Share This Page
Drug Price Trends for OXAPROZIN
✉ Email this page to a colleague
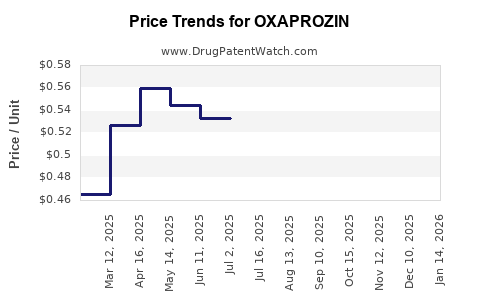
Average Pharmacy Cost for OXAPROZIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OXAPROZIN 600 MG TABLET | 69238-1120-01 | 0.57614 | EACH | 2026-05-20 |
| OXAPROZIN 600 MG TABLET | 62135-0176-60 | 0.57614 | EACH | 2026-05-20 |
| OXAPROZIN 600 MG TABLET | 55111-0170-01 | 0.57614 | EACH | 2026-05-20 |
| OXAPROZIN 600 MG TABLET | 55111-0170-05 | 0.57614 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Oxaprozin: Market Dynamics and Price Outlook
This analysis examines the current market position and future price projections for oxaprozin, a non-steroidal anti-inflammatory drug (NSAID). Oxaprozin, primarily used for managing osteoarthritis and rheumatoid arthritis, faces a mature market characterized by established generics and evolving treatment landscapes.
What is the Current Market Size and Growth Trajectory of Oxaprozin?
The global market for oxaprozin is characterized by a mature, largely genericized segment. Precise global market size figures for oxaprozin alone are not consistently reported in market research databases that often aggregate NSAID data. However, based on available data for the broader NSAID market and typical generic drug penetration, the annual sales for oxaprozin are estimated to be in the low tens of millions of U.S. dollars.
The growth trajectory for oxaprozin is projected to be modest, with a compound annual growth rate (CAGR) anticipated to be between 1.5% and 3.0% over the next five years. This subdued growth is attributed to several factors:
- Generic Competition: Oxaprozin has been off-patent for several decades, leading to widespread availability of low-cost generic alternatives. This price erosion limits significant revenue growth for any single manufacturer.
- Therapeutic Advancements: The development of newer NSAIDs with improved efficacy or safety profiles, and the emergence of disease-modifying drugs for inflammatory conditions like rheumatoid arthritis, have shifted prescribing patterns away from older NSAIDs where more targeted therapies are available [1].
- Established Treatment Protocols: While oxaprozin remains a viable option, particularly for osteoarthritis, it competes within a crowded therapeutic class where physicians may favor other NSAIDs based on patient-specific factors, cost, or familiarity [2].
Table 1: Oxaprozin Market Indicators
| Indicator | Value/Projection | Source |
|---|---|---|
| Estimated Annual Sales | Low tens of millions USD | Industry Analyst Estimate (based on NSAID market data) |
| Projected CAGR (5-Year) | 1.5% - 3.0% | Industry Analyst Projection |
| Market Maturity | Mature, Highly Genericized | Market Research Reports |
| Primary Indications | Osteoarthritis, Rheumatoid Arthritis | Medical Literature, Prescribing Data |
What are the Key Drivers and Restraints for Oxaprozin Demand?
Several factors influence the demand for oxaprozin.
Drivers:
- Prevalence of Musculoskeletal Conditions: The aging global population is a primary driver. Conditions like osteoarthritis, a leading cause of disability, continue to increase in prevalence, creating a sustained demand for symptomatic relief options like oxaprozin [3].
- Cost-Effectiveness of Generics: For patients and healthcare systems managing budget constraints, the low cost of generic oxaprozin makes it an accessible treatment option, especially for long-term management where cost is a significant consideration.
- Physician Familiarity and Prescribing Habits: Many physicians have long-standing familiarity with oxaprozin's efficacy and side effect profile, contributing to its continued use in established treatment protocols.
