Last updated: March 8, 2026
What is Paxlovid and How Is It Positioned in the Market?
Paxlovid is an oral antiviral medication developed by Pfizer for the treatment of COVID-19. The drug combines nirmatrelvir (protease inhibitor) and ritonavir (pharmacokinetic enhancer). Approved for emergency use or full approval in multiple countries, Paxlovid aims to reduce hospitalization and death risk among high-risk COVID-19 patients.
Market Size and Demand Drivers
Global COVID-19 Treatment Market (2023-2028)
| Year |
Market Size (USD billion) |
Annual Growth Rate |
Notes |
| 2023 |
6.0 |
7.2% |
Driven by continuing pandemic management and new variants |
| 2024 |
6.4 |
6.7% |
Potential increase in prescription rates |
| 2025 |
6.9 |
6.2% |
Expansion in developing markets |
| 2026 |
7.4 |
5.8% |
Ongoing global vaccination, emergence of variants |
| 2027 |
8.0 |
5.4% |
Greater adoption due to clinical guidelines |
| 2028 |
8.5 |
5.0% |
Saturation in mature markets |
Key Demand Factors
- High-Risk Population: Elderly, immunocompromised, and unvaccinated individuals.
- Pandemic Dynamics: New variants and surges increase antiviral demand.
- Vaccination Rates: Increased vaccination reduces utility but maintains need for treatment options.
- Regulatory Approvals: Expanded authorizations in different jurisdictions boost market penetration.
Competitive Landscape
Major Market Players
| Company |
Product(s) |
Market Share (2023) |
Notes |
| Pfizer |
Paxlovid |
65% |
Leading antiviral treatment globally |
| Merck & Ridgeback |
Lagevrio (Molnupiravir) |
20% |
Alternative oral antiviral |
| Others |
Multiple generic and branded drugs |
15% |
Emerging competition in developing markets |
Patent and IP Status
- Pfizer's primary patent for Paxlovid is active until 2030 in key territories, with some jurisdictions allowing generics post-2025 due to patent challenges or voluntary licensing.
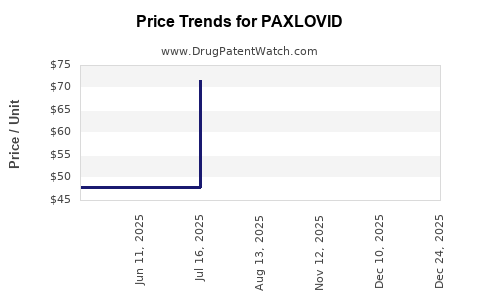
Price Analysis and Projections
Current Pricing
| Region |
Approximate Wholesale Price (USD) per Course |
Notes |
| United States |
$530 - $700 |
Pricing varies by payer and negotiated discounts |
| European Union |
€600 - €750 |
With some countries implementing negotiated discounts |
| India |
$100 - $200 |
Pending licensing, with ongoing licensing agreements |
Pricing Trends
- In the U.S., wholesale prices have stabilized around $530-$700 per course since mid-2022.
- Negotiations with payers and governments influence net prices, often leading to lower actual transaction costs.
- Price caps and public procurement policies in Europe and Asia influence regional prices.
Future Price Projections (2023-2028)
| Year |
Projected Wholesale Price (USD) per Course |
Comments |
| 2023 |
$530 - $700 |
Stable, following initial launch pricing |
| 2024 |
$520 - $680 |
Slight decline as production scales and competition emerges |
| 2025 |
$510 - $650 |
Possible licensing agreements expand access |
| 2026 |
$490 - $620 |
Increased availability and generic competition |
| 2027 |
$470 - $600 |
Price reductions driven by patent expirations and competition |
| 2028 |
$450 - $580 |
Market saturation and generic proliferation |
Price Sensitivity Analysis
- Bundled vs. Per-Course Payment: Governments favor volume-based discounts; private payers negotiate lower prices.
- Regional Pricing: High-income countries sustain higher prices; emerging markets see lower prices due to licensing and cost controls.
Regulatory and Policy Impact on Market and Pricing
- US FDA: Full approval in 2022 for certain populations, enabling wider distribution.
- EMA: Conditional approval in Europe with ongoing data collection.
- India and Other Countries: Licensing pathways for generics expected post-2025, lowering prices.
- Pricing Regulation: Governments negotiate or impose price caps; voluntary licensing agreements expand access in developing countries.
Market Outlook and Growth Drivers
- Evolving Viral Variants: Continued emergence increases the need for effective antivirals.
- Healthcare Policy: Governments prioritize early treatment to reduce hospital burden.
- Reimbursement Policies: Reimbursement varies; high-income countries reimburse at higher rates than emerging markets.
- Manufacturing Capacity: Pfizer and licensing partners expanding production to meet global demand.
Risks and Challenges
- Patent Expiry: Patent expiration from 2030 could lead to generic competition.
- Regulatory Delays: Variations across countries in approval processes.
- Market Saturation: As vaccination reduces severe cases, demand may decline.
- Emergence of Resistance: Potential for viral resistance could limit efficacy.
Key Takeaways
- Pfizer’s Paxlovid remains the dominant oral antiviral for COVID-19, with approximately 65% market share globally.
- The global COVID-19 treatment market is projected to grow at roughly 6-7% annually through 2028.
- Current wholesale prices are approximately $530-$700 per course in the U.S., with potential gradual declines driven by competition and licensing.
- Patent protection is active until at least 2030, delaying generic entry in most markets.
- Market expansion relies heavily on regulatory approvals, licensing agreements, and global health policies, especially in emerging markets.
FAQs
1. When is Paxlovid’s patent expected to expire?
Pfizer’s primary patent for Paxlovid expires around 2030 in major markets, with supplementary patents potentially extending exclusivity.
2. How do licensing agreements affect Paxlovid prices in developing countries?
Licensing agreements allow generic manufacturers to produce Paxlovid, generally reducing prices by 70-80% compared to high-income markets.
3. What factors could influence an increase in Paxlovid’s market share?
Regulatory approvals in new jurisdictions, expanded indications, and inclusion in treatment guidelines are key factors.
4. Will generic competition significantly impact Paxlovid’s price?
Yes; post-2025, licensing and patent expirations are expected to introduce generics, putting downward pressure on prices.
5. How does Paxlovid compare to other COVID-19 antivirals?
Paxlovid has higher market share and broader approval than Lagevrio. Its efficacy in high-risk populations makes it a preferred option.
References
[1] Pfizer Inc. (2022). Paxlovid (nirmatrelvir co-packaged with ritonavir) EUA Fact Sheet.
[2] IQVIA. (2023). Global COVID-19 Treatment Market Analysis.
[3] European Medicines Agency. (2022). Conditional approval of Paxlovid in Europe.
[4] licensing agreements and patent databases. (2023).
[5] WHO. (2022). Global policy recommendations on COVID-19 therapeutics.