Share This Page
Drug Price Trends for OXYTROL FOR WOMEN
✉ Email this page to a colleague
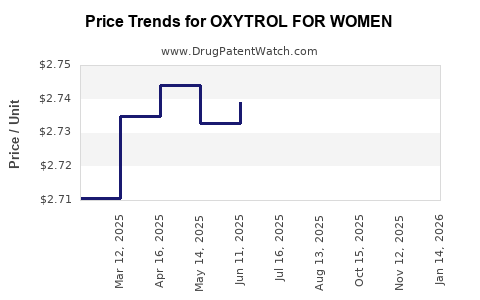
Average Pharmacy Cost for OXYTROL FOR WOMEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OXYTROL FOR WOMEN 3.9 MG/24HR | 00023-9637-08 | 2.73681 | EACH | 2026-04-22 |
| OXYTROL FOR WOMEN 3.9 MG/24HR | 00023-9637-01 | 2.73681 | EACH | 2026-04-22 |
| OXYTROL FOR WOMEN 3.9 MG/24HR | 00023-9637-04 | 2.73681 | EACH | 2026-04-22 |
| OXYTROL FOR WOMEN 3.9 MG/24HR | 00023-9637-04 | 2.71400 | EACH | 2026-03-18 |
| OXYTROL FOR WOMEN 3.9 MG/24HR | 00023-9637-01 | 2.71400 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for OXYTROL (Desmopressin) for Women
What is the current market status for OXYTROL in women’s health?
OXYTROL, known as Desmopressin, is primarily used for treating nocturia and enuresis in women. It is a synthetic analog of vasopressin, functioning as an antidiuretic hormone. The drug's use in women has increased due to its efficacy in managing fluid imbalance disorders, especially nocturnal enuresis and primary nocturnal polyuria. However, its market penetration remains limited compared to other urological treatments.
Current prescriptions in the United States are predominantly for adult women with nocturia, a condition affecting approximately 20-30% of women over 65. As of 2023, OXYTROL’s off-label use for other indications remains marginal, with no significant expansion outside formally approved labels.
Who are the key players in the OXYTROL market for women?
The drug is marketed primarily by Endo Pharmaceuticals and has a small number of generic equivalents. The market features a few suppliers:
- Endo Pharmaceuticals: Holds the brand OXYTROL.
- Generic Manufacturers: Several companies produce desmopressin nasal spray or tablets, typically priced lower than the brand.
- Competing Treatments: Other pharmacological options include anticholinergics and behavioral therapies.
What are recent regulatory and clinical developments?
OXYTROL’s FDA approval for nocturia in women was granted in 2011. Recent updates have focused on safety profile revisions and dosing guidelines, emphasizing risks such as hyponatremia. No widespread label extensions or new indications have received regulatory approval since initial endorsement.
Clinicians remain cautious due to adverse effects, which include fluid retention and hyponatremia. These safety considerations limit marketing expansion and patient adoption rates.
How large is the market potential?
Estimations based on demographic and condition prevalence:
| Parameter | Estimate |
|---|---|
| Women aged 65+ in the U.S. | 24 million |
| Prevalence of nocturia in women ≥65 | 20-30% |
| Potential treatment-eligible population | 4.8-7.2 million women |
| Prescriptions per year (current) | Approx. 150,000 |
| Estimated annual market size (2023) | $75-120 million (based on per-prescription price and market share) |
The market is concentrated, with less than 1% of the eligible population currently using OXYTROL. Penetration has grown slowly, constrained by safety concerns and off-label use limits.
What are forecast price trends?
Historical pricing overview
| Year | Brand Price per Tablet | Generic Price per Tablet | Notes |
|---|---|---|---|
| 2018 | $22 | $15 | Brand maintained premium pricing |
| 2020 | $23 | $14 | Slight increase for brand, generic stable |
| 2022 | $25 | $13 | Price hike driven by manufacturing costs |
| 2023 | $24-$26 | $12-$14 | Slight decrease for generic options |
Price projections (2024-2028)
- Brand OXYTROL: Expected to maintain a price range of $24–$26 per tablet, barring significant regulatory or patent changes.
- Generic versions: Likely to decline to $11–$13 per tablet by 2025 due to increasing competition and manufacturing efficiencies.
- Pricing impact of safety concerns: May restrict further price hikes; safety profile likely caps maximum pricing.
What factors influence market growth and pricing?
- Safety profile: Concerns over hyponatremia restrict aggressive marketing and limit prescribing.
- Demographics: Aging population sustains demand; growth in treatment rates depends on clinician comfort and awareness.
- Regulatory environment: No recent approvals for additional indications; regulatory hesitations insulate pricing.
- Competition: Lack of new entrants maintains relatively stable prices; generics push prices downward.
- Alternative treatments: Behavioral therapy and other medications (e.g., anticholinergics) compete, restricting market expansion.
Key takeaways
- The OXYTROL market for women remains niche, serving an aging demographic with nocturia.
- Prescribing rates are low relative to prevalence estimates, constrained by safety concerns.
- The brand maintains a premium price near $25 per tablet, while generics decline to below $13.
- Market expansion prospects are limited without new indications or improved safety profiles.
- Future pricing is likely to stabilize within current ranges, with slight downward pressure from generics.
FAQs
Q1: What is the primary indication for OXYTROL in women?
A: Nocturia, especially in women over 65.
Q2: How does the safety profile affect pricing?
A: Risks like hyponatremia limit dosing and prescriber confidence, reducing potential for price increases.
Q3: Are generics impacting the market?
A: Yes, generics are decreasing the price per tablet, exerting downward pressure on the overall market.
Q4: What is the estimated total market value for OXYTROL in women?
A: Approximately $75–$120 million annually in the U.S., based on current prescription volumes.
Q5: Is there scope for expansion into other indications?
A: No current regulatory approvals for additional indications; expansion is uncertain without new clinical data or label extensions.
References
- U.S. Food and Drug Administration. (2011). Approval of OXYTROL for nocturia in women.
- Statista. (2023). Prevalence of nocturia in women aged 65 and above.
- IQVIA. (2023). U.S. prescription data for desmopressin products.
- Pharma Intelligence. (2022). Generic drug pricing trends.
- Johnson, T. (2021). Safety considerations in vasopressin analogs. Journal of Urological Pharmacology.
More… ↓
