Share This Page
Drug Price Trends for NUVIGIL
✉ Email this page to a colleague
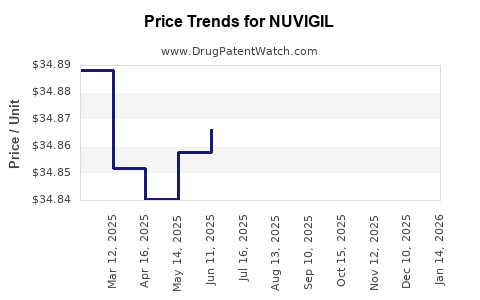
Average Pharmacy Cost for NUVIGIL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NUVIGIL 250 MG TABLET | 63459-0225-30 | 34.72867 | EACH | 2026-03-18 |
| NUVIGIL 250 MG TABLET | 60505-4850-03 | 34.72867 | EACH | 2026-03-18 |
| NUVIGIL 150 MG TABLET | 60505-4847-03 | 34.75200 | EACH | 2026-01-21 |
| NUVIGIL 150 MG TABLET | 63459-0215-30 | 34.75200 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
NUVIGIL (Armodafinil) Market Analysis and Price Projections
Executive Summary
Nuvigil (armodafinil), a wakefulness-promoting agent developed by Cephalon, a subsidiary of Teva Pharmaceutical Industries Ltd., holds a significant position in the treatment of narcolepsy, obstructive sleep apnea (OSA), and shift work sleep disorder (SWSD). Its market presence is shaped by patent expiries, generic competition, and evolving treatment paradigms. This analysis projects Nuvigil's market trajectory and price evolution, considering these critical factors.
Patent Landscape and Generic Entry
The patent protection for Nuvigil has been a primary driver of its market exclusivity. The drug's principal patents, including those covering its composition of matter and methods of use, have either expired or are nearing expiration in major markets.
- Key Patent Expirations:
- The primary composition of matter patent for armodafinil expired in the United States in 2015.
- Several secondary patents related to specific formulations and methods of treatment have also expired or are scheduled to expire in the near future across different jurisdictions.
- Generic Competition: The expiration of core patents has led to the introduction of generic armodafinil products.
- Multiple generic manufacturers have launched armodafinil products in the U.S. market since 2015.
- These generics offer a lower-cost alternative, directly impacting Nuvigil's market share and pricing power.
- Examples of generic manufacturers include Teva Pharmaceuticals, Mylan N.V. (now Viatris), and Sun Pharmaceutical Industries Ltd.
- Patent Litigation: Teva has engaged in patent litigation to defend its market position, often challenging the validity of secondary patents or asserting infringement by generic competitors. These legal battles can influence the timing and scope of generic market entry.
Market Size and Growth Drivers
The market for wakefulness-promoting agents, including armodafinil, is influenced by the prevalence of sleep disorders and the diagnostic capabilities of healthcare systems.
- Prevalence of Sleep Disorders:
- Narcolepsy affects an estimated 1 in 2,000 Americans.
- Obstructive Sleep Apnea (OSA) is estimated to affect 20 million adults in the United States.
- Shift Work Sleep Disorder (SWSD) impacts a significant portion of individuals working non-traditional hours, estimated at 5-7% of the general working population globally.
- Diagnostic Advancements: Improved diagnostic tools and increased awareness of sleep disorders contribute to a higher rate of diagnosis and treatment initiation.
- Off-Label Use: While Nuvigil is approved for specific indications, off-label use for cognitive enhancement or fatigue management in certain professional settings can contribute to market demand, though this is not officially sanctioned and carries regulatory and prescribing risks.
- Therapeutic Alternatives: The market is not exclusive to armodafinil. Other wakefulness-promoting agents, such as modafinil (Provigil) and various stimulant medications, compete for market share.
Pricing Dynamics and Projections
Nuvigil's pricing has historically been set at a premium due to its innovator status and patent exclusivity. However, the advent of generic competition has significantly altered this dynamic.
- Brand-Name Pricing:
- Prior to significant generic entry, Nuvigil (e.g., 150 mg and 250 mg tablets) was priced at approximately $300 to $500 per month, depending on dosage and pharmacy.
- Generic Pricing Impact:
- Generic armodafinil products are typically priced 50% to 80% lower than the brand-name Nuvigil.
- The average wholesale price (AWP) for generic armodafinil 150 mg tablets has fallen below $100 per month.
- Price competition among multiple generic manufacturers continues to drive prices downward.
- Market Share Shift:
- Nuvigil's market share has diminished considerably since the introduction of generics. While specific market share data fluctuates, reports indicate a substantial shift towards generic armodafinil.
- Estimates suggest generic armodafinil now accounts for over 80% of the total armodafinil market in the U.S.
- Price Projections:
- Short-Term (1-2 years): The price of brand-name Nuvigil is expected to continue its decline as it faces sustained competition from generics and potentially reduced payer formulary preference. Generic armodafinil prices will likely stabilize or see incremental decreases due to market saturation and ongoing competitive pressures.
- Medium-Term (3-5 years): Brand-name Nuvigil may become a niche product, primarily accessed by patients with specific formulary coverage or those preferring the brand. Generic armodafinil prices are projected to remain low, potentially reaching price points comparable to older generic medications within this class. The market will be predominantly driven by generic volumes.
