Share This Page
Drug Price Trends for PATADAY
✉ Email this page to a colleague
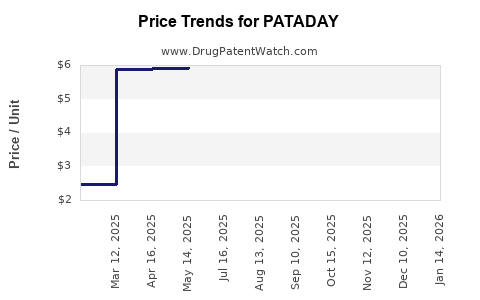
Average Pharmacy Cost for PATADAY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PATADAY ONCE DAILY 0.7% DROPS | 00065-0816-04 | 6.17197 | ML | 2026-05-20 |
| PATADAY TWICE DAILY 0.1% DROPS | 00065-4274-01 | 2.47641 | ML | 2026-05-20 |
| PATADAY ONCE DAILY 0.2% DROPS | 00065-8150-01 | 5.86856 | ML | 2026-05-20 |
| PATADAY TWICE DAILY 0.1% DROPS | 00065-4274-01 | 2.47633 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Pataday Market Overview and Price Projections
Pataday (olopatadine ophthalmic solution 0.2%) is an antihistamine eye drop used primarily for allergic conjunctivitis. The drug is marketed by Alcon and is available over-the-counter (OTC) in the United States and various other regions.
Market Size and Growth
The global allergic conjunctivitis treatment market was valued at approximately USD 750 million in 2022. Expected compound annual growth rate (CAGR) from 2023 to 2030 is around 4.8%, driven by rising allergy prevalence, increased awareness, and OTC availability.
Key regional markets:
| Region | Market Size (2022 USD millions) | CAGR (2023-2030) | Drivers |
|---|---|---|---|
| North America | 350 | 4.6% | High allergy awareness, OTC sales, healthcare access |
| Europe | 200 | 4.4% | Increased allergy diagnostics, OTC availability |
| Asia-Pacific | 130 | 5.2% | Rising urban allergies, expanding OTC markets |
| Latin America | 50 | 4.8% | Growing healthcare infrastructure, OTC sales |
Market Drivers and Challenges
Drivers:
- Increasing allergy prevalence globally, with some estimates indicating up to 30% of teens and adults affected.
- Over-the-counter availability of Pataday enhances accessibility.
- The shift towards self-medication reduces healthcare consultation barriers.
Challenges:
- Competitive landscape includes other antihistamines like Zaditor (ketotifen), Alaway, and prescription options such as Olopatadine (prescription-only formulations).
- Patent expiry on some formulations has resulted in generic competition, pressuring prices.
Price Trends and Projections
Current Pricing (as of 2023):
- Retail price per 0.2% bottle (roughly 2.5 mL): USD 20-25 in the US OTC market.
- Discounted or generic alternatives cost USD 10-15 per bottle.
- Prices vary based on location, pharmacy chains, and insurance coverage.
Historical Price Changes:
- Post-patent expiry of the patented formulations in late 2010s, prices dropped approximately 30-40%.
- Brand loyalty sustains premium pricing despite generics.
Projected Price Evolution (2023-2030):
| Year | Expected Retail Price Range (USD) | Key Factors |
|---|---|---|
| 2023 | 20-25 | Stable, market penetration, generic entries |
| 2025 | 18-24 | Increased generic market share |
| 2027 | 17-22 | Competition intensifies, price pressure |
| 2030 | 15-20 | Saturation, patent expirations, new entrants |
Price declines to USD 15-20 per bottle expected by 2030, with continued discounting on generics and store brands. Brand premiums might persist in premium retail outlets.
Competitive Landscape and Pricing Implications
| Product | Formulation | Market Status | Average Retail Price (USD) | Notes |
|---|---|---|---|---|
| Pataday | 0.2% ophthalmic solution | Market leader, OTC | 20-25 | Brand, perceived quality, marketing |
| Zaditor | 0.035% ketotifen | OTC, generic available | 10-15 | Similar efficacy, lower cost |
| Olopatadine | Prescription-only formulations | Prescription, OTC access | 30-35 | Higher price, prescription-required |
Regulatory and Pricing Influences
- Conversion of prescription formulations to OTC status in the US facilitates broad access but constrains pricing.
- Price regulation policies (e.g., in Europe and Canada) generally aim for affordability but may impact international pricing.
- Reimbursement schemes influence consumer out-of-pocket costs, especially in markets with insurance coverage.
Summary and Market Outlook
Pataday’s market dynamics align with typical over-the-counter allergy medication trends. Its pricing is influenced by brand identity, generic competition, and consumer preferences. Prices are expected to decline gradually over the next seven years, with stabilization around USD 15-20 per bottle by 2030. Market growth continues fueled by rising allergy incidence and increased OTC access.
Key Takeaways
- The global allergic conjunctivitis market is projected to grow at near 5% annually through 2030, reaching approximately USD 1 billion.
- Pataday remains a leading OTC antihistamine eye drop, with current retail prices averaging USD 20-25.
- Price declines will occur due to increasing generic competition, with expectation of USD 15-20 per bottle by 2030.
- Market expansion will be driven largely by rising allergy prevalence and OTC accessibility.
- Competitive pressures from lower-cost alternatives and generics will influence brand pricing and market share.
FAQs
1. What factors influence Pataday’s pricing in different markets?
Pricing depends on patent status, regulatory environment, local competition, healthcare policies, and consumer purchasing power.
2. How does Pataday compare in price to generic antihistamines?
Generics typically retail at approximately USD 10-15, about 50-25% less than Pataday, exerting downward pressure on branded prices.
3. When is Pataday expected to face increased generic competition?
Many formulations have already lost patent protection, with generics and store brands active in the market. Continued patent expirations will further increase competition through 2025-2027.
4. What regional differences exist in Pataday pricing?
North America and Europe tend to have higher prices due to brand loyalty and healthcare structure, while Asia-Pacific and Latin America show lower prices due to market maturity levels.
5. What is the outlook for Pataday’s market share?
While brand recognition supports maintained share, generic entry and price competition may reduce its market dominance over time, especially in price-sensitive markets.
References
[1] MarketWatch, "Global Allergic Conjunctivitis Market Size," 2023.
[2] Statista, "OTC Eye Drop Market Overview," 2022.
[3] Alcon Investor Relations, "Pataday Product Info," 2023.
[4] GoodRx, "Eye Drop Prices," 2023.
[5] Deloitte, "Pharmaceutical Patent Expirations and Market Impact," 2022.
More… ↓