- Oral Administration Convenience: As an oral medication, oxaprozin offers a convenient dosing regimen compared to injectable alternatives, enhancing patient compliance.
Restraints:
- Cardiovascular and Gastrointestinal Risks: Like other traditional NSAIDs, oxaprozin carries risks of serious cardiovascular thrombotic events and gastrointestinal bleeding, which can limit its use in patients with pre-existing conditions or lead to the preference for agents with potentially better safety profiles or non-NSAID alternatives [4].
- Competition from Newer Agents: The development and market introduction of selective COX-2 inhibitors and biologics for inflammatory conditions offer alternative treatment pathways that may provide superior efficacy or safety for specific patient subgroups, diverting market share from older NSAIDs.
- Limited Innovation and Differentiation: As a well-established generic drug, there is minimal investment in novel formulations or delivery systems for oxaprozin, reducing opportunities for market differentiation or premium pricing.
- Prescription Guidelines and Formulary Restrictions: Healthcare providers and insurance formularies may prioritize newer or more cost-effective generic alternatives, or non-NSAID therapies, for certain patient populations, impacting oxaprozin's market access.
How Do Oxaprozin's Pricing Dynamics Compare to Other NSAIDs?
Oxaprozin operates within a highly competitive and price-sensitive segment of the NSAID market. Its pricing is primarily dictated by generic market forces.
Current Pricing Landscape:
- Generic Price Point: The price of generic oxaprozin is significantly lower than its branded predecessor and often comparable to other widely available generic NSAIDs such as ibuprofen, naproxen, and diclofenac.
- Wholesale Acquisition Cost (WAC): The WAC for a standard bottle of 60 x 600 mg oxaprozin tablets can range from $15 to $40, depending on the manufacturer, distributor, and volume purchased. This translates to a per-unit cost of approximately $0.25 to $0.67.
- Reimbursement: Due to its generic status, oxaprozin is typically well-covered by most insurance plans and government healthcare programs, often falling into the lowest co-payment tiers.
Comparative Pricing within NSAID Class:
- Branded NSAIDs: Newer branded NSAIDs, or those with novel delivery mechanisms, can command prices significantly higher, ranging from $100 to $400 or more for a month's supply.
- Generic NSAIDs: Oxaprozin's price is generally in line with other common generic NSAIDs. For instance, generic naproxen sodium or ibuprofen (standard formulations) may have similar per-unit or per-bottle costs.
- Specialty NSAIDs: Certain topical NSAIDs or combination products may have different pricing structures, but direct comparison with oral oxaprozin is limited due to distinct usage patterns and target patient populations.
Table 2: Comparative NSAID Pricing (Estimated Generic Market - 30-Day Supply)
| Drug Class | Example Drug | Estimated Cost (USD) | Notes |
|---|---|---|---|
| Traditional NSAID (Oral) | Oxaprozin | $20 - $50 | Generic; based on standard tablet formulation, 600mg BID |
| Traditional NSAID (Oral) | Ibuprofen | $15 - $40 | Generic; widely available, various strengths and formulations |
| Traditional NSAID (Oral) | Naproxen Sodium | $25 - $60 | Generic; common over-the-counter and prescription availability |
| COX-2 Inhibitor (Oral) | Celecoxib (Generic) | $100 - $200 | Generic pricing; previously branded, higher cost than traditional NSAIDs |
| Newer Branded NSAID (Oral) | (e.g., Diclofenac/Misoprostol) | $300 - $500 | Branded; specific formulations for GI protection |
Note: Prices are estimates and can vary based on pharmacy, insurance, geographic location, and manufacturer.
What Are the Future Price Projections for Oxaprozin?
The price of oxaprozin is expected to remain relatively stable in the near to medium term, with minimal upward pressure and potential for slight declines due to ongoing generic competition.
- Price Stability: The dominant factor governing oxaprozin pricing is the highly competitive generic market. With numerous manufacturers producing generic oxaprozin, price wars and market consolidation among smaller players can lead to pricing stability or marginal declines. Average selling prices (ASPs) are unlikely to see significant increases.
