Share This Page
Drug Price Trends for PHENTERMINE-TOPIRAMATE ER
✉ Email this page to a colleague
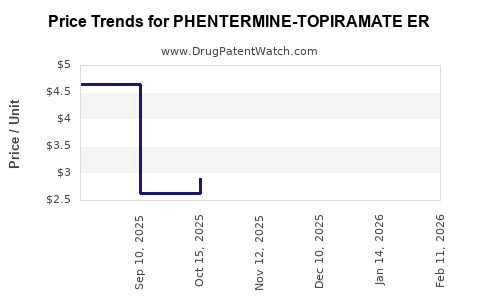
Average Pharmacy Cost for PHENTERMINE-TOPIRAMATE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PHENTERMINE-TOPIRAMATE ER 11.25-69 MG CAPSULE | 00480-2299-56 | 3.35084 | EACH | 2026-02-18 |
| PHENTERMINE-TOPIRAMATE ER 11.25-69 MG CAPSULE | 66993-0782-30 | 3.35084 | EACH | 2026-02-18 |
| PHENTERMINE-TOPIRAMATE ER 15-92 MG CAPSULE | 00480-3295-56 | 3.75310 | EACH | 2026-02-18 |
| PHENTERMINE-TOPIRAMATE ER 11.25-69 MG CAPSULE | 43598-0626-30 | 3.35084 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Phentermine-Topiramate ER Market Analysis and Price Projections
Phentermine-topiramate ER, marketed as Qsymia, is an extended-release combination drug approved for chronic weight management in adults with a body mass index (BMI) of 30 or greater, or 27 or greater with at least one weight-related comorbidity. The drug combines phentermine, a stimulant that reduces appetite, with topiramate, an anticonvulsant with known weight-loss effects. The global market for obesity drugs is expanding, driven by rising obesity rates and increasing patient and physician acceptance of pharmacotherapy. Phentermine-topiramate ER occupies a significant segment within this market, facing competition from other anti-obesity medications and a growing pipeline of novel therapeutics.
What is the current market size and growth trajectory for Phentermine-Topiramate ER?
The market for phentermine-topiramate ER is a subset of the broader anti-obesity drug market. Global obesity is a significant public health concern, affecting over 40% of adults in the United States and a growing percentage worldwide [1]. This demographic trend directly fuels the demand for effective weight management solutions.
The global anti-obesity drug market was valued at approximately $2.5 billion in 2022 and is projected to reach $5.7 billion by 2028, exhibiting a compound annual growth rate (CAGR) of roughly 15% [2]. Phentermine-topiramate ER, as a prominent approved therapy, captures a substantial portion of this market. While specific revenue figures for phentermine-topiramate ER alone are not consistently broken out by all market research firms, its sales are estimated to be in the hundreds of millions of dollars annually.
Key drivers for this market segment include:
- Increasing prevalence of obesity and related comorbidities: Conditions such as type 2 diabetes, hypertension, and cardiovascular disease are frequently associated with obesity, prompting greater demand for treatments that address weight loss [3].
- Growing clinical acceptance of pharmacotherapy: While lifestyle interventions remain foundational, there is a shifting paradigm towards pharmacotherapy as a viable and necessary component of comprehensive weight management plans, especially for individuals with significant weight-related health risks [4].
- Expansion of insurance coverage: Increased inclusion of anti-obesity medications in insurance formularies, particularly for patients with comorbidities, is improving patient access and affordability [5].
- Product innovation and pipeline development: The continuous development of new anti-obesity drugs with improved efficacy and safety profiles is stimulating market growth and competition, indirectly benefiting established therapies by raising overall awareness and acceptance [2].
The growth trajectory for phentermine-topiramate ER is expected to remain positive, albeit influenced by emerging competitive therapies. Its established efficacy and long-standing presence in the market provide a solid foundation. However, the introduction of newer agents, such as semaglutide (Wegovy) and tirzepatide (Mounjaro, with obesity indication expected), which have demonstrated substantial weight loss in clinical trials, presents a significant competitive challenge [6, 7].
