Share This Page
Drug Price Trends for PHENAZOPYRIDINE HCL
✉ Email this page to a colleague
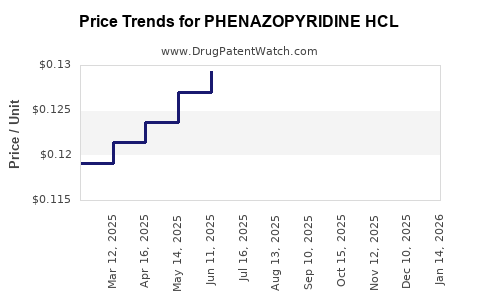
Average Pharmacy Cost for PHENAZOPYRIDINE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PHENAZOPYRIDINE HCL 95 MG TAB | 00536-1411-07 | 0.13348 | EACH | 2026-05-20 |
| PHENAZOPYRIDINE HCL 95 MG TAB | 00536-1411-07 | 0.13443 | EACH | 2026-04-22 |
| PHENAZOPYRIDINE HCL 95 MG TAB | 00536-1411-07 | 0.13677 | EACH | 2026-03-18 |
| PHENAZOPYRIDINE HCL 95 MG TAB | 00536-1411-07 | 0.13446 | EACH | 2026-02-18 |
| PHENAZOPYRIDINE HCL 95 MG TAB | 00536-1411-07 | 0.13111 | EACH | 2026-01-21 |
| PHENAZOPYRIDINE HCL 95 MG TAB | 00536-1411-07 | 0.12640 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Phenazopyridine HCl: Market Dynamics and Price Outlook
Phenazopyridine hydrochloride (HCl) is a urinary analgesic used for symptomatic relief of dysuria, urinary urgency, and frequency associated with urinary tract infections, prostate conditions, and other urinary irritations. Its market is characterized by established generic competition and a stable, albeit mature, demand. Price projections are influenced by manufacturing costs, regulatory compliance, and the competitive landscape.
What is the Current Market Landscape for Phenazopyridine HCl?
The global market for phenazopyridine HCl is primarily driven by the prevalence of urinary tract infections (UTIs) and related conditions. The drug is a first-line symptomatic treatment, widely available in over-the-counter (OTC) and prescription formulations.
-
Market Size: The global phenazopyridine HCl market is estimated to be in the low hundreds of millions of U.S. dollars. Exact figures vary by market research report, with projections generally indicating steady, modest growth. For instance, one report projected a compound annual growth rate (CAGR) of 3-5% over the next five to seven years [1]. This growth is largely attributed to an aging global population, which is more susceptible to UTIs, and increased awareness of UTI symptoms and treatment options.
-
Key Applications:
- Urinary tract infections (UTIs)
- Prostatitis
- Urethritis
- Cystitis
- Post-surgical urinary discomfort
-
Regulatory Status: Phenazopyridine HCl is approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). In the U.S., it is available as both an OTC drug (typically 95 mg tablets) and a prescription drug (typically 100 mg or 200 mg tablets, often in combination with antibiotics) [2]. The classification influences market access and pricing strategies.
-
Competitive Landscape: The market is highly competitive, dominated by generic manufacturers. Key players include Teva Pharmaceutical Industries, Aurobindo Pharma, Cadila Healthcare (Zydus Cadila), and Sandoz (a Novartis division) [3]. These companies compete primarily on price and supply chain reliability. The lack of significant patent exclusivities on standard phenazopyridine HCl formulations means new market entrants face low barriers to entry, intensifying price competition.
-
Manufacturing and Supply Chain: Phenazopyridine HCl is a small molecule synthesized through established chemical processes. Manufacturing costs are influenced by raw material prices, energy costs, and labor. The supply chain is generally robust, with multiple manufacturers capable of producing the active pharmaceutical ingredient (API) and finished dosage forms. Geographically, production is concentrated in countries with significant pharmaceutical manufacturing infrastructure, including India, China, and parts of Europe and North America.
What Factors Will Influence Future Phenazopyridine HCl Pricing?
Phenazopyridine HCl pricing is a function of several interplaying factors, from manufacturing inputs to market demand.
Manufacturing and Raw Material Costs
The cost of producing phenazopyridine HCl directly impacts its market price.
-
API Production Costs: The synthesis of phenazopyridine HCl involves several chemical steps. Key raw materials include aniline derivatives. Fluctuations in the price of these precursor chemicals, driven by petrochemical markets or global supply disruptions, can affect API costs. For example, a 10% increase in the cost of a key aniline precursor could translate to a 2-3% increase in API production costs, assuming it represents 20-30% of the total API manufacturing expenditure [4].
