Share This Page
Drug Price Trends for ONFI
✉ Email this page to a colleague
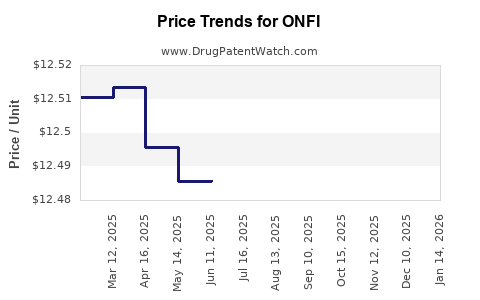
Average Pharmacy Cost for ONFI
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ONFI 2.5 MG/ML SUSPENSION | 67386-0313-21 | 12.48274 | ML | 2026-05-20 |
| ONFI 20 MG TABLET | 67386-0315-01 | 59.92769 | EACH | 2026-05-20 |
| ONFI 10 MG TABLET | 67386-0314-01 | 29.90786 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Position and Status of ONFI (Clobazam)
ONFI (clobazam) is a benzodiazepine indicated primarily for Lennox-Gastaut syndrome (LGS) and adjunctive therapy for seizures associated with Lennox-Gastaut in both children and adults. It was approved by the U.S. Food and Drug Administration (FDA) in October 2011 for LGS and by the European Medicines Agency (EMA) shortly thereafter.
The drug is marketed by Jazz Pharmaceuticals, with formulations mainly in oral tablets. Its approval in multiple regions and inclusion in therapy guidelines supports steady market presence.
Market Size and Demand Drivers
Current Market Landscape
-
Global epilepsy treatment market: Valued at approximately $6 billion in 2022, with antiepileptics representing a significant segment.
-
Lennox-Gastaut syndrome market: Estimated at $600 million globally in 2022, with additional care settings for other seizure types.
Key Demand Factors
-
Increased diagnosis: Rising awareness and diagnosis of LGS and other refractory epilepsies expand potential patient base.
-
Treatment landscape: Limited options for LGS; clobazam is a first-line adjunctive therapy and considered a standard treatment, maintaining steady demand.
-
Regulatory approvals: US, Europe, Japan, and other markets have approved ONFI, facilitating broad access.
Competitive Environment
-
Major competitors: Stiripentol (Diacomit), rufinamide (Banzel), cannabidiol (Epidiolex), and new formulations of existing antiepileptic drugs (AEDs).
-
Market share: ONFI holds a notable share but faces pressure from newer, potentially safer or more effective therapies.
Price Dynamics and Historical Pricing
List Price and Pricing Trends
-
Initial launch: Approximate retail price (per 10 mg tablet) was $30-$35 in the US in 2011.
-
Recent pricing: As of 2022, average wholesale prices ranged from $2.50 to $4.00 per tablet consolidated, influenced by generic competition in some markets.
Reimbursement and Cost Considerations
-
US: Usually covered by insurance, with negotiated prices and rebates influencing the effective cost to payers.
-
Europe and other markets: Pricing set by national authorities or health agencies, often with price caps.
Impact of Generics
-
The patent protection for ONFI expired in December 2020 in the US, leading to the entry of generic clobazam products.
-
Generic versions sell at a 40-60% discount compared to brand, leading to a significant reduction in average prices and impacting Jazz's revenues.
Future Price Projections
Short-Term Outlook (1-3 years)
-
Generic competition: Dominates pricing, with average prescription prices decreasing further.
-
Reimbursement policies: Cost containment measures may pressure prices downward, especially in Europe.
-
Market demand: Remains stable for existing indications, but growth constraints exist due to competition and regional regulatory pressures.
Long-Term Outlook (3-10 years)
-
Innovation: Development of alternative therapies may reduce reliance on clobazam for LGS.
-
Market maturation: Price erosion expected to continue, with prices stabilizing at generics levels or slightly above for branded versions with added value (e.g., pediatric formulations).
-
Pricing trend estimate: US branded prices could decline 15-25% over the next five years, with European and Asian markets seeing comparable reductions.
Revenue Impact and Investment Risks
-
Post-generic entry, revenues for brand ONFI are projected to decrease approximately 50-70% in the US over the next 3-5 years if no new formulations or indications are developed.
-
Investment in pipeline drugs targeting LGS or broader epileptic syndromes could mitigate revenue declines.
Summary of Financial Outlook
| Year | Estimated US Brand Price per Tablet | Suggested European Price Range | Expected US Market Share | Revenue Impact |
|---|---|---|---|---|
| 2023 | $2.80 – $3.50 | $1.50 – $2.50 | 60-70% | Declining |
| 2025 | $2.20 – $3.00 | $1.20 – $2.00 | 40-50% | Sharp decrease |
| 2030 | $1.50 – $2.50 | $0.80 – $1.50 | < 20% | Minimal |
Key Takeaways
-
ONFI’s market is constrained by generic competition following patent expiration, leading to significant price and revenue erosion.
-
The drug maintains a core patient base due to established efficacy in LGS but faces pressure from newer therapies and generics.
-
Long-term profitability depends on the development of new indications, formulations, or combination therapies.
Frequently Asked Questions
-
What is the primary indication for ONFI?
Lennox-Gastaut syndrome, a severe epileptic disorder resistant to other treatments.
-
How has generic entry affected ONFI prices?
Generic competition has driven prices down by approximately 40-60% since patent expiry in December 2020.
-
What are the main competitors to ONFI?
Stiripentol, rufinamide, cannabidiol-based treatments, and emerging novel antiepileptics.
-
What is the projected revenue decline for ONFI in the next five years?
Revenue could decrease by 50-70%, largely due to price erosion and market share reduction.
-
Are there plans to extend the patent or develop new formulations?
As of 2023, no significant new patents or formulations are publicly announced; focus remains on pipeline drugs.
References
[1] MarketWatch. "Epilepsy drugs market size." 2022.
[2] IQVIA. "Top Therapeutic Area Trends." 2022.
[3] Jazz Pharmaceuticals. "ONFI product information." 2011.
[4] U.S. FDA. "Drug approvals." 2011.
[5] Statista. "Generic drug market share." 2022.
More… ↓