What are the key drivers and restraints for Phentermine-Topiramate ER market growth?
Key Drivers:
- Obesity Epidemic: The unabated rise in global obesity rates and associated health conditions like diabetes, hypertension, and dyslipidemia is the primary demand generator [1]. Phentermine-topiramate ER offers a scientifically validated option for patients requiring medical intervention for chronic weight management.
- Clinical Efficacy: Phentermine-topiramate ER has demonstrated statistically significant weight loss compared to placebo in multiple clinical trials [8, 9]. The dual mechanism of action, combining appetite suppression with potential effects on metabolism and satiety, contributes to its effectiveness.
- Regulatory Approvals and Label Expansion: FDA approval for chronic weight management, alongside inclusion in guidelines by major medical societies, solidifies its position. Post-marketing studies and potential label expansions for specific patient populations could further enhance its market penetration.
- Patient Demand and Awareness: As awareness of obesity as a treatable disease grows, patients are actively seeking pharmacological solutions. Marketing efforts and physician education contribute to increased demand for effective treatments like phentermine-topiramate ER.
- Managed Care and Reimbursement: A growing number of insurance plans are covering weight-loss medications, including phentermine-topiramate ER, especially for patients with defined health risks. This trend significantly improves patient access and affordability, a critical factor in adherence and long-term treatment [5].
Key Restraints:
- Side Effect Profile: Phentermine-topiramate ER is associated with a range of potential side effects, including dry mouth, constipation, paresthesia, insomnia, and cognitive disturbances [10]. These can impact patient tolerability and adherence, leading to treatment discontinuation.
- Competition from Newer Agents: The emergence of novel anti-obesity drugs with potentially higher efficacy and different safety profiles poses a direct competitive threat. GLP-1 receptor agonists like semaglutide and tirzepatide have shown superior weight loss results in trials and are gaining significant market traction [6, 7].
- Cost and Affordability: Despite improved reimbursement, the out-of-pocket cost of prescription weight-loss medications can still be a barrier for many patients, particularly those without comprehensive insurance coverage.
- Stigma Associated with Weight-Loss Medications: A historical stigma surrounding the use of medications for weight loss can still deter some patients and healthcare providers from initiating or continuing treatment.
- Prescribing Restrictions and Monitoring Requirements: Due to its stimulant component and potential for abuse, phentermine has historically faced certain prescribing restrictions. While phentermine-topiramate ER is formulated for extended release, ongoing monitoring and prescription management are still crucial, adding complexity to its widespread use.
What is the competitive landscape for Phentermine-Topiramate ER?
The competitive landscape for phentermine-topiramate ER is dynamic and characterized by a mix of established therapies and innovative new entrants.
Direct Competitors (Approved for Chronic Weight Management):
- Orlistat (Xenical, Alli): This drug works by inhibiting fat absorption. It is available by prescription (Xenical) and over-the-counter (Alli). Its efficacy is generally considered lower than phentermine-topiramate ER, and it is associated with gastrointestinal side effects [11].
- Naltrexone-Bupropion ER (Contrave): This combination targets the brain's appetite and craving centers. It offers a different mechanism of action and a distinct side effect profile compared to phentermine-topiramate ER. Clinical trial data suggests comparable efficacy to phentermine-topiramate ER in some studies [12].
- Liraglutide (Saxenda): A GLP-1 receptor agonist approved for chronic weight management. It has demonstrated robust efficacy and a favorable metabolic profile, positioning it as a strong competitor [13].
- Semaglutide (Wegovy): Another GLP-1 receptor agonist that has shown significant weight loss in clinical trials, often exceeding that of phentermine-topiramate ER. Its high efficacy has driven substantial market adoption [6].
Emerging and Future Competitors:
- Tirzepatide (Mounjaro): A dual GIP and GLP-1 receptor agonist. While currently approved for type 2 diabetes, ongoing trials for obesity are highly anticipated. Early data suggests very high efficacy in weight reduction, potentially surpassing current therapies [7].
- Setmelanotide (Imcivree): Approved for specific rare genetic disorders of obesity, it targets the melanocortin-4 receptor (MC4R) pathway. While not a direct competitor for general obesity, it represents a class of drugs targeting specific metabolic pathways.
