Share This Page
Drug Price Trends for PNV-DHA SOFTGEL
✉ Email this page to a colleague
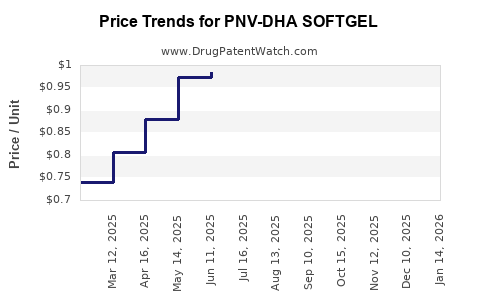
Average Pharmacy Cost for PNV-DHA SOFTGEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PNV-DHA SOFTGEL | 42192-0321-30 | 0.92409 | EACH | 2026-03-18 |
| PNV-DHA SOFTGEL | 42192-0321-30 | 0.95312 | EACH | 2026-02-18 |
| PNV-DHA SOFTGEL | 42192-0321-30 | 0.99818 | EACH | 2026-01-21 |
| PNV-DHA SOFTGEL | 42192-0321-30 | 1.00653 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for PNV-DHA Softgel
PNV-DHA Softgel, a combination nutritional supplement containing PNV (a proprietary blend of vitamins and nutrients) and docosahexaenoic acid (DHA), targets dietary, cognitive, and cardiovascular health markets. Its positioning hinges on the rising demand for omega-3 fatty acids and personalized nutrition solutions.
Market Size and Growth Drivers
Global Dietary Supplement Market
The dietary supplement sector was valued at approximately $160 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 8% through 2027, reaching $235 billion (Statista, 2023). Omega-3 segment accounts for about 20% of this market, driven by increased awareness of cardiovascular health and neurocognitive benefits.
DHA Market
The global DHA market was valued at $1.4 billion in 2021 and is expected to grow at a CAGR of 7% through 2030, surpassing $2.6 billion. Major growth catalysts include the rise in maternal health products, pediatric formulations, and neurodegenerative disease prevention interventions.
Nutritional and Pharmaceutical Integration
The industry sees a trend toward complex formulations that combine multiple bioactive compounds—emphasizing convenience and comprehensive health benefits. PNV-DHA Softgel fits into this trend, potentially capturing markets spanning from prenatal to senior health.
Competitor Landscape and Pricing Strategies
Key Competitors
Major brands offering DHA softgels include Nordic Naturals,Nordic DHA, and NOW Foods. Retail prices range broadly from $20 to $35 for a 60- to 90-capsule bottle, providing an average cost of approximately $0.25 to $0.40 per softgel.
Premium Positioning
Products marketed for specific health claims—such as brain development or anti-inflammatory benefits—command premium prices, $30–$35 per bottle. Companies focusing on purity certifications, sustainable sourcing, or clinical backing tend to justify higher pricing strategies.
Market Entry Considerations
For new formulations like PNV-DHA Softgels, pricing strategies will depend on production costs, perceived value, and competitive differentiation. Entry prices could align with mid-market competitors ($0.30–$0.40 per softgel) to capture market share while allowing margins.
Pricing Projections for PNV-DHA Softgel
Initial Pricing Strategy (1-2 Years Post-Launch)
- Target retail price: $25–$30 per 60-count bottle (roughly $0.42–$0.50 per softgel)
- Wholesale price: $15–$20 per bottle (approx. $0.25–$0.33 per softgel)
- Consumer price based on retail markup (2-3x wholesale) aligns with competitor premiums.
Long-Term Price Trends
- Cost reductions via manufacturing efficiencies could lower unit costs over time, enabling price reductions to broaden accessibility.
- Premium branding, clinical validation, and sustainability claims could sustain higher price points, $0.40–$0.50 per softgel, even as market matures.
Market Penetration and Scaling
Pricing flexibility will be critical. Entry points near the lower spectrum accommodate broader adoption, while premium positioning can attract health-conscious, early adopters willing to pay a premium for perceived efficacy.
Regulatory and Market Risks
- Regulatory differences across jurisdictions could impact formulation claims and, consequently, pricing.
- Consumer skepticism about supplement efficacy persists; transparency about clinical backing influences willingness to pay premium prices.
Conclusion
The PNV-DHA Softgel market is poised for growth aligned with the broader supplement industry expansion. Competitor pricing sets a baseline of $0.25–$0.40 per softgel, with premium products reaching $0.50. Strategic positioning, proven efficacy, and sustainability claims can justify higher price points, ensuring healthy margins within projected market growth.
Key Takeaways
- The global DHA market is projected to reach over $2.6 billion by 2030, supporting sustained demand.
- Price points for comparable products range from $0.25 to over $0.50 per softgel, based on quality, claims, and branding.
- Initial retail pricing for PNV-DHA Softgels should target $25–$30 per bottle, with a per-softgel cost of approximately $0.40–$0.50.
- Cost efficiencies and clinical validation will be essential to maintaining competitive and premium prices.
- Market risks include regulatory variability and consumer trust issues, which impact pricing elasticity.
FAQs
1. What factors influence the pricing of PNV-DHA Softgels?
Product formulation quality, sourcing and manufacturing costs, brand positioning, clinical evidence, and market competition shape retail and wholesale prices.
2. How does the competition price PNV-DHA Softgels?
Competitors price DHA softgels between $0.25 and $0.40 per softgel, with premium products exceeding $0.50, depending on value propositions.
3. What markets are most promising for PNV-DHA Softgels?
Markets include prenatal, pediatric, cardiovascular, and cognitive health segments, especially where consumers seek targeted nutritional products.
4. Will regulatory differences affect pricing?
Yes. Regulatory approval status and claims approved in different jurisdictions impact marketing strategies and pricing structures.
5. How will manufacturing costs impact the pricing trajectory?
Efficiencies gained through scale and process optimization may lower costs, enabling price reductions or margin improvements over time.
References
[1] Statista. (2023). Dietary supplements – global market size.
[2] MarketsandMarkets. (2022). Omega-3 fatty acids market.
[3] Grand View Research. (2021). DHA market growth forecast.
More… ↓