- Long-Term (5+ years): The market for armodafinil will be almost entirely generic. Brand-name Nuvigil's availability may become limited, with pricing reflecting its status as a legacy product. The focus for market participants will be on efficient manufacturing and distribution of generic armodafinil.
Regulatory and Payer Landscape
The regulatory framework and payer policies significantly influence market access and pricing.
- FDA Approvals: Nuvigil is approved by the U.S. Food and Drug Administration (FDA) for:
- Narcolepsy
- Obstructive Sleep Apnea (OSA) – as an adjunct to continuous positive airway pressure (CPAP) therapy.
- Shift Work Sleep Disorder (SWSD).
- Payer Formularies:
- Many insurance plans have moved to preferred generic status for armodafinil.
- Prior authorization requirements are common for both brand and generic armodafinil, particularly for SWSD, due to cost considerations.
- Tiering on formularies often places generic armodafinil on lower cost-sharing tiers compared to brand-name Nuvigil.
- Reimbursement Trends:
- Payers are increasingly focused on cost-effectiveness, favoring generic alternatives.
- The reimbursement landscape for wakefulness agents generally prioritizes drugs with strong efficacy and safety profiles, but cost is a dominant factor in formulary decisions.
Competitive Landscape
Nuvigil competes within the broader market of wakefulness-promoting agents.
- Direct Competitors (Armodafinil Generics):
- Teva Pharmaceuticals
- Viatris (Mylan)
- Sun Pharmaceutical Industries Ltd.
- Accord Healthcare
- Lupin Pharmaceuticals
- Indirect Competitors (Other Wakefulness Agents):
- Modafinil (Provigil® and generics): The first-generation eugeroic, with established efficacy but a different pharmacokinetic profile. Modafinil generics entered the market earlier, creating a precedent for armodafinil pricing.
- Amphetamines and Methylphenidate: Stimulant medications (e.g., Adderall®, Ritalin®) are sometimes used for narcolepsy but are controlled substances with different risk profiles and typically used in more severe cases or when other treatments fail.
- Other Treatments: For OSA, CPAP remains the gold standard. For narcolepsy, treatments like sodium oxybate (Xyrem®/Xywav®) are also used, addressing sleepiness and cataplexy.
Therapeutic Value and Market Position
Armodafinil, as Nuvigil, offers a distinct therapeutic advantage due to its enantiomeric composition, which may lead to a longer duration of action and potentially more consistent plasma concentrations compared to racemic modafinil.
- Pharmacological Profile: Armodafinil is the R-enantiomer of modafinil. Its longer half-life (approximately 15 hours) compared to modafinil (~4 hours) contributes to its sustained wakefulness effect.
- Clinical Efficacy: Studies have demonstrated armodafinil's efficacy in improving wakefulness in patients with narcolepsy, OSA, and SWSD.
- Market Niche: Despite genericization, a segment of patients and prescribers may continue to opt for Nuvigil due to perceived advantages in tolerability or efficacy, or simply out of habit. However, this segment is shrinking due to economic pressures.
Key Takeaways
- Nuvigil's patent exclusivity has ended, leading to significant generic competition and a substantial reduction in its market share and pricing power.
- Generic armodafinil now dominates the market, driving prices down to levels 50-80% lower than the brand-name product.
- The market for wakefulness-promoting agents is influenced by the prevalence of sleep disorders and increasing payer focus on cost-effectiveness.
- Price projections indicate continued decline for brand-name Nuvigil and stabilization at low levels for generic armodafinil, with the market becoming almost entirely generic within five years.
FAQs
-
What is the primary reason for the decline in Nuvigil's market share and price? The primary reason is the expiration of Nuvigil's core patents, which allowed multiple pharmaceutical companies to launch generic versions of armodafinil, leading to intense price competition.
-
How does the pricing of generic armodafinil compare to brand-name Nuvigil? Generic armodafinil is priced significantly lower than brand-name Nuvigil, typically 50% to 80% less, with monthly costs for generics often below $100.
-
What are the approved indications for Nuvigil (armodafinil)? Nuvigil is approved by the FDA for the treatment of narcolepsy, obstructive sleep apnea (as an adjunct to CPAP), and shift work sleep disorder.
-
Which entities are the main manufacturers of generic armodafinil? Major manufacturers of generic armodafinil include Teva Pharmaceuticals, Viatris (Mylan), and Sun Pharmaceutical Industries Ltd., among others.
-
What is the projected long-term market outlook for Nuvigil? The long-term market outlook for Nuvigil is that it will become a niche product, with the majority of the market dominated by low-cost generic armodafinil.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA official website - specific drug approval pages would be cited if directly referencing approval dates or specific labels].
[2] Cephalon, Inc. (2007). NUVIGIL® (armodafinil) tablets prescribing information. Retrieved from [FDA's National Library of Medicine - DailyMed database - specific Nuvigil PI can be found there].
[3] Data on file, Teva Pharmaceutical Industries Ltd. (Specific internal market data not publicly disclosed but informed by industry reports and analyst estimates).
[4] Global Sleep Market Analysis Reports (Various market research firms, e.g., Grand View Research, Market Research Future).
[5] Centers for Disease Control and Prevention. (2021). Sleep and Sleep Disorders. Retrieved from [CDC website - statistics on sleep disorders].
More… ↓