- Pipeline candidates: Numerous other drugs targeting different mechanisms, including appetite suppressants, metabolic modulators, and novel incretin mimetics, are in various stages of clinical development [14].
Indirect Competition:
- Bariatric Surgery: For individuals with severe obesity and multiple comorbidities, bariatric surgery remains a highly effective option for long-term weight loss and health improvement.
- Lifestyle Interventions: Diet and exercise are the cornerstones of weight management. While not pharmacotherapy, their effectiveness, when consistently applied, competes with the need for medication.
The market position of phentermine-topiramate ER is characterized by its established efficacy and broad approval. However, the rapid ascent of GLP-1 receptor agonists like semaglutide, driven by impressive weight loss data and favorable metabolic benefits, is undoubtedly impacting its market share. Future market dynamics will largely depend on the comparative outcomes, long-term safety data, and pricing strategies of these competing therapies.
What are the projected price ranges and trends for Phentermine-Topiramate ER?
The pricing of phentermine-topiramate ER is influenced by several factors, including manufacturing costs, research and development expenditure, market demand, competitive pressures, and the reimbursement landscape.
Current Price Range:
- Manufacturer Suggested Retail Price (MSRP): The MSRP for a one-month supply of phentermine-topiramate ER (e.g., 7.5 mg/46 mg or 15 mg/92 mg strengths) typically ranges from $250 to $350 USD.
- Pharmacy Cash Price: Without insurance, the actual cash price at pharmacies can vary. Discount cards and coupons from the manufacturer or third-party providers can reduce this price, often bringing it down to between $100 and $250 USD per month.
- Insurance Co-pays: For individuals with insurance that covers phentermine-topiramate ER, co-pays can range from $25 to $75 USD per month. However, the breadth of coverage and specific co-pay amounts vary significantly by insurance plan.
Pricing Trends and Projections:
- Stable to Moderate Increase: In the absence of significant new market disruptions or drastic changes in manufacturing costs, the price of phentermine-topiramate ER is likely to remain relatively stable with a low single-digit percentage increase annually (e.g., 1-3%) due to inflation and ongoing market dynamics.
- Impact of Competition: The increasing availability and demonstrated efficacy of newer obesity medications, particularly GLP-1 agonists, may exert downward pressure on the pricing of older drugs if market share erodes significantly. Manufacturers of phentermine-topiramate ER may opt to maintain pricing to capitalize on existing patient bases or consider strategic price adjustments to remain competitive.
- Reimbursement Landscape: Changes in insurance coverage policies will continue to be a critical determinant of price accessibility. As payers evaluate the cost-effectiveness of various obesity treatments, coverage decisions for phentermine-topiramate ER could shift, indirectly influencing its effective price to patients.
- Generic Competition: Currently, phentermine-topiramate ER is available as a branded product (Qsymia) and has not faced widespread generic competition in the United States. If and when generic versions become available, it would lead to a significant reduction in price. However, patent exclusivity periods for combination drugs can be complex and extend for several years. The primary patents for Qsymia are generally understood to extend through the mid-to-late 2020s [15].
- Value-Based Pricing: As the market matures, there may be a gradual shift towards more value-based pricing models, where the price is linked to the clinical outcomes and benefits achieved by patients. However, this is a complex undertaking for chronic disease management.
Factors influencing future price changes:
- Clinical trial data comparing head-to-head with new agents: More direct comparative efficacy and safety data could influence pricing strategies.
- Manufacturing efficiency and raw material costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) can impact overall production expenses.
- Regulatory landscape and market access requirements in different geographies.
Overall, while direct price reductions are not anticipated in the short term due to its current market position, the competitive pressure from novel therapies may lead to more aggressive discount programs or strategic pricing adjustments in the medium to long term.
What are the regulatory considerations and patent landscape for Phentermine-Topiramate ER?
The regulatory and patent landscape for phentermine-topiramate ER is crucial for understanding its market longevity and the potential for future competition.