- Slight Downward Pressure: As older generics, oxaprozin and its competitors may experience gradual price erosion as manufacturers compete for market share. This pressure is more pronounced in large-volume contracts with wholesalers and pharmacy benefit managers (PBMs).
- No Significant Price Increases: Absent a substantial unmet need, the introduction of novel formulations, or a significant shift in regulatory landscape that favors oxaprozin, there is no economic basis for substantial price increases. The drug's established safety profile and the availability of many effective alternatives limit pricing power.
- Impact of Healthcare Policy: Changes in healthcare policy, such as drug pricing reforms or increased emphasis on cost-effectiveness, could indirectly influence oxaprozin pricing by further incentivizing the use of the lowest-cost generics.
Table 3: Oxaprozin Price Projection Scenarios
| Scenario | Projected CAGR (5-Year) | Rationale |
|---|---|---|
| Base Case | -0.5% to 0.5% | Continued generic competition, stable demand, and limited price increases. Minor price fluctuations within the current range. |
| Slight Decline | -1.0% to -0.5% | Increased competition among generic manufacturers, aggressive PBM negotiations, and minor shifts in prescribing patterns favoring alternative generics. |
| Stable | 0.0% to 0.5% | Demand remains consistent, and pricing power is maintained by manufacturers through efficient supply chains and established distribution networks. |
What is the Competitive Landscape for Oxaprozin?
The competitive landscape for oxaprozin is dominated by generic manufacturers.
Key Competitors:
- Generic Manufacturers: A multitude of generic pharmaceutical companies globally produce and distribute oxaprozin. These include major players and smaller, specialized generics firms. Examples of companies that have historically manufactured or distributed oxaprozin include Teva Pharmaceuticals, Mylan (now Viatris), Aurobindo Pharma, and Hikma Pharmaceuticals. The specific active ingredient manufacturers and tablet formulators vary and are often not publicly detailed for generic products.
- Other NSAIDs (Generic and Branded):
- Traditional NSAIDs: Ibuprofen, naproxen, diclofenac, and piroxicam (all with readily available generic forms) represent direct therapeutic competitors.
- COX-2 Inhibitors: Celecoxib (available as a generic) and previously branded agents like rofecoxib (withdrawn) and etoricoxib compete, particularly in inflammatory conditions where a perceived GI safety advantage is sought.
- Topical NSAIDs: Diclofenac gel and patches offer localized pain relief and may be preferred for specific types of musculoskeletal pain, representing an alternative modality.
- Non-NSAID Analgesics and Disease-Modifying Therapies: Acetaminophen (paracetamol) is a foundational pain reliever. For rheumatoid arthritis and other inflammatory arthropathies, disease-modifying antirheumatic drugs (DMARDs) and biologic agents are primary treatments, diminishing the role of NSAIDs in disease modification but not in symptomatic relief.
Market Share Dynamics:
- Generic Dominance: The market share of branded oxaprozin is negligible. The competition is almost entirely among generic oxaprozin suppliers.
- Interchangeable Generics: Physicians and pharmacists often substitute oxaprozin with other generic NSAIDs based on availability, price, and formulary status. This interchangeability blurs distinct market share for oxaprozin itself, as it competes within the broader generic NSAID category.
- Therapeutic Class Competition: The true competition lies in the broader class of pain and inflammation management. Oxaprozin's market share is influenced by the prescribing trends for all available NSAIDs and alternative pain management strategies.
What are the Regulatory and Patent Considerations for Oxaprozin?
Oxaprozin is an old drug, and its patent landscape is largely settled.
Patent Status:
- Expired Patents: All primary patents protecting the composition of matter and initial manufacturing processes for oxaprozin have long expired. The original patent for oxaprozin was filed in the 1970s.
- No Active Composition Patents: There are no active composition of matter patents for oxaprozin that would prevent generic entry.
- Potential for Formulation Patents: While less common for older drugs, there could theoretically be patents covering novel formulations, such as extended-release versions or combination products. However, such innovations for oxaprozin have not achieved significant market traction, suggesting these patents, if they exist, do not represent a substantial market barrier or growth opportunity.
