Share This Page
Drug Price Trends for PALIPERIDONE ER
✉ Email this page to a colleague
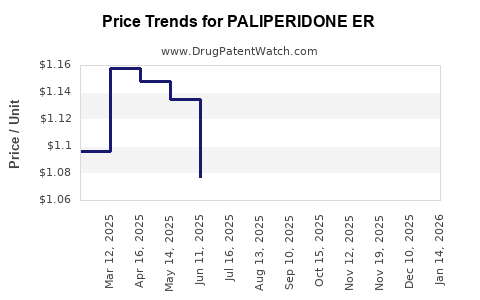
Average Pharmacy Cost for PALIPERIDONE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PALIPERIDONE ER 9 MG TABLET | 72819-0160-03 | 1.31889 | EACH | 2026-03-18 |
| PALIPERIDONE ER 1.5 MG TABLET | 16714-0866-01 | 0.97761 | EACH | 2026-03-18 |
| PALIPERIDONE ER 1.5 MG TABLET | 27808-0222-01 | 0.97761 | EACH | 2026-03-18 |
| PALIPERIDONE ER 1.5 MG TABLET | 43975-0349-03 | 0.97761 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for PALIPERIDONE ER
What is PALIPERIDONE ER?
Paliperidone ER (extended release) is an atypical antipsychotic used primarily to treat schizophrenia and schizoaffective disorder. It is a once-daily oral formulation that delivers the active metabolite of risperidone.
Current Market Position
Paliperidone ER is marketed by Janssen Pharmaceuticals since its FDA approval in 2009. It competes directly with drugs like risperidone, risperidone-long acting injectable, aripiprazole, quetiapine, and other atypical antipsychotics.
Market Size and Growth
Global Schizophrenia Treatment Market
The schizophrenia pharmaceutical market was valued at approximately USD 4.2 billion in 2022 and is projected to grow at a CAGR of 3.8% through 2030[1].
Key Factors Influencing the Market
- Rising prevalence of schizophrenia (approximately 1 in 100 worldwide)
- Increased acceptance of atypical antipsychotics
- Patent expiry of some competing drugs (risperidone, olanzapine)
- Emerging markets adopting newer medications
Competitive Landscape
| Drug Class | Major Products | Market Share (2022) | Patent Status |
|---|---|---|---|
| Atypical Antipsychotics | Paliperidone ER, risperidone, aripiprazole | 60% | Active patents (Paliperidone ER) |
| Typical Antipsychotics | Haloperidol, chlorpromazine | 15% | Off-patent |
| Long-acting Injectables | Risperidone, paliperidone palmitate | 25% | Patents expiring 2024-2028 |
Pricing Overview
Current pricing varies across geographies, with the U.S. marketplace presenting the most detailed data.
U.S. Pricing (2023)
- Brand Name (Invega ER): Approx. USD 850-1000 per month for a typical dose (6 mg daily)
- Generic Pricing: Not available yet, but expected to decrease to USD 600-700 upon biosimilar entry, likely in 2025-2026
Cost Per Dose
| Dose (mg) | Approximate Monthly Cost | Cost per Tablet (USD) |
|---|---|---|
| 3 | 850 | 28 |
| 6 | 950 | 32 |
| 9 | 1050 | 35 |
Prices are based on wholesale acquisition costs with variation depending on pharmacy and insurance rebates.
Price Projections (2024-2030)
Assumptions
- Patent expiry for the innovator drug in 2024; biosimilar competition emerges shortly after
- Patent litigation may delay biosimilar market entry until 2025-2026
- Market penetration of biosimilars expected to be 30-50% within five years of entry
- Pricing discounts for biosimilars anticipated at 30-50%
Projected Price Trends
| Year | Brand Price (USD/month) | Biosimilar Price (USD/month) | Market Share (biosimilar) |
|---|---|---|---|
| 2024 | 850-1000 | Not available | 0% |
| 2025 | 820-950 | 600-700 | 10-20% |
| 2026 | 780-900 | 550-650 | 30-50% |
| 2027 | 750-870 | 520-620 | 50-60% |
| 2028 | 700-850 | 500-600 | 60-70% |
| 2029 | 680-820 | 480-580 | 70-80% |
| 2030 | 650-800 | 460-560 | 80-90% |
Revenue Implications
- U.S. peak branded sales could decline by 20-30% post-biosimilar entry due to price reductions.
- Global markets may experience slower price erosion due to different patent laws and reimbursement policies.
Key Drivers and Risks
Drivers
- Increasing schizophrenia prevalence
- Cost-containment measures favoring biosimilars
- Continued differentiation through formulations and delivery systems
Risks
- Patent litigation delays
- Biosimilar quality perception issues
- Market resistance to price reductions
- Regulatory delays in emerging markets
Summary of Price Volatility and Opportunities
| Period | Opportunity | Risk |
|---|---|---|
| Pre-2024 | Maintain premium pricing | Patent risk, generic competition |
| 2024-2026 | Market share gains for biosimilars | Price erosion, slower uptake |
| 2027+ | Stabilization at lower price point | Pricing war, regulatory hurdles |
Key Takeaways
- Paliperidone ER commands high prices in major markets, with USD 850-1,000 monthly in the U.S.
- Patent expiry expected in 2024 opens the biosimilar market, likely reducing prices by 30-50%.
- Biosimilar entry is projected to lead to significant market share shifts by 2026, with prices declining accordingly.
- The overall market is poised for moderate growth, driven by rising schizophrenia prevalence and adoption of novel formulations.
- Companies should monitor patent litigation and biosimilar approvals as key risk factors influencing future pricing.
FAQs
1. When is patent expiry for Paliperidone ER?
Expected around 2024, with ongoing litigation potentially delaying biosimilar entry until 2025-2026.
2. What is the expected price of biosimilars upon entry?
Biosimilars are projected to be priced at 30-50% less than the brand, around USD 520-650 per month initially.
3. How will biosimilar competition affect revenue?
Initially, a temporary revenue decline; long-term, increased market penetration may stabilize overall sales volume.
4. Are there regulatory hurdles in emerging markets?
Yes; patent protections and regulatory pathways vary, affecting biosimilar launch timelines and prices.
5. What factors could accelerate price declines?
Patent challenges favoring early biosimilar approval, regulatory approvals in multiple jurisdictions, and market acceptance.
References
- Grand View Research. (2023). Schizophrenia Treatment Market Size, Share & Trends Analysis. Retrieved from https://www.grandviewresearch.com/industry-analysis/schizophrenia-treatment-market
More… ↓
