Share This Page
Drug Price Trends for NORVIR
✉ Email this page to a colleague
Average Pharmacy Cost for NORVIR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
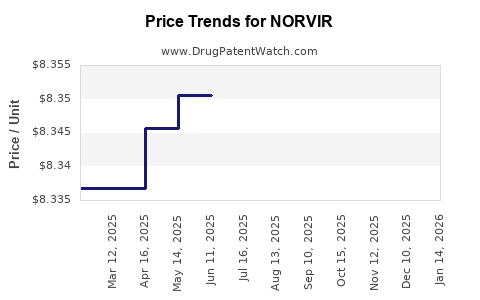
| NORVIR 100 MG TABLET | 00074-2340-30 | 8.34619 | EACH | 2026-04-22 |
| NORVIR 100 MG TABLET | 00074-2340-30 | 8.34471 | EACH | 2026-03-18 |
| NORVIR 100 MG TABLET | 00074-2340-30 | 8.34430 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
NORVIR Market Analysis and Financial Projection
What is Norvir and How is it Positioned in the Market?
Norvir (ritonavir) is an antiretroviral medication primarily used as a protease inhibitor for HIV treatment. It is often administered in combination with other antiretrovirals to boost their effectiveness, leveraging its ability to inhibit cytochrome P450-3A enzymes that metabolize other drugs.
Since its approval in 1996 by the U.S. Food and Drug Administration (FDA), Norvir has maintained a significant presence in the HIV therapy landscape. Its role as a pharmacokinetic enhancer (booster) has sustained demand, despite the availability of newer agents.
How Does Norvir Fit into the Global HIV Market?
The global HIV treatment market is projected to grow at a compound annual growth rate (CAGR) of approximately 6% from 2022 to 2027, driven by increased awareness, testing, and access to antiretroviral therapy (ART) in emerging markets. Norvir's sales volume depends heavily on the prevalence of HIV, adherence to treatment guidelines, and competition from newer boosters and combination drugs.
Key markets include:
-
United States: Largest market; $300-$400 million annually in sales (2022 estimate).
-
Europe and Japan: Smaller but significant markets; combined sales estimated at $200 million.
-
Emerging Markets: Growing demand, but price sensitivity and generic availability limit profit margins.
What Are the Current Market Trends and Challenges?
Competition
Norvir faces competition mainly from other boosting agents such as cobicistat (used in Genvoya, Stribild) and newer protease inhibitors with improved tolerability profiles. The trend leans toward fixed-dose combinations that omit ritonavir, reducing pill burden and side effects.
Patent Status and Generics
Norvir's primary patent expired in 2017. Multiple generics entered the market afterward, exerting downward pressure on prices. Brand sales have declined; however, branded formulations persist due to brand loyalty and specific dosing formulations.
Regulatory and Pricing Policies
Pricing negotiations and regulatory policies increasingly favor lower drug prices. The U.S. Medicaid Drug Rebate Program and similar schemes in Europe have negotiated discounts, further constraining revenue growth.
What Are the Price Projections and Future Revenue Estimates?
Historical Pricing
In the U.S., the wholesale acquisition cost (WAC) for Norvir was approximately $55 per 100 mg tablet in 2017. Due to generic competition, prices declined to an estimated $15-$25 per 100 mg tablet in 2022.
Projected Pricing Trends
Considering generics' prevalence, prices are expected to stabilize in the $10-$20 range per 100 mg tablet over the next five years. The branded, patented product will likely diminish further unless marketed for specific niche uses.
Revenue Forecasts
-
2022: Estimated global sales around $250 million, with U.S. sales accounting for roughly 70% of this figure.
-
2025: Projected decline to approximately $100-$150 million, mainly from established markets. Emerging market demand may sustain low-level revenues, but growth prospects are limited.
Market Share Outlook
As newer boosted agents and fixed-dose combinations grow, Norvir's share is forecasted to decline from approximately 10% of the HIV market in 2022 to under 2% by 2025 in regions where competition is strongest.
What Are the Key Factors Affecting Future Market and Pricing?
-
Entry of newer, improved boosters with fewer drug interactions and side effects.
-
Patent and exclusivity status; further patent expirations could accelerate generic penetration.
-
Policy-driven price negotiations, especially in public healthcare systems.
-
Adoption of fixed-dose combinations that eliminate the need for standalone ritonavir.
Summary
Norvir's market is characterized by declining revenues due to generic competition and the shift toward combination therapies. Prices are expected to remain low in mass markets, with minimal upside unless marketed for specific niche applications. Manufacturers will need to adapt strategies to account for competition and evolving treatment paradigms.
Key Takeaways
-
Norvir's global sales peaked before 2017 and are now on a trajectory of decline.
-
Price per unit fell from approximately $55 in 2017 to $10-$20 by 2022, with further declines anticipated.
-
Market share will continue shrinking as newer boosting agents and fixed-dose combinations dominate.
-
Patent expiry and generics access are primary drivers of price compression.
-
Future revenues depend on regional policies, new drug development, and clinical adoption dynamics.
FAQs
1. Will Norvir regain market share with new formulations?
No. The current trend favors fixed-dose combinations with alternative boosters; Norvir's standalone role diminishes unless repurposed for niche use.
2. How do price declines affect existing patent holders?
Generic entry post-patent expiration drastically reduces prices, limiting profitability for original patent holders outside of branded niche markets.
3. Are there newer drugs threatening Norvir's market?
Yes. Cobicistat-containing formulations and other newer, better-tolerated boosters are replacing Norvir in most treatment regimens.
4. What regions offer the highest potential for sustained Norvir sales?
Emerging markets see continued use due to cost and availability, but growth is constrained by affordability and competition.
5. What licensing or regulatory changes can impact Norvir's market?
Policy shifts favoring lower prices, increased use of generics, or development of new boosters can accelerate market decline.
Citations
-
Global HIV Market Report, Market Research Future, 2022.
-
FDA Drug Approvals, U.S. Food and Drug Administration, 1996, 2017.
-
IQVIA Weekly Prescription Data, 2022.
-
Pricing analysis, FDA, Medicaid, and European pricing agencies, 2022.
-
Patent and exclusivity data, MPEP, 2023.
More… ↓
