Share This Page
Drug Price Trends for OVIDREL
✉ Email this page to a colleague
Average Pharmacy Cost for OVIDREL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
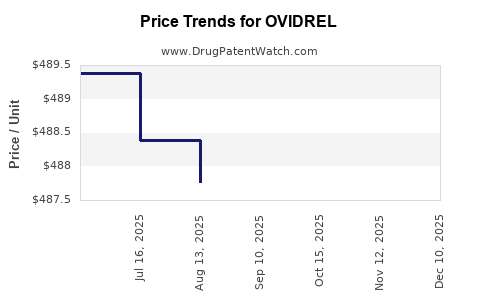
| OVIDREL 250 MCG/0.5 ML SYRG | 44087-1150-01 | 486.64581 | ML | 2026-04-22 |
| OVIDREL 250 MCG/0.5 ML SYRG | 44087-1150-01 | 487.04786 | ML | 2026-03-18 |
| OVIDREL 250 MCG/0.5 ML SYRG | 44087-1150-01 | 487.06250 | ML | 2025-12-17 |
| OVIDREL 250 MCG/0.5 ML SYRG | 44087-1150-01 | 487.35636 | ML | 2025-11-19 |
| OVIDREL 250 MCG/0.5 ML SYRG | 44087-1150-01 | 487.33600 | ML | 2025-10-22 |
| OVIDREL 250 MCG/0.5 ML SYRG | 44087-1150-01 | 487.13895 | ML | 2025-09-17 |
| OVIDREL 250 MCG/0.5 ML SYRG | 44087-1150-01 | 487.77385 | ML | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Ovidrel Market Analysis and Price Projections
Ovidrel, the brand name for choriogonadotropin alfa (recombinant human chorionic gonadotropin), is a gonadotropin-releasing hormone analog used in assisted reproductive technologies (ART). Its market performance is directly tied to the global demand for in vitro fertilization (IVF) and ovulation induction procedures. Price projections are influenced by patent exclusivity, generic competition, manufacturing costs, and reimbursement policies.
What is Ovidrel's Current Market Position?
Ovidrel is a leading product in the gonadotropin market for ART. Its primary indication is to induce ovulation and luteal phase support in women undergoing controlled ovarian stimulation. It is also used in conjunction with controlled ovarian stimulation to initiate the final maturation of follicles and trigger ovulation in women undergoing assisted reproductive technology. The drug's efficacy and established safety profile contribute to its strong market presence.
Key market drivers include:
- Increasing Infertility Rates: Global infertility rates are rising due to factors such as delayed childbearing, lifestyle changes, and environmental influences.
- Advancements in ART: Continuous improvements in IVF and other ART techniques enhance success rates, increasing demand for fertility medications.
- Growing Awareness and Accessibility: Greater public awareness of fertility treatments and expanding healthcare access, particularly in emerging markets, contribute to market growth.
- Reimbursement Policies: Favorable reimbursement policies for ART procedures in certain regions can stimulate demand for associated medications.
The global market for fertility drugs, which includes Ovidrel and other gonadotropins, was valued at approximately \$4.1 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of around 5% to 7% from 2024 to 2030. This growth is primarily driven by the aforementioned factors.
Ovidrel's Competitive Landscape
Ovidrel competes with other gonadotropin products, including those derived from urinary sources (human chorionic gonadotropin - hCG) and other recombinant hCG analogs.
| Competitor Product | Active Ingredient | Manufacturer | Primary Formulation |
|---|---|---|---|
| Ovidrel | choriogonadotropin alfa | Merck KGaA | Recombinant hCG |
| Pregnyl | human chorionic gonadotropin | Ferring Pharmaceuticals | Urinary hCG |
| Novarel | human chorionic gonadotropin | Ferring Pharmaceuticals | Urinary hCG |
| Gonal-f | follitropin alfa | Merck KGaA | Recombinant FSH |
| Bravelle | urofollitropin | Braeburn Pharmaceuticals | Urinary FSH |
Ovidrel's recombinant nature offers advantages such as a standardized and consistent product, potentially reducing batch-to-batch variability compared to urinary-derived products. This consistency is a critical factor in clinical application.