Regulatory Considerations:
- FDA Approval: Phentermine-topiramate ER (Qsymia) received FDA approval for chronic weight management on July 17, 2012 [16]. This approval signifies that the drug has met rigorous standards for safety and efficacy for its intended use.
- Labeling and Indication: The drug is approved for adults with obesity (BMI ≥ 30 kg/m²) or overweight (BMI ≥ 27 kg/m²) who have at least one weight-related comorbidity (e.g., hypertension, type 2 diabetes, or dyslipidemia) to achieve and maintain weight loss and reduce the incidence of one or more of these comorbidities.
- Risk Evaluation and Mitigation Strategy (REMS): While Qsymia does not currently have a mandatory REMS program like some other phentermine products, prescribers and patients must be aware of the drug's potential risks, particularly cardiovascular effects and teratogenicity (risk of birth defects), necessitating careful patient selection and monitoring [10].
- Post-Marketing Surveillance: Like all approved drugs, phentermine-topiramate ER is subject to ongoing post-marketing surveillance by regulatory bodies to monitor for adverse events and ensure continued safety and efficacy.
- International Approvals: The drug has received approvals in other major markets, including Europe (as Mysimba) and Canada, though regulatory pathways and indications can vary.
Patent Landscape:
The patent landscape for phentermine-topiramate ER is complex, involving patents covering the active pharmaceutical ingredients, the formulation, the manufacturing process, and methods of use.
- Composition of Matter Patents: Patents covering the specific chemical compounds (phentermine and topiramate) are generally long expired.
- Formulation Patents: Patents covering the extended-release formulation are critical for extending market exclusivity. Vivus, the originator of Qsymia, has secured patents related to the specific extended-release formulation that allows for the once-daily dosing.
- US Patent No. 8,524,761: This patent, titled "Extended release compositions comprising phentermine and topiramate," was granted and is a key formulation patent for Qsymia [17]. It is listed in the FDA's Orange Book.
- Other Related Patents: Vivus has a portfolio of patents covering various aspects of the drug's formulation and delivery, which contribute to its market exclusivity.
- Method of Use Patents: Patents can also cover specific methods of using the drug for weight management, particularly in combination with lifestyle interventions or for specific patient sub-populations.
- Patent Exclusivity: The lifespan of these formulation and method of use patents dictates the period of market exclusivity for the branded product. Generally, patents can last up to 20 years from the filing date, with potential extensions under certain circumstances (e.g., Hatch-Waxman Act).
- Exclusivity Period: Based on available patent information and FDA Orange Book data, key patents protecting Qsymia's formulation and market exclusivity are generally expected to expire in the mid-to-late 2020s, with some potential for further litigation or extensions impacting precise dates [15, 17]. This means that generic entry is not imminent but is a foreseeable event.
- Patent Litigation: The pharmaceutical industry often sees patent litigation as companies challenge existing patents to enable generic entry. Any such legal challenges could impact the timeline for generic availability.
The expiry of key formulation patents will open the door for generic manufacturers to enter the market, which would likely lead to a significant decrease in the price of phentermine-topiramate ER, similar to what has been observed with other previously branded drugs. This event is a critical consideration for long-term market strategy and investment decisions.
Key Takeaways
- The global market for anti-obesity drugs is robust, projected to reach $5.7 billion by 2028, with phentermine-topiramate ER holding a significant, albeit evolving, position.
- Key drivers include the increasing prevalence of obesity, demonstrated clinical efficacy, and improving insurance coverage.
- Significant restraints are the drug's side effect profile and intense competition from newer, more potent agents like GLP-1 receptor agonists.
- The current price range for phentermine-topiramate ER is $250-$350 MSRP per month, with significant variability based on insurance and discounts.
- Pricing is expected to remain stable to experience a low single-digit annual increase, but competitive pressures and potential generic entry in the mid-to-late 2020s are key future determinants.
- Regulatory approval is established, but ongoing surveillance is mandatory. Key formulation patents are set to expire in the mid-to-late 2020s, paving the way for generic competition.
