Share This Page
Drug Price Trends for PIOGLITAZONE-GLIMEPIRIDE
✉ Email this page to a colleague
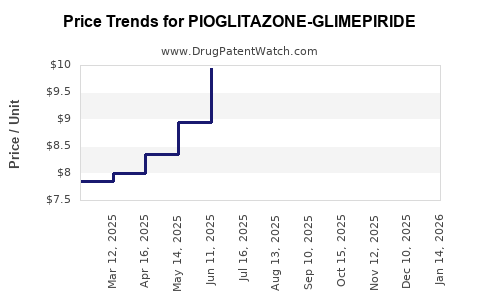
Average Pharmacy Cost for PIOGLITAZONE-GLIMEPIRIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PIOGLITAZONE-GLIMEPIRIDE 30-2 | 66993-0821-30 | 10.80087 | EACH | 2026-02-18 |
| PIOGLITAZONE-GLIMEPIRIDE 30-4 | 66993-0822-30 | 11.27016 | EACH | 2026-02-18 |
| PIOGLITAZONE-GLIMEPIRIDE 30-4 | 66993-0822-30 | 11.30412 | EACH | 2026-01-21 |
| PIOGLITAZONE-GLIMEPIRIDE 30-2 | 66993-0821-30 | 10.80087 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Pioglitazone-Glimepiride
What is the Current Market Landscape for Pioglitazone-Glimepiride?
Pioglitazone-Glimepiride is a combination oral antidiabetic medication used to manage type 2 diabetes mellitus. The combination offers a dual mechanism by improving insulin sensitivity and stimulating insulin secretion. Its primary competitors include other fixed-dose combinations like Metformin-Glimepiride, Sitagliptin-Glimepiride, and standalone drugs such as Pioglitazone and Glimepiride.
The market for diabetes treatments has experienced consistent growth, driven by the increasing prevalence of type 2 diabetes globally. The compound's market share is influenced by regulatory approvals, patent status, safety profiles, and pricing strategies differing across regions (North America, Europe, Asia-Pacific).
Regulatory Status and Patent Landscape
As of 2023, pioglitazone patents have mostly expired or are close to expiry in key markets, opening the market to generic manufacturers. Glimepiride's patent expired earlier, leading to competition from generics.
- Generic Availability: Multiple generics of pioglitazone and glimepiride are accessible globally, pressuring branded prices.
- Market Entrants: Major pharmaceutical companies such as Teva, Sun Pharmaceuticals, and Mylan produce generic versions.
Pricing Trends and Projections
Current Pricing Spectrum
| Region | Brand Name | Average Price per Pack | Generic Price Range | Key Notes |
|---|---|---|---|---|
| North America | Actos (brand) | $150 (30 tablets) | $50–$100 (generics) | Branded prices are significantly higher; insurance often covers branded drugs. |
| Europe | Pioglitazone-Glimepiride (generic) | €10–€15 (30 tablets) | €5–€12 (generics) | European markets see a wider range depending on healthcare systems. |
| Asia-Pacific | Local generics | $5–$8 (30 tablets) | similar prices | Price sensitivity drives demand for generics. |
Future Projections (Next 5 Years)
- The global market is projected to grow at a compound annual growth rate (CAGR) of approximately 4.5% through 2028.
- Price reductions for branded formulations, especially in mature markets, will continue as more generics enter the market.
- Patent expiries will further push prices downward, with generic prices potentially declining by 50% over the next five years.
- Regulatory developments in emerging markets could increase competition and reduce prices further.
Market Drivers and Risks
Drivers:
- Rise in global diabetes prevalence, expected to reach 700 million people by 2045.
- Growing preference for fixed-dose combination drugs for adherence.
- Healthcare initiatives reducing costs through generic substitution.
Risks:
- Safety concerns associated with pioglitazone's links to bladder cancer may restrict usage in some populations.
- Regulatory restrictions or safety warnings could dampen demand and affect pricing.
Competitive Landscape
| Competitor | Focus Area | Market Share (Estimated) | Price Positioning |
|---|---|---|---|
| Teva | Generics of pioglitazone-glimepiride | 20% | Low, prioritizing volume |
| Novartis | Branded formulations | 25% | Premium, focused on patent products |
| Local Asian Firms | Generics and local brands | 35% | Very low, high volume, price-sensitive |
Key Takeaways
- The Pioglitazone-Glimepiride market is consolidating with increased generic competition.
- Prices are expected to decline significantly across regions over the next five years, especially in markets with patent expiries.
- Regulatory issues surrounding pioglitazone safety profiles might influence pricing and market shares.
- The overall market growth remains steady, driven by rising diabetes prevalence and demand for combination therapies.
FAQs
1. How will patent expiries influence the Pioglitazone-Glimepiride price in the next five years?
Patent expiries will increase generic competition, leading to a potential price decline of up to 50% in key markets, depending on regulatory approvals and market dynamics.
2. Are there significant safety concerns affecting the market?
Yes. Pioglitazone has associations with bladder cancer, leading to regulatory warnings in some regions, which can limit its use and influence pricing strategies.
3. Which regions will see the highest price reductions?
European and North American markets will experience the most significant reductions due to widespread generic availability and ongoing price competition.
4. What factors could disrupt projected market growth?
Emerging safety concerns, regulatory restrictions, and shifts toward newer drug classes like SGLT2 inhibitors could limit growth.
5. How do price trends for Pioglitazone-Glimepiride compare with other fixed-dose combinations?
Prices are similar, with generics generally priced lower than branded counterparts. Fixed-dose products combining newer drug classes tend to have higher prices, maintaining a premium segment.
References
- World Health Organization. (2022). Diabetes Fact Sheet. https://www.who.int/news-room/fact-sheets/detail/diabetes
- IMS Health. (2022). Global Diabetes Market Report.
- U.S. Food and Drug Administration. (2022). Pioglitazone Safety Labeling Changes.
- European Medicines Agency. (2022). Zynquista (sitagliptin-glimepiride) assessment report.
- MarketWatch. (2023). Diabetes Drugs Market Trends.
More… ↓