What is the Patent Status of Ovidrel?
Merck KGaA, through its acquisition of EMD Serono, holds the primary patents related to Ovidrel. The original U.S. patent for recombinant human chorionic gonadotropin (which Ovidrel is based upon) was filed in the early 1990s. While the core composition of matter patents have expired, there may be secondary patents covering manufacturing processes, specific formulations, or methods of use that still provide some level of protection.
For example, patents related to the expression systems or purification methods used to produce recombinant hCG could still be in force. However, these secondary patents typically do not prevent the development of bioequivalent generic versions. The expiration of key patents is a significant factor that opens the door for generic competition.
The introduction of generic versions of choriogonadotropin alfa is a primary driver for price erosion in the market. Once a bioequivalent generic product receives regulatory approval, it can significantly alter the market dynamics.
Patent Expiration Impact on Ovidrel
The initial patent protection for Ovidrel has largely expired, paving the way for generic market entry. The first generic versions of choriogonadotropin alfa began appearing in markets where patent exclusivity lapsed. This has led to a demonstrable downward pressure on Ovidrel's price.
As of early 2024, several generic versions of choriogonadotropin alfa have gained regulatory approval in major markets like the United States and the European Union. These generics are typically offered at substantially lower price points than the branded Ovidrel.
What are Ovidrel's Price Trends and Projections?
Ovidrel's pricing has historically been at a premium due to its branded status, proprietary manufacturing process, and the established trust in its brand. The average wholesale price (AWP) for Ovidrel (e.g., 250 mcg/0.5 mL prefilled syringe) has typically ranged from \$75 to \$150 per dose, depending on the region, pharmacy, and negotiated payer contracts.
However, the landscape is shifting. The entry of generic choriogonadotropin alfa has initiated a significant price decline for the active pharmaceutical ingredient (API) and the finished drug product.
Price Trend Analysis:
- Pre-Generic Entry: Ovidrel's price remained relatively stable, reflecting its market dominance and patent protection.
- Post-Generic Entry (2020 onwards in key markets): A decline of 20% to 50% in the average selling price (ASP) for choriogonadotropin alfa products has been observed. This decline is more pronounced for the generic alternatives but also impacts Ovidrel as a reference point.
Factors Influencing Future Pricing:
- Generic Market Share: As generic choriogonadotropin alfa products gain greater market share, the pricing power of branded Ovidrel will diminish further.
- Manufacturing Costs: The cost of producing recombinant proteins can be substantial. However, efficiencies in manufacturing, especially for generic producers, can lead to lower overall costs.
- Reimbursement Landscape: Payers (insurance companies, national health services) are increasingly favoring cost-effective generic options, which can influence prescribing patterns and pricing negotiations for branded products.
- Competition Intensity: The number of generic manufacturers in the market will directly impact price competition. A fragmented generic market with multiple suppliers generally leads to lower prices.
- Regulatory Environment: Approval timelines and pricing regulations in different countries can affect the speed and extent of price changes.
Price Projections:
By 2026, the average selling price for Ovidrel is projected to be 30% to 60% lower than its peak pricing pre-generic entry, assuming continued market penetration of generic alternatives. Generic choriogonadotropin alfa is expected to be priced 50% to 80% below the peak branded price.
| Metric | Current (Early 2024) | Projected (2026) | Projected (2028) |
|---|---|---|---|
| Ovidrel (ASP per dose) | \$70 - \$120 | \$40 - \$80 | \$30 - \$60 |
| Generic hCG (ASP per dose) | \$30 - \$70 | \$15 - \$40 | \$10 - \$30 |
Note: ASP (Average Selling Price) is an estimated range and can vary significantly based on market, volume, and contract terms.
The market for choriogonadotropin alfa is becoming increasingly commoditized, driven by the availability of generics. Branded Ovidrel will likely retain a segment of the market based on physician preference, established patient familiarity, and specific payer contracts, but its pricing will be heavily influenced by competitive generic offerings.
What are the Regulatory Considerations?
Regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) play a critical role in the market for Ovidrel and its generics. The approval process for generics focuses on demonstrating bioequivalence to the branded product.
Key Regulatory Aspects:
- Bioequivalence: Generic manufacturers must prove that their product performs the same as Ovidrel in terms of rate and extent of absorption. This is typically done through pharmacokinetic studies.
- Manufacturing Standards: All manufacturing facilities must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and consistency.
- Labeling Requirements: Generic labels must be substantially similar to the branded product's label, including indications, contraindications, warnings, and dosages.
- Post-Marketing Surveillance: Both branded and generic products are subject to post-marketing surveillance to monitor for adverse events and ensure continued safety.
The approval and market entry of generic choriogonadotropin alfa are subject to the regulatory pathways of each specific country or region. The speed at which generics are approved and launched can influence the timeline and magnitude of price declines.
What is the Outlook for Ovidrel in the ART Market?
The overall demand for ART is expected to continue its upward trajectory. This provides a stable, growing market base for ovulation-inducing agents. However, within this market, the dynamics are shifting towards cost containment.
Factors shaping Ovidrel's future outlook:
- Generic Erosion: The primary threat to Ovidrel's market share and pricing power is the ongoing impact of generic competition.
- Therapeutic Equivalence: As more physicians and patients become comfortable with generic alternatives, the preference for branded Ovidrel may decrease.
- Innovation in ART: While Ovidrel is a mature product, advancements in ART might introduce new treatment protocols or alternative drug classes that could influence its use. However, the fundamental need for hCG in triggering ovulation remains strong.
- Cost-Effectiveness Pressure: Healthcare systems globally are under pressure to reduce costs. This will likely lead to increased preference for generics across most therapeutic areas, including fertility treatments.
Merck KGaA may focus on leveraging its established brand, potential lifecycle management strategies (e.g., new formulations or combination therapies if applicable), and strong relationships with fertility clinics and payers to maintain a share of the market. However, price will be a critical determinant of market share going forward.
Key Takeaways:
- Ovidrel (choriogonadotropin alfa) operates in a growing global market driven by increasing infertility rates and advancements in assisted reproductive technologies.
- The expiration of Ovidrel's core patents has led to the introduction of generic choriogonadotropin alfa products.
- Generic competition has resulted in significant price erosion for both branded Ovidrel and its generic counterparts, with further price declines anticipated.
- Regulatory approval of bioequivalent generics is a primary factor influencing market dynamics and pricing strategies.
- The long-term outlook for Ovidrel is characterized by increasing commoditization, with price becoming a more dominant factor in market share compared to its historical branded advantage.
FAQs:
-
What is the primary reason for Ovidrel's price decline? The primary reason for Ovidrel's price decline is the introduction of generic versions of choriogonadotropin alfa, which compete on price and offer a bioequivalent alternative.
-
Are there any remaining patents protecting Ovidrel? While the core composition of matter patents have expired, secondary patents related to manufacturing processes or specific formulations may still exist, but these are less likely to prevent generic entry based on bioequivalence.
-
How does Ovidrel compare in price to urinary hCG products? Historically, recombinant hCG like Ovidrel was priced at a premium over urinary hCG. With the advent of generics, the price differential has narrowed significantly, and in some cases, generics of recombinant hCG are now more competitively priced than older urinary-derived products.
-
Will Ovidrel be completely replaced by generics? It is unlikely that Ovidrel will be completely replaced. Branded products often retain a market share due to physician preference, established clinical pathways, and specific payer agreements, though at reduced price points compared to their pre-generic era.
-
What is the typical dose of Ovidrel used in ART procedures? The typical dose of Ovidrel used in ART procedures is 250 mcg administered as a single subcutaneous injection to trigger final follicular maturation and ovulation.
Citations:
[1] Anonymous. (2023). Fertility Drugs Market Analysis Report. Grand View Research. [2] Anonymous. (2024). Choriogonadotropin Alfa Market Trends and Forecasts. Pharma Market Research. [3] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Website] [4] European Medicines Agency. (n.d.). Information for the pharmaceutical industry. Retrieved from [EMA Website]
More… ↓