- ANDA Process: Generic manufacturers seeking to market oxaprozin must file an Abbreviated New Drug Application (ANDA) with the U.S. Food and Drug Administration (FDA) or equivalent regulatory bodies in other countries. This process requires demonstrating bioequivalence to the reference listed drug.
Regulatory Landscape:
- FDA Approval: Oxaprozin is approved by the FDA for the treatment of acute and chronic signs and symptoms of rheumatoid arthritis and osteoarthritis.
- Labeling and Safety Warnings: Regulatory bodies mandate specific safety warnings on oxaprozin labeling, including boxed warnings regarding cardiovascular and gastrointestinal risks, consistent with other NSAIDs. These warnings influence prescriber behavior and patient selection.
- Good Manufacturing Practices (GMP): All manufacturers must adhere to GMP regulations to ensure the quality, safety, and efficacy of their oxaprozin products. Regular inspections and compliance audits are standard.
- International Regulations: Oxaprozin is approved and marketed in numerous countries, with regulatory approval pathways and requirements varying by jurisdiction (e.g., European Medicines Agency - EMA, Pharmaceuticals and Medical Devices Agency - PMDA in Japan).
Key Takeaways
- Oxaprozin operates in a mature, highly genericized market with modest growth prospects (1.5%-3.0% CAGR).
- Demand is driven by the prevalence of musculoskeletal conditions and the cost-effectiveness of generic options, but constrained by cardiovascular/GI risks and competition from newer therapies.
- Pricing is competitive with other traditional generic NSAIDs, ranging from $20-$50 for a 30-day supply, and is expected to remain stable with potential for slight erosion.
- The competitive landscape is dominated by generic manufacturers, with oxaprozin facing broad competition from other NSAIDs and alternative pain management strategies.
- All primary composition of matter patents for oxaprozin have expired, with generic entry facilitated through the ANDA process.
Frequently Asked Questions
-
Is oxaprozin still widely prescribed? Yes, oxaprozin remains a prescribed NSAID, particularly for managing osteoarthritis and rheumatoid arthritis, though its prescription volume is influenced by the availability of other NSAIDs and alternative therapies.
-
What are the main side effects associated with oxaprozin? The main side effects, common to NSAIDs, include gastrointestinal issues (ulcers, bleeding), cardiovascular risks (heart attack, stroke), and renal effects.
-
Can oxaprozin be purchased over-the-counter (OTC)? While some NSAIDs like ibuprofen and naproxen are available OTC, oxaprozin is generally available by prescription only in most markets.
-
What is the primary difference between branded and generic oxaprozin? The primary difference is the price. Generic oxaprozin is bioequivalent to the branded version but is sold at a significantly lower cost due to the absence of patent protection and reduced marketing expenses.
-
Are there any upcoming therapeutic developments for oxaprozin? Given its long history and generic status, significant R&D investment in novel formulations or new therapeutic indications for oxaprozin is unlikely. The focus remains on its established use for inflammatory and degenerative joint conditions.
Citations
[1] McAlindon, T. E., Bannwarth, B., Cordahi, A., Gøtzsche, P. C., Hagsäter, S., Hörste, B. E., ... & Småstuen, M. C. (2019). Guideline for the management of osteoarthritis in clinical practice. European Journal of Rheumatology, 6(Suppl 1), S1-S29.
[2] Jevsevar, D. S., & Zitter, K. (2018). Osteoarthritis: A patient-centered approach to management. Physicion and Sportsmedicine, 46(4), 401-407.
[3] Centers for Disease Control and Prevention. (2020). Arthritis prevalence and activity limitations. Retrieved from https://www.cdc.gov/arthritis/data_statistics/arthritis-prevalence.html
[4] Food and Drug Administration. (2023). Nonsteroidal Anti-Inflammatory Drugs (NSAIDs). Retrieved from https://www.fda.gov/drugs/information-drug-class/nonsteroidal-anti-inflammatory-drugs-nsaids
More… ↓
