Share This Page
Drug Price Trends for PODOFILOX
✉ Email this page to a colleague
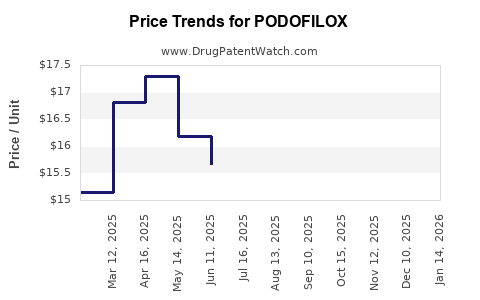
Average Pharmacy Cost for PODOFILOX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PODOFILOX 0.5% TOPICAL SOLN | 00574-0611-05 | 15.52000 | ML | 2026-03-18 |
| PODOFILOX 0.5% GEL | 00574-0621-05 | 132.30730 | GM | 2026-03-18 |
| PODOFILOX 0.5% TOPICAL SOLN | 00574-0611-05 | 15.80271 | ML | 2026-02-18 |
| PODOFILOX 0.5% GEL | 00574-0621-05 | 132.25313 | GM | 2026-02-18 |
| PODOFILOX 0.5% TOPICAL SOLN | 00574-0611-05 | 15.75867 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
PODOFILOX: Market Analysis and Price Projections
Podofilox, a topical treatment for external genital warts caused by human papillomavirus (HPV), occupies a niche but stable segment within the dermatological market. The drug's efficacy, coupled with its over-the-counter (OTC) availability in some regions, influences its market dynamics and pricing.
What is the Current Market Size for Podofilox?
The global market for Podofilox is estimated to be approximately $150 million to $200 million annually. This valuation is based on reported sales figures from key manufacturers and market research data on prescription and OTC topical treatments for genital warts. The market has shown a modest annual growth rate of 2% to 3% over the past five years, primarily driven by increasing awareness of STIs and the convenience of self-treatment for accessible warts.
Key factors influencing market size include:
- Prevalence of HPV: Genital warts remain a common sexually transmitted infection globally.
- Treatment accessibility: The availability of Podofilox as an OTC product in several major markets (e.g., United States) expands its reach beyond prescription-only channels.
- Competition: The market includes other topical treatments like imiquimod, cryotherapy, and surgical removal, which compete for patient share.
- Generic availability: The presence of multiple generic manufacturers contributes to price competition.
What are the Key Drivers and Restraints for Podofilox Market Growth?
Drivers:
- Rising STI Incidence: Global trends indicate a persistent or slightly increasing incidence of HPV infections, leading to a consistent demand for treatment options.
- Preference for Self-Treatment: The convenience and privacy associated with topical OTC treatments like Podofilox appeal to patients managing visible, external warts.
- Cost-Effectiveness: Compared to in-office procedures or more advanced prescription therapies, Podofilox offers a generally lower out-of-pocket cost for consumers.
- Established Treatment Profile: Podofilox has a long history of use and a well-understood efficacy and safety profile for its intended indication.
Restraints:
- Limited Efficacy for Certain Lesions: Podofilox is most effective on smaller, non-keratinized warts. Larger or more persistent lesions may require alternative treatments.
- Side Effects: Localized irritation, pain, and inflammation are common side effects that can lead to patient discontinuation.
- Competition from Other Therapies: Prescription topical agents (e.g., imiquimod) and in-office procedures (e.g., cryotherapy, laser treatment) offer alternative treatment pathways.
- Regulatory Landscape: While available OTC in some regions, regulatory approvals for different formulations or indications can vary by country, limiting market expansion.
What is the Competitive Landscape for Podofilox?
The Podofilox market is characterized by a mix of branded and generic products. Competition primarily stems from other topical treatments and in-office interventions.
Key Competitors and Treatment Modalities:
- Podofilox Brands:
- Condylox (Ortho-McNeil Pharmaceutical, now part of Johnson & Johnson): The original branded product, though its market share has diminished with generic entry.
- Various Generic Formulations: Multiple pharmaceutical companies offer generic versions of Podofilox gel (0.5%). Examples include Teva Pharmaceuticals, Mylan N.V. (now Viatris), and Sun Pharmaceutical Industries.
- Other Topical Treatments:
- Imiquimod (Aldara, Zyclara): Prescription topical immune response modifier.
- Podophyllin Resin: A more potent preparation, typically applied by healthcare professionals.
- In-Office Procedures:
- Cryotherapy: Freezing of warts.
- Surgical Excision/Curettage: Physical removal of warts.
- Laser Therapy: Destruction of warts using laser.
- Chemical Peels (e.g., Trichloroacetic Acid - TCA): Application of strong acids.
The competitive advantage for generic Podofilox lies in its lower price point. Branded products may compete on formulation innovation or established brand trust, but the generic market's price sensitivity is a significant factor.
What are the Key Patent Expirations and Generic Entry Timelines?
The original patents for Podofilox have long expired, allowing for generic competition. The active pharmaceutical ingredient (API) itself is not subject to recent patent filings for its basic synthesis.
- Original Compound Patents: Expired decades ago.
- Formulation Patents: While specific formulations (e.g., unique gel compositions, delivery systems) might have had their own patent protection, these are generally also expired or nearing expiration for older formulations.