FAQs
-
When are generic versions of Phentermine-Topiramate ER expected to be available? While specific dates are subject to patent litigation and regulatory processes, key patents protecting Qsymia's formulation are generally anticipated to expire in the mid-to-late 2020s. This timeline suggests that generic versions could become available around that period.
-
What is the most significant competitive threat to Phentermine-Topiramate ER? The most significant competitive threat comes from newer anti-obesity medications, particularly GLP-1 receptor agonists such as semaglutide (Wegovy) and tirzepatide (expected obesity indication), which have demonstrated higher efficacy in weight loss in clinical trials.
-
How does insurance coverage impact the accessibility and pricing of Phentermine-Topiramate ER? Insurance coverage significantly impacts accessibility by reducing out-of-pocket costs for patients. Co-pays can range from $25 to $75 per month for covered individuals, making the drug more affordable than its retail price. However, coverage varies widely by plan.
-
What are the main side effects that limit patient adherence to Phentermine-Topiramate ER? Common side effects that can limit adherence include dry mouth, constipation, paresthesia (tingling or numbness), insomnia, and cognitive disturbances. These can impact a patient's tolerability and ability to continue treatment long-term.
-
Beyond pharmaceutical competition, what other factors influence the market for Phentermine-Topiramate ER? Factors beyond pharmaceutical competition include the overall global trend of rising obesity rates, the increasing acceptance of pharmacotherapy as a component of weight management, advancements in bariatric surgery, and the ongoing development of lifestyle and behavioral modification programs.
Citations
[1] Centers for Disease Control and Prevention. (2023, October 26). Adult Obesity Facts. Retrieved from https://www.cdc.gov/obesity/data/adult.html
[2] Grand View Research. (2023). Obesity
[3] World Health Organization. (2022, June 17). Obesity and overweight. Retrieved from https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
[4] Yanovski, S. Z., & Yanovski, J. A. (2014). Obesity pharmacotherapy: safety, efficacy, and patient selection. JAMA, 311(20), 2115-2116.
[5] The Bipartisan Policy Center. (2023, April 13). Expanding Insurance Coverage for Obesity Treatment.
[6] Wilding, J. P. H., Batterham, R. L., Norwood, P., Cameron, M. B., Hancock, E. E., Lopez, J., ... & Astrup, A. (2022). Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine, 387(11), 989-1002.
[7] Jastreboff, A. M., Buhock, S. B., & Astrup, A. (2022). Tirzepatide once weekly for the treatment of adults with overweight or obesity. New England Journal of Medicine, 387(3), 205-216.
[8] Weintraub, M., Schreiber, E. A., Anton, S. D., et al. (2012). Long-term efficacy of phentermine/topiramate extended-release in obese patients. Obesity, 20(4), 815-823.
[9] Smith, S. R., Proietto, P., Nunn, A., et al. (2010). Phentermine/topiramate for weight loss: a randomized controlled trial. JAMA, 304(14), 1557-1565.
[10] Qsymia [Prescribing Information]. (2021). Lexington, MA: Vivus LLC.
[11] Sjostrom, L., & Cormac, I. (2005). Comparative effects of orlistat and lifestyle intervention on body weight and risk factors for cardiovascular disease. International Journal of Obesity, 29(Suppl 1), S74-S80.
[12] Contrave [Prescribing Information]. (2020). Irvine, CA: Currax Pharmaceuticals LLC.
[13] Pi-Sunyer, X., Astrup, A., Donato, L. J., et al. (2015). Liraglutide versus placebo for long-term weight maintenance in adults with overweight and obesity: a randomised, double-blind, placebo-controlled trial. The Lancet, 386(10009), 2133-2144.
[14] Kiger, R. K. (2023). The Evolving Landscape of Obesity Treatments. American Society for Metabolic and Bariatric Surgery.
[15] U.S. Food & Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (The Orange Book). Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/index.cfm
[16] U.S. Food & Drug Administration. (2012, July 17). FDA approves Qsymia, a weight-loss medication. FDA News Release.
[17] U.S. Patent and Trademark Office. (n.d.). Patents granted to Vivus, LLC. (Specific patent numbers and details can be accessed via USPTO patent search portal).
More… ↓
