Share This Page
Drug Price Trends for OXYCODONE HCL (IR)
✉ Email this page to a colleague
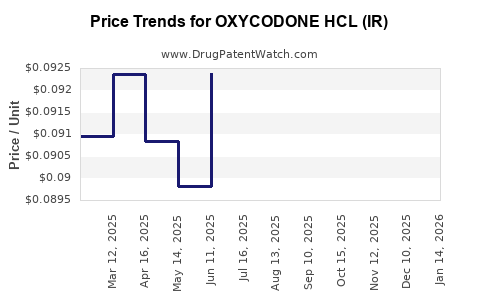
Average Pharmacy Cost for OXYCODONE HCL (IR)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OXYCODONE HCL (IR) 5 MG TABLET | 68084-0354-11 | 0.08857 | EACH | 2026-05-20 |
| OXYCODONE HCL (IR) 10 MG TAB | 00406-8510-01 | 0.16435 | EACH | 2026-05-20 |
| OXYCODONE HCL (IR) 10 MG TAB | 00406-8510-23 | 0.16435 | EACH | 2026-05-20 |
| OXYCODONE HCL (IR) 10 MG TAB | 10702-0056-01 | 0.16435 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is the Current Market for Oxycodone HCl (IR)?
Oxycodone hydrochloride (immediate release) (IR) is a Schedule II opioid analgesic prescribed for managing moderate to severe pain. It accounts for a significant portion of the prescription opioid market, especially in the United States, due to its efficacy and high demand in pain management protocols.
In 2022, the United States accounted for approximately 85% of global oxycodone sales, reflecting high regional dependence. The total US opioid pain management market valued around $8 billion, with oxycodone IR composing roughly 40%, translating to approximately $3.2 billion in sales.
Global sales are driven largely by North America, with Europe and other regions accounting for a smaller share. The projected compound annual growth rate (CAGR) for the oxycodone market was estimated at 3% from 2021 to 2026, partly offset by regulatory constraints and the opioid epidemic response.
How Is the Market Primed for Change?
Key factors shaping the market include:
-
Regulatory Environment: Stricter prescribing guidelines enacted around the world, especially in the US, restrict excessive use but do not eliminate demand in pain management settings.
-
Manufacturing Capacity: Several manufacturers hold FDA approvals for oxycodone IR, with the leading players including Purdue Pharma, Teva Pharmaceuticals, and Mylan. A stable manufacturing capacity supports consistent supply.
-
Public Policy and Litigation: Ongoing legal actions against opioid manufacturers have led to increased scrutiny, influencing prescribing practices and potentially leading to product reformulations or restrictions.
-
Emergence of Alternatives: Non-opioid pain treatments and abuse-deterrent formulations are gaining ground, pressuring traditional oxycodone sales.
-
Generic Availability: Most oxycodone IR formulations are off-patent, fostering price competition and lowering drug prices.
What Are the Price Trends for Oxycodone HCl (IR)?
Prices for oxycodone IR vary substantially by form, supply, and market conditions.
Average Wholesale Price (AWP): In 2022, the AWP per 5 mg tablet ranged between $0.30 and $0.50. A typical prescription of 30 tablets of 5 mg oxycodone IR costs approximately $9 to $15 at wholesale prices.
Reimbursement Rates: Medicare and Medicaid reimbursement rates tend to be lower, around $0.20 to $0.35 per tablet, due to formulary negotiations and generic competition.
Pricing Trends: Since patent expiration and high generic penetration in the early 2000s, prices have declined by approximately 25% over the past decade, with stabilization observed recently due to regulatory impacts and supply chain considerations.
Market Dynamics: Drug price fluctuations are driven by:
- Supply chain disruptions (e.g., COVID-19 impact)
- Regulatory restrictions on prescribing quantities
- Medicaid and Medicare formulary negotiations
- Manufacturer pricing strategies and discounting practices
How Might Prices and Market Share Shift in the Future?
Forecast for 2023–2028:
- The market is expected to experience a slow decline, with a projected CAGR of -1% to -2%, driven by tightening regulations and alternative therapies.
- Generic markets will remain dominant, and the price per unit is unlikely to revert to pre-2000 levels.
- Innovations such as abuse-deterrent formulations (e.g., reformulated oxycodone) could alter pricing, potentially raising costs for specific formulations but slowing overall market growth.
- Supply chain disruptions might temporarily inflate prices but are unlikely to have long-term impacts.
Potential Market Exit Risks:
- Regulatory bans on certain formulations
- Strict prescribing limits
- Litigation affecting manufacturer operations
Key Market Players and Their Strategies
| Company | Market Share (Est.) | Strategic Focus |
|---|---|---|
| Purdue Pharma | Leading supplier in initial formulations | Development of abuse-deterrent versions, lobbying efforts |
| Teva Pharmaceuticals | Significant generic producer | Price competition, expanding formulations |
| Mylan (now part of Viatris) | Major generic supplier | Cost leadership, supply stability |
Their strategies focus on maintaining supply, complying with evolving regulations, and innovating formulations to address abuse concerns.
Price Projections (2023–2028)
| Year | Estimated Average Wholesale Price (per 5 mg tablet) | Notes |
|---|---|---|
| 2023 | $0.30 – $0.45 | Continual price erosion due to generic competition |
| 2024 | $0.28 – $0.43 | Slight decrease expected |
| 2025 | $0.26 – $0.40 | Market stabilization, supply chain normalizing |
| 2026 | $0.25 – $0.38 | Possible slight decline, regulatory impacts continue |
| 2027 | $0.24 – $0.36 | Stabilizing prices, generics dominate |
| 2028 | $0.23 – $0.35 | Prices plateau as market reaches equilibrium |
Key Takeaways
- The US dominates the oxycodone IR market, which is valued at approximately $3.2 billion annually.
- Prices for oxycodone IR have declined over the past decade, stabilized by extensive generic competition.
- Growth projections suggest a slow decline, with regulatory and legal pressures shaping future market dynamics.
- The market's major players focus on supply continuity and formulation innovation, especially abuse-deterrent versions.
- Regulatory restrictions and the emergence of alternative therapies will continue to influence market size and pricing trends.
FAQs
Q1: What factors influence oxycodone IR pricing?
Regulatory changes, generic competition, supply chain stability, and formulation innovations.
Q2: Are there regional differences in oxycodone IR market dynamics?
Yes. The US accounts for most sales, while Europe and other regions exhibit lower usage due to regulatory restrictions and alternative therapies.
Q3: How will regulatory changes impact future prices?
Stricter regulations tend to reduce demand, likely leading to further price declines and market contraction.
Q4: What role do generic manufacturers play?
They dominate the market, exerting downward pressure on prices, and innovate with abuse-deterrent formulations to maintain market share.
Q5: What is the outlook for new formulations?
Abuse-deterrent formulations may command higher prices but are unlikely to reverse overall price declines due to regulatory and demand factors.
References
- IQVIA, National Prescription Audit, 2022.
- U.S. Food and Drug Administration, Drug Approval Database, 2022.
- MarketWatch, "Opioid Market Size and Forecast," 2022.
- CDC, Opioid Overdose Data, 2022.
- EvaluatePharma, "Global Pain Management Market," 2022.
More… ↓