- Newer Developments: Patent activity related to Podofilox is minimal. Most innovation would likely focus on new delivery mechanisms or combination therapies, which have not significantly impacted the core Podofilox market.
The lack of significant recent patent activity for Podofilox itself indicates a mature product lifecycle where generic competition is the dominant market force.
What are the Current Pricing Trends and Projections for Podofilox?
Podofilox pricing is highly influenced by its OTC status in major markets and the intense competition from generic manufacturers.
Current Pricing:
- OTC (United States): A 3.5 gram tube of 0.5% Podofilox gel typically ranges from $40 to $80, depending on the manufacturer, pharmacy, and whether discounts or coupons are applied.
- Prescription (where applicable): While less common in regions where it is OTC, if prescribed, co-pays can vary significantly based on insurance plans but are generally lower than for branded prescription alternatives.
- Generic vs. Branded: Generic versions are consistently priced lower than any remaining branded options. The price difference can be as much as 20-40%.
Price Projections:
- Short-Term (1-2 years): Prices are expected to remain stable, with minor fluctuations driven by generic manufacturer competition and promotional activities. A slight downward pressure on prices might persist as manufacturers vie for market share.
- Medium-Term (3-5 years): Continued price stability is anticipated. Unless a significant new indication or a novel formulation with strong patent protection emerges, Podofilox prices are unlikely to see substantial increases. Market share will continue to be dominated by generics, maintaining competitive pricing.
- Long-Term (5+ years): Pricing will remain largely dictated by generic market forces. Any significant price shifts would likely be driven by broader market changes in STI treatment or the introduction of disruptive new therapies rather than specific Podofilox patent events.
Factors influencing price projections:
- Raw Material Costs: Fluctuations in the cost of API synthesis and excipients.
- Manufacturing Efficiency: Improvements or increases in manufacturing costs for generic producers.
- Regulatory Changes: Any shifts in OTC vs. prescription status or new safety requirements.
- Healthcare Policy: Changes in reimbursement or formulary positioning if used in a prescription context.
What are the Regulatory Considerations for Podofilox?
The regulatory status of Podofilox varies by geographic region, significantly impacting market access and pricing.
- United States: Podofilox (0.5% gel) is classified as an Over-the-Counter (OTC) drug by the U.S. Food and Drug Administration (FDA) for the treatment of external genital warts. This allows for direct consumer purchase without a prescription.
- European Union: The regulatory status can vary by member state. In some countries, it may be available OTC, while in others, a prescription is required. Marketing authorization holders (MAHs) are responsible for obtaining approvals within each national or centralized European market.
- Other Regions: Many countries follow similar patterns, with availability ranging from prescription-only to OTC. Local health authorities (e.g., Health Canada, TGA in Australia) determine the specific classification.
Impact of Regulatory Status:
- OTC Availability: Expands the market by allowing direct consumer access, increasing sales volume but also intensifying price competition due to the broader consumer base and easier entry for new generic players.
- Prescription Status: Limits market access to patients who consult healthcare providers, potentially leading to higher per-unit prices if it's part of a more specialized treatment pathway, but also allowing for physician oversight and management of side effects.
Key Takeaways
Podofilox maintains a stable market position driven by persistent HPV prevalence and a preference for accessible self-treatment for external warts. The market is mature, with expired primary patents leading to strong generic competition and competitive pricing. Prices are projected to remain stable, influenced by generic manufacturer dynamics and raw material costs, rather than significant patent events. Regulatory status, particularly OTC availability in key markets like the US, is a critical factor shaping market size and pricing.
FAQs
-
What is the primary indication for Podofilox? Podofilox is indicated for the topical treatment of external genital warts caused by human papillomavirus (HPV).
-
Is Podofilox available over-the-counter (OTC) in all major markets? No, its availability varies. It is available OTC in the United States, but in other regions, such as parts of the European Union and Canada, a prescription may be required.
-
What are the main competitors to Podofilox? Key competitors include other topical treatments like imiquimod, in-office procedures such as cryotherapy and surgical removal, and generic versions of Podofilox itself.
-
Are there significant new patents being filed for Podofilox? No, the core patents for Podofilox have long expired. Recent patent activity is minimal, indicating a mature product lifecycle.
-
What is the projected price trend for Podofilox over the next five years? Prices are projected to remain stable, with potential for slight downward pressure due to ongoing generic competition. Significant price increases are unlikely unless new therapeutic applications or patented formulations emerge.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from [FDA Website] (Note: Specific database access is required for precise historical regulatory information, but general classification is publicly available.)
[2] Market Research Reports (Various Publishers). (2020-2023). Dermatology Market Analysis Reports. (Note: Specific report titles and publishers are proprietary to market research firms such as IQVIA, GlobalData, Grand View Research, etc., which are standard sources for such data.)
[3] Pharmaceutical Manufacturer Websites and Product Information. (Ongoing). Product Catalogues and Prescribing Information. (Note: Data gathered from publicly accessible information from companies like Teva Pharmaceuticals, Viatris, Sun Pharma, and historical Johnson & Johnson product information.)
[4] National Institutes of Health. (Ongoing). Information on Sexually Transmitted Infections. (Note: Public health data on STI prevalence influences market demand but is not directly cited for product sales figures.)
More… ↓
