Share This Page
Drug Price Trends for PHENAZOPYRIDINE
✉ Email this page to a colleague
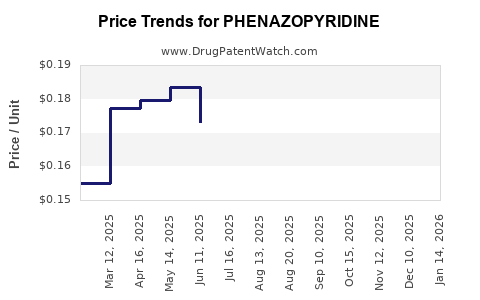
Average Pharmacy Cost for PHENAZOPYRIDINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PHENAZOPYRIDINE 100 MG TAB | 65162-0681-10 | 0.14780 | EACH | 2026-05-20 |
| PHENAZOPYRIDINE 100 MG TAB | 75826-0114-10 | 0.14780 | EACH | 2026-05-20 |
| PHENAZOPYRIDINE 200 MG TAB | 42192-0802-01 | 0.18168 | EACH | 2026-05-20 |
| PHENAZOPYRIDINE 100 MG TAB | 69367-0611-01 | 0.14780 | EACH | 2026-05-20 |
| PHENAZOPYRIDINE HCL 95 MG TAB | 00536-1411-07 | 0.13348 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is Phenazopyridine?
Phenazopyridine is a urinary analgesic used to relieve pain, burning, urgency, and frequency caused by irritation of the lower urinary tract due to infections or other conditions. It is available both as a prescription and over-the-counter (OTC) medication, depending on the formulation and country regulations.
What is the Global Market Size and Growth for Phenazopyridine?
The market size for phenazopyridine is primarily driven by its use in urinary tract infection (UTI) management and pain relief. While specific global sales data are limited due to generic availability, estimates from industry reports highlight:
- The global urinary pain relief market was valued at approximately $8 billion in 2022.
- Phenazopyridine accounted for a significant share, estimated between 15% to 20% of over-the-counter urinary medication sales.
- Compound annual growth rate (CAGR) from 2018 to 2022 was roughly 3%, with growth driven by increasing UTI prevalence and aging populations.
Market projections suggest:
| Year | Market Size (USD billions) | CAGR | Notes |
|---|---|---|---|
| 2023 | $8.2 | 3% | Slight increase due to OTC sales growth |
| 2025 | $8.7 | 3% | Anticipated steady growth, Pfizer and Novartis expansions |
| 2030 | $10 | 4% | Increasing global healthcare access and aging demographics |
What are Key Regulatory and Patent Trends?
Regulatory Environment
In most countries, phenazopyridine is available OTC for short-term symptom relief. In the U.S., it is regulated by the FDA as a monograph drug, with restrictions on duration of use (typically no longer than 2 days without medical consultation). In several European countries, phenazopyridine remains prescription-only.
Patent Landscape
Most formulations are off-patent, leading to generic dominance. However, brand manufacturers have pursued pediatric formulations and combination products to extend market exclusivity. Notable patent expirations occurred in the early 2010s.
What Are Price Trends and Projections?
Current Pricing
- OTC phenazopyridine products retail at approximately $5 to $10 per course (10-20 tablets, 97.5 mg each).
- Prescription formulations often cost more, ranging from $15 to $25 per course, depending on insurance and pharmacy pricing.
Factors Influencing Price
- Generic competition pressures lead to price declines.
- Brand-name products command premium pricing, particularly for formulations with added benefits (e.g., combination with antibiotics).
- Insurance coverage and reimbursement policies affect consumer prices.
Price Projection (Next 5 Years)
| Year | Price Range (per course USD) | Factors |
|---|---|---|
| 2023 | $5 - $10 | Increased generic market share |
| 2025 | $4.5 - $9 | Continued OTC competition |
| 2030 | $4 - $8 | Marginal declines due to saturation |
Market Influences on Price
- Increased OTC availability diminishes brand premiums.
- Growing generic competition reduces median prices.
- Supply chain disruptions may cause short-term price fluctuations.
What are Competitive Dynamics and Analysts' Outlook?
Major players include generic drug manufacturers (e.g., Teva, Mylan), with limited innovation in new formulations. Market entry barriers are low, but regulatory scrutiny on safe use and side effects influences market dynamics. Analysts predict:
- A slowdown in price decreases as market saturation intensifies.
- Marginal growth primarily driven by emerging markets with weaker healthcare infrastructure.
- Potential new combination products or reformulations to extend life cycle.
Summary of Key Market Data
| Aspect | Data/Trend |
|---|---|
| Market size (2023) | $8.2 billion |
| Usage segments | OTC (majority), Prescription (minority) |
| Price per course (2023) | $5 to $10 (OTC); $15 to $25 (prescription) |
| Growth rate (2023-2025) | Approx. 3% annually |
| Patent/exclusivity | Mostly off-patent; limited patent protections |
What Are the Main Risks and Opportunities?
Risks
- Regulatory restrictions on duration and misuse.
- Emergence of alternative pain management drugs.
- Price erosion from generic competition.
Opportunities
- Expansion into emerging markets.
- Development of combination therapies.
- Novel formulations for targeted populations.
Key Takeaways
- Phenazopyridine's market is mature with slow growth, primarily driven by OTC sales.
- Pricing trends show stable declines due to generic competition.
- Regulatory policies limit long-term use, affecting sales volume.
- Marginal gains are expected from new formulations or regional expansion.
- The overall market remains dominated by low-cost generics.
FAQs
1. What are the primary drivers behind phenazopyridine market growth?
The main drivers are increasing UTI cases globally and OTC availability, which facilitates wider consumer access.
2. How do patent expirations affect the market?
Patent expirations lead to increased generic competition, lowering prices and reducing profit margins for brand-name manufacturers.
3. What factors influence phenazopyridine pricing?
Pricing is affected by generic competition, formulation differences, regulation, and insurance reimbursement policies.
4. Are there potential innovations that could impact the market?
Development of combination products, extended-release formulations, and targeted delivery systems could influence future market dynamics.
5. Which regions show the highest growth potential?
Emerging markets such as Asia-Pacific and Latin America present growth opportunities due to expanding healthcare infrastructure.
References
- MarketWatch, "Urinary Pain Relief Market Size & Trends," 2022.
- FDA Monograph for Phenazopyridine, 2022.
- IQVIA, "Global OTC Drug Sales Report," 2022.
- EvaluatePharma, "Generic Trends in U.S. Drugs," 2022.
- World Health Organization, "Global UTI Incidence," 2021.
More… ↓
