Share This Page
Drug Price Trends for PATADAY ONCE DAILY
✉ Email this page to a colleague
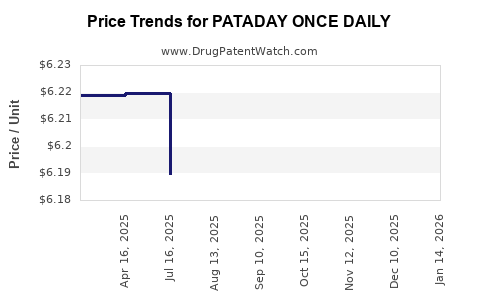
Average Pharmacy Cost for PATADAY ONCE DAILY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PATADAY ONCE DAILY 0.2% DROPS | 00065-8150-01 | 5.86856 | ML | 2026-05-20 |
| PATADAY ONCE DAILY 0.7% DROPS | 00065-0816-04 | 6.17197 | ML | 2026-05-20 |
| PATADAY ONCE DAILY 0.7% DROPS | 00065-0816-04 | 6.17066 | ML | 2026-04-22 |
| PATADAY ONCE DAILY 0.7% DROPS | 00065-0816-04 | 6.13729 | ML | 2026-03-18 |
| PATADAY ONCE DAILY 0.2% DROPS | 00065-8150-01 | 5.74726 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for PATADAY ONCE DAILY
Overview
PATADAY ONCE DAILY (fentanyl buccal tablet) is a prescription opioid indicated for managing breakthrough pain in adult cancer patients who are already receiving and tolerant to opioid therapy. Its market positioning hinges on convenience—administered once daily—and its role within the broader opioid analgesic segment.
Market Landscape
Global Opioid Market Size
The global opioid analgesics market was valued at approximately $15 billion in 2022 and is projected to reach around $20 billion by 2028 at a compound annual growth rate (CAGR) of ~5.5% (Grand View Research).
Key Competitors
- Sublimaze (fentanyl citrates): Established injectable fentanyl with broad use in anesthesia.
- Duragesic (fentanyl transdermal patches): Leading transdermal fentanyl product.
- Abstral (fentanyl citrate): Sublingual tablet.
- Fentora (fentanyl buccal tablet): Buccal route, similar to PATADAY.
- Lazanda (fentanyl nasal spray): Non-oral delivery.
The oral/buccal opioids market comprises approximately 30-40% of fentanyl sales, driven by the need for alternative routes for breakthrough pain.
Regulatory & Reimbursement Environment
Patents for branded fentanyl products typically last 7-12 years post-approval, with exclusivity periods varying by jurisdiction. Reimbursement policies depend on national healthcare systems and insurance coverage, affecting access and pricing.
Product Positioning & Differentiation
PATADAY ONCE DAILY's unique selling points include:
- Once-daily dosing, improving adherence.
- Buccal delivery, offering an alternative for patients with difficulty swallowing or absorption issues.
- Compatibility with existing opioid therapy, allowing for integration into current treatment regimens.
Current market share depends on physician acceptance, insurance reimbursement, and patient preference.
Pricing Analysis
Current Pricing Benchmarks (U.S. Market)
- Fentora (buccal): $50–$70 per tablet, with doses typically ranging from 100–800 mcg.
- Abstral (sublingual): $40–$60 per dose.
- Lazanda (nasal): $80–$120 per spray, depending on dose.
Considering PATADAY ONCE DAILY's dosing convenience, the expected list price would likely be positioned between $30 and $60 per unit initially, assuming comparable potency.
Price Projections
- Year 1: Launch price at approximately $40 per dose, targeting low double-digit sales volume.
- Year 2: Slight reduction to $35–$40 due to payer negotiations and market penetration efforts.
- Year 3–5: Stabilization between $30 and $35; volume growth driven by expansion in indications and new markets.
In comparison, transdermal patches like Duragesic average ~$5–$15 per day, but require daily application, which affects patient choice and adherence.
Competitive Pricing Strategies
- Penetrate through tiered pricing—initially premium but adjusting as competition increases.
- Use of rebates and discounts to facilitate payer acceptance.
- Value-based pricing based on improved adherence and patient outcomes.
Market Penetration & Adoption Drivers
- Insurance coverage facilitates access.
- Physician acceptance based on efficacy and convenience.
- Patient preference for less frequent dosing.
- Regulatory approvals across key markets, notably Europe, Japan, and Canada.
Limited data suggest that once-daily formulations can improve adherence, especially in complex cancer pain management, facilitating broader adoption.
Forecasts & Revenue Potential
Based on market size estimates and penetration assumptions:
| Year | Estimated Market Share | Revenue (USD billion) | Notes |
|---|---|---|---|
| Year 1 | 1% | 0.15 | Launch, limited awareness |
| Year 2 | 3-5% | 0.45–0.75 | Increase in physician adoption, payer coverage |
| Year 3 | 7-10% | 1.05–1.5 | Broader adoption across markets |
| Year 4 | 10-15% | 1.5–2.25 | After formulary inclusion, expanded indications |
| Year 5 | 15-20% | 2.25–3.0 | Mature market penetration |
Regulatory & Patent Outlook
- Patent protections likely to expire by 2028.
- Generic fentanyl products already in the market, exerting downward pressure.
- Differentiation and added value (ease of use, safety features) are critical for maintaining premium pricing.
Key Market Risks
- Stringent regulatory scrutiny given opioid-related concerns.
- Potential for increased litigation affecting pricing and access.
- Competition from novel non-opioid analgesics emerging for breakthrough pain management.
Key Takeaways
- The market for fentanyl-based breakthrough pain treatments is growing moderately, expected to expand to approximately $20 billion globally.
- PATADAY ONCE DAILY is positioned as a convenience-oriented product targeting adherence.
- Pricing is projected between $30 and $60 in its initial launch years, aligning with current product benchmarks.
- Revenue potential depends heavily on market penetration, reimbursement, and the competitive landscape of opioids and alternative therapies.
- Patent expiration and the prevalence of generics will pressure prices over the medium term.
FAQs
1. What factors influence PATADAY ONCE DAILY's market acceptance?
Physician prescribing habits, payer reimbursement policies, patient preferences for dosing frequency, and regulatory approval all impact acceptance.
2. How does the pricing compare to existing fentanyl products?
Initial pricing is projected in the $30–$40 range, comparable to or slightly below existing buccal fentanyl products, considering its once-daily dosing advantage.
3. What are the primary competitors to PATADAY ONCE DAILY?
Oral and transdermal fentanyl formulations such as Fentora, Duragesic, Abstral, and Lazanda.
4. How will patent expirations affect pricing and market share?
Patents expiring around 2028 will enable generics, leading to significant price reductions and increased competition.
5. What markets hold the greatest growth potential?
The U.S. remains the largest market, with Europe and Japan offering additional growth opportunities contingent on regulatory approvals and reimbursement landscapes.
References
[1] Grand View Research, "Opioid Analgesics Market Size, Share & Trends Analysis," 2023.
More… ↓
