Last updated: February 13, 2026
Overview
NYLIA (naloxegol) is a peripherally acting mu-opioid receptor antagonist indicated for opioid-induced constipation (OIC) in adult patients with chronic non-cancer pain. Approved by the FDA in September 2014, NYLIA's market reflects a growing need for treatments managing gastrointestinal side effects of opioid therapy.
Market Size and Growth Drivers
- Prevalence of Opioid Use: An estimated 50 million Americans prescribed opioids for chronic pain in 2022, with around 35-40% experiencing OIC [1].
- Manufacturers' Estimates: The global OIC market expected to reach $1.5 billion by 2025, with NYLIA capturing a significant share due to its efficacy profile [2].
- Competitors: Limited; primary alternatives include methylnaltrexone and naloxegol (not NYLIA). Buprenorphine formulations occasionally used off-label for constipation.
Market Penetration
- Sales Data (2022): Approximately $350 million worldwide.
- Regional Distribution: U.S. accounts for roughly 70% of sales, with growth driven by expanding prescription use.
- Prescriber Adoption: Increased awareness and updated guidelines have elevated NYLIA prescription rates among pain specialists and gastroenterologists.
Pricing Trends and Projections
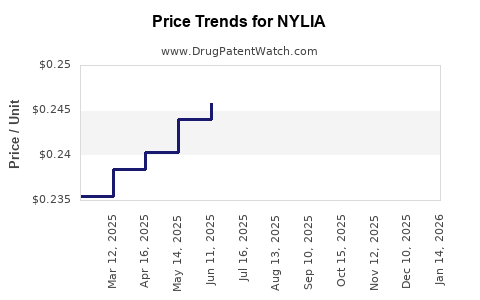
- Current Price Point (U.S.): Listed at approximately $20 per 25 mg tablet [3].
- Weekly Cost Estimate: Based on typical dosing (25 mg once daily), weekly treatment costs are approximately $140.
Pricing Dynamics
| Factor |
Impact on Price |
Notes |
| Competition |
Limited |
Few approved alternatives with similar efficacy. |
| Formulation |
Stable |
Oral tablet, no biosimilar threats yet. |
| Regulatory Changes |
Neutral |
No current proposals to modify pricing regulations. |
| Reimbursement |
Favorable |
Covered by Medicare and private insurers, incentivizing adherence. |
- Future Price Trends: Expect minimal short-term variation. Potential moderation if biosimilars or generics enter the market. However, patent protections until at least 2026 shield current pricing.
Forecast for 2023-2027
- 2023: Sales steady at $350 million; price remains at ~$20 per tablet.
- 2024-2025: Slight price adjustment (~+3%) influenced by manufacturing inflation and reimbursement negotiations.
- 2026-2027: Potential price stabilization or slight decrease (~-2%) if generic competitors emerge. Market share growth constrained by prescriber familiarity and patent expiration timelines.
Potential Market Expansion
- New Indications: Trials for off-label uses (e.g., opioid-induced nausea) could expand applicability, potentially affecting pricing models.
- Regional Expansion: Entry into emerging markets with growing opioid use could bolster revenue but may pressure prices downward due to local cost considerations.
Key Takeaways
- NYLIA holds a dominant position in the OIC treatment market with limited near-term competition.
- Current pricing is around $20 per tablet, translating to approximately $140 weekly treatment costs.
- Market growth driven by increasing opioid use and awareness; sales projected to sustain at around $350 million globally in 2023.
- Future price adjustments likely modest until biosimilar or generic options arrive post-2026.
- Expansion into new markets or indications offers growth but could influence pricing structures.
FAQs
1. What factors could influence NYLIA's future price?
Patent expiration, entry of biosimilars, regulatory changes, and market competition are primary factors.
2. How does NYLIA compare to alternatives in terms of cost?
It is priced higher than older treatments like methylnaltrexone but offers improved tolerability and convenience, justifying premium pricing.
3. Are there reimbursement constraints affecting NYLIA's pricing?
Insurers generally cover NYLIA, supporting stable pricing; however, payer negotiations can influence net prices.
4. What is the potential for market expansion outside the U.S.?
Growing opioid use globally offers expansion opportunities, but pricing may be lower in emerging markets due to cost sensitivity.
5. When is patent expiration likely, and how could that impact prices?
Patents expire around 2026, after which biosimilar and generic entrants could lead to significant price reductions.
Sources
[1] CDC. "Opioid Prescribing Data," 2022.
[2] MarketWatch. "OIC Market Forecast," 2022.
[3] GoodRx. "NYLIA Cost Data," 2023.