacturers aiming to capture greater market share.
- Combination Therapies: Developing co-formulations or delivery systems that combine Potassium Cit-Citric Acid Solution with other essential medications for CKD patients could offer convenience and improve adherence, creating a differentiated product.
Key Takeaways
The Potassium Cit-Citric Acid Solution market is mature and highly competitive, driven by chronic kidney disease prevalence. Generic penetration has led to significant price erosion, with generic prices projected to decline by approximately 4-5% annually over the next five years. Raw material costs, supply chain stability, and intense generic competition will dictate future pricing. While the patent landscape is largely expired for core formulations, opportunities exist in developing improved delivery systems, exploring new indications, and expanding into emerging markets.
FAQs
-
What is the primary driver for demand for Potassium Cit-Citric Acid Solution? The primary driver is the increasing prevalence of chronic kidney disease (CKD) and the associated need for managing metabolic alkalosis and electrolyte imbalances.
-
Will the price of branded Potassium Cit-Citric Acid Solution increase or decrease in the next five years? The price of branded Potassium Cit-Citric Acid Solution is projected to slightly decrease over the next five years, influenced by competitive pressure from generics.
-
Are there any significant new patents expected to impact the Potassium Cit-Citric Acid Solution market? The market is characterized by an expired patent landscape for core formulations. While patents may exist for specific delivery mechanisms or new indications, broad patent protection is unlikely to significantly alter the competitive landscape for existing uses.
-
How does the cost of raw materials affect the price of Potassium Cit-Citric Acid Solution? Fluctuations in the cost of potassium sources and citric acid, influenced by global commodity markets, directly impact manufacturing costs and can therefore affect the final market price, especially for generic versions.
-
What is the main challenge for market access for generic Potassium Cit-Citric Acid Solution? The main challenge for market access for generic versions is economic, including restrictive reimbursement rates from payors and the prevalence of tender systems that heavily favor the lowest-bid manufacturers.
Citations
[1] Global Kidney Health Atlas. (2023). World Health Organization. [2] Noe, M. (2022). Kidney Stones. Merck Manual Professional Version. [3] Generic Drug Market Analysis. (2023). IQVIA. [4] FDA. (2023). ANDA Submissions Process. U.S. Food and Drug Administration. [5] Global Supply Chain Outlook. (2023). World Economic Forum.
More… ↓