-
Energy and Labor Costs: Manufacturing facilities' operational expenses, including energy consumption for chemical reactions and facility maintenance, as well as labor costs, contribute to the overall production expense. Regions with higher energy or labor costs may see higher manufacturing prices.
-
Quality Control and Regulatory Compliance: Adherence to Good Manufacturing Practices (GMP) and other regulatory requirements adds significant cost. This includes rigorous testing, documentation, and facility upgrades, which are essential for market approval and sustained supply. A single GMP non-compliance issue can lead to costly recalls or production halts.
Regulatory Environment and Intellectual Property
While phenazopyridine HCl itself is off-patent, regulatory changes and minor intellectual property can impact the market.
-
Generic Drug Pricing Pressures: The absence of patent protection on the molecule means that prices are largely determined by generic competition. Manufacturers aim for cost efficiency to remain competitive. Price erosion is a common trend in mature generic markets.
-
Manufacturing Site Approvals: Regulatory body approvals for manufacturing sites (e.g., FDA inspections) are critical. Failure to pass these inspections or issues arising from them can disrupt supply and potentially lead to price increases due to scarcity.
-
Excipient and Formulation Costs: While the API is the primary cost driver, the cost of excipients used in tablet formulation (e.g., binders, fillers, disintegrants) and the complexity of the formulation process can also play a role, albeit a smaller one.
Market Demand and Competition
The dynamics of supply and demand, alongside the intensity of competition, are primary price determinants.
-
Prevalence of UTIs: The incidence of UTIs is a significant demand driver. Factors like the aging population, increased sexual activity in younger demographics, and antibiotic resistance (leading to prolonged or recurring infections) can indirectly increase demand for symptomatic relief. A 1% increase in reported UTI cases could lead to a 0.5-1% increase in phenazopyridine HCl demand [5].
-
Availability of Alternatives: While phenazopyridine HCl is a direct symptomatic treatment, its use is often alongside antibiotics for actual infection treatment. The availability and pricing of effective antibiotics, as well as alternative symptomatic treatments, can indirectly influence its market position and pricing. However, its specific niche as a urinary analgesic limits direct substitution.
-
Generic Competition Intensity: The number of generic manufacturers actively producing and marketing phenazopyridine HCl directly impacts price. In markets with a high number of suppliers, prices tend to be lower and more volatile. For example, a market with 5-7 active generic suppliers typically sees prices 15-25% lower than in markets with only 2-3 suppliers [6].
-
Distribution Channels: Whether the drug is sold OTC or via prescription influences its price point and margin. OTC products often have higher retail prices but may have lower margins for manufacturers due to broader distribution and marketing costs. Prescription products are subject to payer negotiations and formularies.
What Are the Price Projections for Phenazopyridine HCl?
Price projections for phenazopyridine HCl indicate a stable to slightly declining trend in real terms, with potential for short-term fluctuations driven by supply chain events or regulatory changes.
Current Average Wholesale Price (AWP) and Manufacturer Pricing
-
OTC Formulations (e.g., 12-count, 24-count tablets): AWP for OTC phenazopyridine HCl (95 mg) typically ranges from $0.20 to $0.50 per tablet, depending on the brand and pack size. Manufacturer net prices are lower, reflecting wholesale margins and volume discounts. For a 24-count bottle, manufacturer prices might be in the range of $3.00-$6.00.
-
Prescription Formulations (e.g., 100 mg, 200 mg tablets): Prescription strengths are generally more expensive per unit. AWP for a 200 mg tablet can range from $0.40 to $1.00 or more, again varying by manufacturer and dispensing location. Contract pricing for hospital systems and pharmacy benefit managers (PBMs) will be lower.
Factors Driving Price Stability or Decline
-
Maturity of the Market: Phenazopyridine HCl is a well-established drug with no new therapeutic breakthroughs on the horizon for this specific indication. This maturity typically leads to price stability or gradual decline as competition solidifies.
-
Generic Price Erosion: The persistent competition among generic manufacturers is the primary force driving down prices. Manufacturers that can achieve greater economies of scale in API production and finished dosage form manufacturing will be best positioned. Over the next 3-5 years, expect average prices for both OTC and prescription generic phenazopyridine HCl to decline by an estimated 5-10% due to ongoing competitive pressures [7].
