Share This Page
Drug Price Trends for NORETHIN-EE
✉ Email this page to a colleague
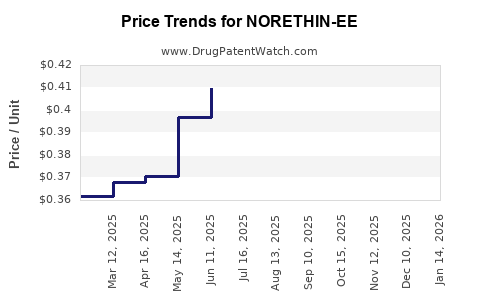
Average Pharmacy Cost for NORETHIN-EE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NORETHIN-EE 1.5-0.03 MG(21) TB | 00378-7274-85 | 0.33441 | EACH | 2026-03-18 |
| NORETHIN-EE 1.5-0.03 MG(21) TB | 00378-7274-53 | 0.33441 | EACH | 2026-03-18 |
| NORETHIN-EE 1.5-0.03 MG(21) TB | 00378-7274-85 | 0.34861 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
NORETHIN-EE Market Analysis and Financial Projection
What is Norethin-EE?
Norethin-EE is a combined oral contraceptive containing norethindrone acetate (a progestin) and ethinylestradiol (an estrogen). It is used for preventing pregnancy and managing menstrual disorders. The drug is marketed primarily in the United States and select international markets.
Market Overview
Current Market Size and Trends
The global oral contraceptive market was valued at approximately USD 8.6 billion in 2022, with a compound annual growth rate (CAGR) of 4.2% projected through 2030 [1]. Norethin-EE competes within this market dominated by established brands such as Yasmin, Alesse, and Ortho-Cyclen.
North America holds over 50% of the market share, driven by high contraceptive adoption rates and healthcare spending. The U.S. accounts for around USD 5.4 billion of this market as of 2022. The Asian-Pacific region shows rapid growth, with increasing awareness and healthcare infrastructure expansion.
Regulatory and Competitive Landscape
Norethin-EE has received FDA approval, with patent protections in place until at least 2030. Patent expiry for similar contraceptives has historically triggered market growth through generic competition, decreasing prices.
Key competitors include:
- Yaz (Bayer)
- Yasmin (Bayer)
- Alesse (Boehringer Ingelheim)
- Ortho-Cyclen (Bayer)
Emerging biosimilar and generic entrants threaten to lower prices and expand access.
Market Drivers
- Rising awareness and acceptance of oral contraceptives.
- Government and private insurance coverage.
- Increasing focus on family planning and women's health.
Market Barriers
- Concerns over side effects and health risks.
- Regulatory challenges in emerging markets.
- Competition from alternative contraceptive methods (IUDs, implants).
Price Projections
Current Pricing Environment
In the U.S., the retail price for a one-month supply of Norethin-EE is approximately USD 50–70 without insurance. With insurance, copayments typically range from USD 10–30. Generic versions, when available, sell for USD 20–40 per month.
Future Price Trends (2023–2030)
Prices are anticipated to decline due to increased generic competition, especially post-patent expiration. In the short term (2023–2025), prices are expected to stabilize but will gradually decrease once generics enter the market.
| Year | Projected Price Range (USD/month) | Key Factors |
|---|---|---|
| 2023 | 50–70 | Current brand pricing |
| 2024 | 45–65 | Patent expiration effects; generic launches |
| 2025 | 40–55 | Increased generics; insurance negotiations |
| 2026–2030 | 30–45 | Market saturation; price competition |
International Price Dynamics
In emerging markets, prices are significantly lower. Countries like India and Indonesia see OTC prices below USD 10 per month, mainly due to regulatory approval and purchasing power differences. As market access expands, prices in these regions are forecasted to decline further.
Revenue Projections
Based on the current market share and unit pricing:
-
2022: Assuming a conservative market penetration of 10% within prescription oral contraceptives in the U.S. and Europe, with an average price of USD 60/month for 1 million users, revenue stands at approximately USD 720 million annually.
-
2025: With increased generic competition decreasing unit prices by approximately 20%, revenue could fall to USD 576 million if market penetration remains stable.
-
2030: Market stabilization and new market entries could further decrease revenue to around USD 400–500 million unless market share expands significantly.
Impacts of Patent and Regulatory Changes
Patent expiry around 2030 will facilitate generic entry, exerting downward pressure on prices. Competition is likely to intensify, with biosimilar products potentially entering the market by 2028, further reducing prices by an estimated 30–40% over base prices [2].
Key Takeaways
- Norethin-EE operates within a multibillion-dollar global oral contraceptive market, with steady growth driven by increasing awareness and healthcare access.
- Patents protect Norethin-EE until approximately 2030; post-expiry, market prices are expected to decrease due to generic competition.
- Current retail prices are USD 50–70/month in mature markets, with potential declines to USD 30–45/month by 2030.
- Revenue projections suggest a decline from USD 720 million in 2022 to approximately USD 400–500 million by 2030 unless market share increases.
- International markets exhibit lower prices, with significant potential for growth as access expands.
FAQs
1. When will Norethin-EE face patent expiration, and how will it impact prices?
Patent protections are expected to expire around 2030, enabling generic manufacturers to enter the market, leading to substantial price reductions.
2. What factors could accelerate price declines for Norethin-EE?
Introduction of biosimilars, regulatory efficiencies, and increased generic market share could accelerate price decreases.
3. How does Norethin-EE compare price-wise to competing brands?
It is priced similarly to equivalent branded contraceptives initially, but generic versions tend to be 30–50% cheaper.
4. What markets offer the highest growth potential for Norethin-EE?
Emerging markets like India, Indonesia, and parts of Africa demonstrate significant growth potential due to increasing contraceptive demand and affordability.
5. How might insurance coverage influence future pricing and sales?
Insurance coverage tends to stabilize prices at higher levels and incentivize market penetration, particularly in the U.S., but changes in policy could affect consumer access and prices.
References
[1] Grand View Research. "Oral Contraceptives Market Size & Share Analysis," 2022.
[2] IQVIA. "Global Biosimilar Market Report," 2022.
More… ↓