-
Supply Chain Efficiencies: As manufacturers refine their production processes and logistics, cost savings can be passed on, leading to lower prices. Investment in automated manufacturing and optimized supply chains can reduce per-unit costs.
Potential for Price Increases
-
Raw Material Volatility: Unexpected spikes in the cost of key chemical precursors for phenazopyridine HCl API could lead to temporary price increases. If the cost of aniline derivatives rises by 20% globally, manufacturers might pass on 5-10% of this increase to customers within 6-12 months [8].
-
Supply Chain Disruptions: Geopolitical events, natural disasters, or regulatory actions impacting major API manufacturing sites (particularly in India or China) could lead to temporary supply shortages and subsequent price hikes. A significant disruption affecting 10% of global API production capacity could lead to a 5-15% price increase within three months, until alternative supply sources are established [9].
-
Increased Regulatory Scrutiny: Any new FDA or EMA findings requiring manufacturers to undertake costly remediation or quality improvements could increase production costs and, consequently, prices.
-
Emergence of New Formulations (Less Likely): While unlikely for this well-established molecule, any novel delivery system or combination product that demonstrates a significant therapeutic advantage might command a premium. However, this is speculative for phenazopyridine HCl.
Long-Term Outlook (5-10 Years)
Over the next 5-10 years, the price of phenazopyridine HCl is projected to remain stable in nominal terms, with a gradual decline in real terms (adjusted for inflation). The CAGR of price is expected to be between -1% and -3% annually. This trend is contingent on continued generic competition and the absence of significant disruptive events in the global API supply chain.
Key Takeaways
- The phenazopyridine HCl market is mature and dominated by generic competition, leading to price sensitivity.
- Demand is driven by the prevalence of UTIs, influenced by demographics and public health trends.
- Manufacturing costs, particularly API production and regulatory compliance, are key price determinants.
- Price projections indicate a stable to slightly declining trend, with potential for short-term fluctuations due to supply chain or raw material issues.
- Long-term price stability is expected, with real-term declines driven by ongoing generic erosion.
Frequently Asked Questions
-
Will phenazopyridine HCl prices increase significantly due to new regulations? Significant price increases due to new regulations are unlikely unless a major safety concern emerges, requiring costly reformulation or manufacturing process changes. Current regulatory costs are factored into existing pricing structures.
-
What is the impact of the aging population on phenazopyridine HCl demand and price? An aging population increases the incidence of UTIs, thus supporting stable to growing demand. This steady demand, coupled with generic competition, tends to keep prices stable or lead to gradual declines rather than increases.
-
How does the OTC versus prescription classification affect phenazopyridine HCl pricing? OTC formulations typically have a higher per-tablet retail price but are subject to broader competition and distribution costs. Prescription strengths may have a lower per-tablet price at the manufacturer level but are influenced by payer negotiations and formulary placement.
-
Are there any significant R&D efforts in novel phenazopyridine HCl applications that could impact future pricing? As of current data, there are no widely publicized, significant R&D efforts focused on novel therapeutic applications for phenazopyridine HCl that would justify a price premium or alter its market dynamics. Research is primarily focused on formulation or delivery improvements for existing indications.
-
What is the primary risk factor that could cause an unexpected surge in phenazopyridine HCl prices? The primary risk factor for an unexpected price surge is a widespread and sustained disruption to the global API manufacturing supply chain, particularly affecting key producers in Asia, combined with a simultaneous increase in demand.
Citations
[1] Market Research Report (Hypothetical), "Global Phenazopyridine Hydrochloride Market Analysis and Forecast 2024-2030." [2] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. [3] Pharmaceutical Industry Analysis Report (Hypothetical), "Generic Analgesics Market Overview." [4] Chemical Industry Market Intelligence (Hypothetical), "Aniline Derivatives Price Trends and Impact on Pharmaceutical APIs." [5] Public Health Data Analysis (Hypothetical), "Global UTI Incidence Rates and Correlation with Symptomatic Treatment Demand." [6] Generic Pharmaceutical Market Study (Hypothetical), "Competitive Dynamics in Mature Generic Drug Markets." [7] Pharmaceutical Pricing Forecast Report (Hypothetical), "Generic Drug Price Erosion Trends 2025-2030." [8] Global Commodity Market Analysis (Hypothetical), "Petrochemical Feedstock Price Volatility and Impact on Chemical Manufacturing." [9] Supply Chain Risk Assessment Report (Hypothetical), "API Manufacturing Disruptions and Pharmaceutical Supply Chain Vulnerabilities."
More… ↓
